A case of anemia caused by combined vitamin B12 and iron deficiency manifesting as short stature and delayed puberty
Article information
Abstract
Anemia caused by vitamin B12 deficiency resulting from inadequate dietary intake is rare in children in the modern era because of improvements in nutritional status. However, such anemia can be caused by decreased ingestion or impaired absorption and/or utilization of vitamin B12. We report the case of an 18-year-old man with short stature, prepubertal sexual maturation, exertional dyspnea, and severe anemia with a hemoglobin level of 3.3 g/dL. He had a history of small bowel resection from 50 cm below the Treitz ligament to 5 cm above the ileocecal valve necessitated by midgut volvulus in the neonatal period. Laboratory tests showed deficiencies of both vitamin B12 and iron. A bone marrow examination revealed dyserythropoiesis and low levels of hemosiderin particles, and a cytogenetic study disclosed a normal karyotype. After treatment with parenteral vitamin B12 and elemental iron, both anemia and growth showed gradual improvement. This is a rare case that presented with short stature and delayed puberty caused by nutritional deficiency anemia in Korea.
Introduction
Nutritional deficiency anemia traditionally includes iron deficiency anemia (IDA), vitamin B12 (cobalamin) deficiency anemia, folate deficiency anemia, protein deficiency anemia, and pyridoxine deficiency anemia1). These key nutrients are thought to be important in growth and development. Of the various anemias, megaloblastic anemia resulting from vitamin B12 deficiency caused by inadequate dietary intake is rare in children in developed countries because of their good diet, but can also be caused by decreased ingestion, impaired absorption from the small intestine, or poor utilization, of vitamin B12. IDA caused by high iron requirements which can be treated by dietary supplementation is common in infancy, and is the most frequent nutritional deficiency anemia seen in children2). However, this anemia is relatively rare after 3 years of age.
A few case reports have been published on growth retardation caused by nutritional deficiency anemias, resulting from inadequate vitamin B12 and iron intake, in Korean children3). The purpose of the present report is to describe the case of an 18 year-old man who had short stature and delayed puberty caused by severe anemia associated with both vitamin B12 and iron deficiency.
Case report
An 18-year-old male patient was admitted to our institution with principal complaints of short stature, prepubertal sexual maturation, and exertional dyspnea, for 1 year. He was severely anemic, with a hemoglobin level of 3.3 g/dL. He had a history of small bowel resection from 50 cm below the Treitz ligament to 5 cm above the ileocecal valve, necessitated by midgut volvulus in the neonatal period. No other abnormal congenital or genetic condition was detected. After surgery he experienced short bowel syndrome (SBS) as a sequela, and total parenteral nutrition had been supplied, which was gradually replaced by enteral feeding. Chronic abdominal pain, intermittent diarrhea, and vomiting had persisted for several years but eventually improved. At 4 years of age, his feeding mode was entirely enteral, and he was lost to follow-up until his recent visit. His family history was unremarkable. He denied use of any abusive substance and was not sexually active.
Review of system on admission revealed long-lasting dizziness and dyspnea on exertion, and these symptoms had become aggravated in the past year. He also complained of early satiety during meals, but denied gastrointestinal symptoms such as abdominal pain, constipation, or diarrhea.
On physical examination, he was alert, but chronically ill-looking and very pale. His blood pressure was 113/77 mmHg, his pulse rate 95/min, his respiratory rate 20/min, and his body temperature 36.1℃. His height was 137.7 cm (below the 3rd percentile for his age) and his weight 28.9 kg (also below the 3rd percentile for his age). His pubic hair was of Tanner stage I, as was his genital development (testis volume <4 mL). Other physical and neurologic examination findings were unremarkable.
The results of initial laboratory studies are listed in Table 1. The complete blood count showed hemoglobin of 3.3 g/dL, hematocrit of 12.5%, mean corpuscular volume (MCV) of 86.2 fl, mean corpuscular hemoglobin (MCH) of 22.8 pg, red cell distribution width (RDW) of 22.2%, and reticulocyte count of 0.59% (reticulocyte production index 0.16). The white blood cell count was 4,900/mm3 with a differential count of 45% neutrophils, 47% lymphocytes, 7% monocytes, and 1% eosinophils. The platelet count was 182,000/mm3. Serum iron was 23 µg/dL (normal 50-170 µg/dL), TIBC was 534 µg/dL (normal 280-400 µg/dL), and serum ferritin was 2.5 ng/mL (normal 20-320 ng/mL). Serum vitamin B12 was 60 pg/mL (normal 211-911 pg/mL) and serum folic acid was 11.3 ng/mL (normal >5.4 ng/mL). Peripheral blood cell morphology (Fig. 1) showed normocytic hypochromic anemia, marked anisocytosis, mild poikilocytosis, hypersegmented neutrophils, and mild thrombocytopenia. Bone marrow aspiration and biopsy revealed normal cellularity (90%) with an myeloid : erythroid (M : E) ratio of 2:1. Dyserythropoiesis, including changes in megaloblastoid levels, decreased amounts of hemosiderin particles, and sideroblasts, were also found. The cytogenetic study revealed a normal 46,XY karyotype. Because our patient was of short stature and was sexually underdeveloped, we performed a hormonal evaluation. Luteinizing hormone (LH) and follicular stimulating hormone (FSH) levels were 3.1 mIU/mL (normal 1.5-9.0 mIU/mL) and 2.0 mIU/mL (normal 2.0-9.2 mIU/mL) respectively, both of which were at prepubertal levels. Testosterone was 0.34 ng/mL (normal 3.5-9.7 ng/mL), insulin-like growth factor-1 (IGF-1) 83.6 ng/mL (normal 239-630 ng/mL), and insulin-like growth factor binding protein (IGFBP-1) 3,565 ng/mL (normal 1,820-6,990 ng/mL). On simple chest radiography, the heart size was normal. Bone age was about 12 years by wrist x-ray (Fig. 2). Bone mineral density (BMD) measured by dual energy x-ray absorptiometry was very low, and the Z score (with respect to bone age) of the BMD of the lumbar spine was -3.9.
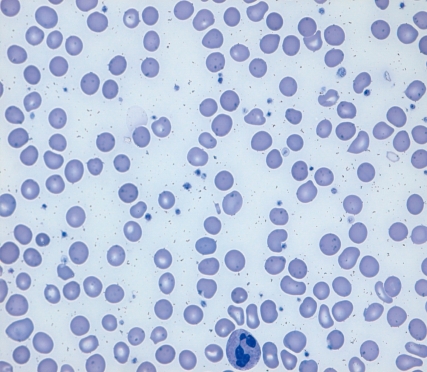
Peripheral blood smear shows hypersegmented neutrophils and normocytic hypochromic red blood cells (Wright stain, ×1,000 magnification).
Based on these findings, the patient was diagnosed with anemia caused by a combined vitamin B12 and iron deficiency attributable to impaired absorption caused by SBS. Parenteral vitamin B12 and iron treatment was initiated. Vitamin B12, as cobalamin, was administrated via intramuscular injection of 100 µg daily for 14 days, followed by 50 µg every other week. Our patient was also given 3 mg/kg elemental iron (ferric form) intravenously every other day for 14 days, followed by the same dose weekly. He was initially placed on a high potassium diet to avoid the transient hypokalemia that could result from rapid erythropoiesis.
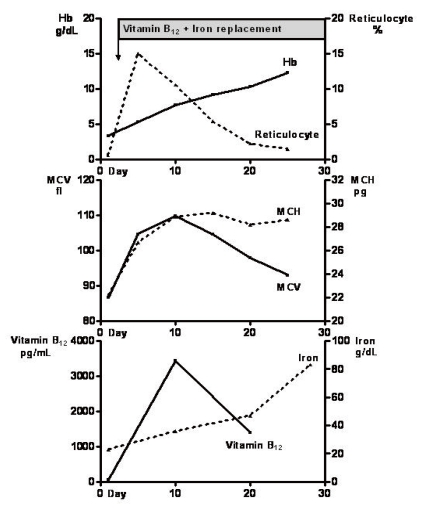
The hematologic indices responded immediately to therapy (Fig. 3). The reticulocyte count peaked at 14.9% on the 6th day of parenteral vitamin B12 and iron therapy. Ten days after treatment began, the hemoglobin concentration had increased to 7.7 g/dL, the hematocrit to 29.2%, vitamin B12 to 1,389 pg/mL, and serum iron to 47 µg/dL. He was discharged on the 10th day after admission with improvement in his dizziness and exertional dyspnea. After discharge, he was maintained on parenteral vitamin B12 and iron supplementation in the outpatient clinic. At 3 months after discharge, his hemoglobin level and hematocrit were 12.3 g/dL and 39%, respectively. Treatment was maintained for 17 months and the level of hemoglobin stabilized at around 12 g/dL. Two years after treatment began, the patient's weight and height had increased to 42.3 kg and 154.2 cm (below the 3rd percentile for his age). He has also attained puberty, with Tanner stage 4 pubic hair and a testicular volume of more than 10 mL.
Discussion
We present the case of a patient with anemia caused by combined vitamin B12 and iron deficiency, with a history of midgut volvulus that was operatively repaired in the neonatal period. This case demonstrates the importance of assessing growth status and nutritional status, including that of vitamins and minerals, in pediatric and adolescent populations with a history of intestinal surgery.
SBS is a malabsorptive state that may follow massive resection of the small intestine because the remaining small bowel absorptive surface is inadequate and enteral nutrient intake cannot support the body mass4, 5). SBS in children usually results from necrotizing enterocolitis and congenital intestinal anomalies, for which surgical resection of the involved intestinal segment is often the only treatment, as exemplified in our patient6).
The length and location of small intestine loss in SBS will generally define the degree of nutrient malabsorption and the likelihood of micronutrient deficiencies7). Duodenal resection, for example, can result in iron and folate deficiencies. Extensive jejunal resection can lead to malabsorption of carbohydrates, proteins, and calcium. The terminal ileum is the primary site of absorption of vitamin B12 and bile salts. Ileocolonic resection can predispose a patient to several problems as a consequence of the loss of the ileocecal valve, resulting in the absence of the ileal break, an increased risk of anastomotic ulcer, and bacterial overgrowth. The extent of nutrient malabsorption in SBS is generally assessed by history and physical examination, including measurement of growth parameters. Our patient experienced SBS after small bowel resection from 50 cm below the Treitz ligament to 5 cm above the ileocecal valve, in which almost the entire jejunoileum was included, and showed severe growth retardation and delayed puberty. As the absorption of iron and vitamin B12 takes place in the duodenum and upper jejunum, and terminal ileum, respectively, it is likely that malabsorption of both iron and vitamin B12 occurred in our patient.
Children with SBS are at the greatest risk for nutritional deficiencies in the interval during which parenteral nutrition support is being tapered, and also after discontinuation. Once parenteral nutrition has ceased, the level of micronutrient absorption is uncertain, and patients should be carefully monitored for nutrient deficiencies. It has been recommended that vitamin and mineral levels should be measured at the time of discontinuation, and every 3 months in the first year, and then as needed based on the requirement for supplementation8). As our patient had been lost to follow-up soon after parenteral nutrition was discontinued, his nutritional status could not be monitored and he became progressively malnourished. If he had received supplementation with vitamins and minerals earlier, he could have achieved a catch-up in growth.
Iron deficiency is the most common nutritional deficiency in children2), and dietary issues contribute significantly to the evolution of IDA in infancy and early childhood. Although typically presenting as a nutritional anemia, IDA may occur as part of a complex medical problem that includes gastrointestinal blood loss, malabsorption syndromes, and chronic inflammatory diseases9). IDA is also a confounding finding in anemia associated with chronic disease. The most common presentation of IDA involves an otherwise asymptomatic, well-nourished infant or child with mild anemia, but moderately severe IDA is associated with decreased work capacity, in part because iron is an essential cofactor for enzyme-driven aerobic metabolism. In our patient, a malabsorption state resulting from SBS was the cause of IDA, and the patient presented with symptoms of exertional dyspnea and dizziness.
Vitamin B12 is necessary for production of tetrahydrofolate, which is important in DNA synthesis. Animal products are the principal source of B12 in humans and gastrointestinal absorption occurs via a well-described mechanism10). The Intrinsic factor-cobalamin complex travels to the ileum where it binds to a specific receptor, and is finally absorbed through the ileal mucosa11). Any abnormality along this pathway can result in vitamin B12 malabsorption and deficiency. In non-vegetarian populations, one of the most common causes of vitamin B12 deficiency is specific malabsorption of vitamin B12 associated with pernicious anemia or gastric disease. Vitamin B12 deficiency is also common in patients who have undergone ileal resection. Vitamin B12 deficiency often presents with nonspecific manifestations, such as developmental delay, irritability, weakness, and failure to thrive, and is not easily detected by pediatricians12). Neurologic changes including paresthesias, sensory deficits, movement disorders, hypotonia, seizures, and neuropsychiatric changes can occur, without hematologic abnormality13, 14). Vitamin B12 deficiency appears to be associated with an increased risk of osteoporosis, possibly because of suppression of osteoblast activity15). Our patient had weakness, failure to thrive, and decreased BMD, but no neurologic symptoms.
The differential diagnoses in a patient with short stature, delayed puberty, and anemia include chronic renal failure, inflammatory bowel disease, inherited anemia, and congenital bone marrow failure syndrome16-19). Growth retardation has many causes in children. Although a growth hormone (GH) stimulation test was not performed, our patient was not considered to have any GH deficiency because there was a definitive cause of severe growth retardation, a long-lasting severe nutritional deficiency anemia attributable to SBS. Concomitant iron and vitamin B12 deficiency caused by malabsorption following small intestinal resection induced bone marrow erythroid megaloblastosis. Peripheral blood macroovalocytosis, frequently found in cases of vitamin B12 deficiency associated with megaloblastic anemia, was masked because of the countervailing tendency of iron deficiency to produce hypochromic microcytic erythrocytes.
Appropriate treatment results in dramatic clinical and laboratory responses in most patients. Our patient showed rapid and significant improvement in growth and hematologic recovery after cobalamin and iron treatment. His clinical response to parenteral injections of these supplements suggests that only vitamin B12 and iron were at inadequate levels. This is supported by the absence of hypoproteinemia, a normal prothrombin time, and no evidence of rickets. He has continued to grow steadily while receiving only parenteral vitamin B12 and iron, and thus appears to be absorbing other factors required for growth and development from his residual intestine.
In conclusion, we report a rare case of a patient who presented with short stature and delayed puberty caused by nutritional deficiency anemia, in Korea. Our observations illustrate the importance of the distal small intestine in absorption of vitamin B12 and iron. The data also support the conclusion that patients who have undergone resection of the distal small intestine should be closely monitored with respect to growth and hematologic parameters, and immediate supplementation in cases of deficiency should be provided to prevent severe anemia and subsequent growth retardation.