Acute tubular necrosis as a part of vancomycin induced drug rash with eosinophilia and systemic symptoms syndrome with coincident postinfectious glomerulonephritis
Article information
Abstract
Drug rash with eosinophilia and systemic symptoms (DRESS) syndrome is a rare and potentially fatal condition characterized by skin rash, fever, eosinophilia, and multiorgan involvement. Various drugs may be associated with this syndrome including carbamazepine, allopurinol, and sulfasalazine. Renal involvement in DRESS syndrome most commonly presents as acute kidney injury due to interstitial nephritis. An 11-year-old boy was referred to the Children's Hospital of Pusan National University because of persistent fever, rash, abdominal distension, generalized edema, lymphadenopathy, and eosinophilia. He previously received vancomycin and ceftriaxone for 10 days at another hospital. He developed acute kidney injury with nephrotic range proteinuria and hypocomplementemia. A subsequent renal biopsy indicated the presence of acute tubular necrosis (ATN) and late exudative phase of postinfectious glomerulonephritis (PIGN). Systemic symptoms and renal function improved with corticosteroid therapy after the discontinuation of vancomycin. Here, we describe a biopsy-proven case of severe ATN that manifested as a part of vancomycin-induced DRESS syndrome with coincident PIGN. It is important for clinicians to be aware of this syndrome due to its severity and potentially fatal nature.
Introduction
Drug rash with eosinophilia and systemic symptoms (DRESS) syndrome, also known as drug-induced hypersensitivity syndrome, is a rare but possible fatal condition characterized by skin rash, fever, eosinophilia and multiorgan involvement such as skin, liver, pancreas and kidney due to drug exposure12). Various drugs including carbamazepine, allopurinol and sulfasalazine can lead to this syndrome3). Renal involvement of DRESS syndrome has been reported to occur in 10% or 30% of patients and commonly presents as acute kidney injury due to interstitial nephritis34). Here we report a case of renal involvement of DRESS syndrome which may have been caused by vancomycin with coincident postinfectious glomerulonephritis (PIGN).
Case Report
An 11-year-old boy was referred to the Children's Hospital of Pusan National University because of persistent fever, rash, abdominal distension and generalized edema. He had been suffering from parotitis for 2 weeks and treated with intravenous antibiotics including vancomycin and ceftriaxone for 10 days at another hospital. He had a bad headache for the previous 3 days while in the previous hospital, and his blood pressure was above 135/80 mmHg. Before admission to that hospital he had been well with no significant past medical and familial history. On our admission, he was febrile in 38.2℃ with heart rate of 100 beats/min, respiratory rate of 22 breaths/min and blood pressure of 110/70 mmHg. He had gained 6.8 kg in the previous 10 days. Physical examination showed generalized maculopapular exanthematous eruption especially around neck, painful cervical lymphadenopathy, facial edema and conjunctival injection. Lab investigations revealed hemoglobin 13.5 mg/dL, white blood cell 22,740/µL with eosinophilia in peripheral blood (24%), erythrocyte sedimentation rate (ESR) 21 mm/hr, C-reactive protein (CRP) 10.54 mg/dL, ASO 420 units aspartate aminotransferase/alanine aminotransferase 9/9 IU/L, blood urea nitrogen 13 mg/dL, and creatinine 0.84 mg/dL. Blood cultures and several viral serological tests were negative. His initial urinalysis showed a specific gravity of 1.010, protein of 2+ and red blood cell of 15.20/HPF (high power field). Urine culture and urine eosinophil were negative. Course of major laboratory findings during admission is shown in Table 1.
We initially suspected Kawasaki disease, but we could not exclude the possibility of an infectious disease such as suppurative parotitis, so treated him with intravenous immunoglobulin of 1 g/kg over 2 days, and started intravenous cefazolin instead of vancomycin and ceftriaxone for suppurative parotitis. However, his fever and skin rash continued and were worsening. An echocardiography performed on the third day demonstrated normal heart structure and good function with intact coronary arteries except for a small pericardial effusion. Neck computed tomography displayed acute lymphadenopathy at neck level (I.VI) combined with suppurative parotitis.
During the following a couple of days, eosinophil count rapidly increased to 18,150/uL (51%) accompanying increased ESR, CRP and leukocytosis. Renal functions mildly deteriorated with serum creatinine rising to 0.94 mg/dL. There was no evidence of bacterial or parasitic infection, hematological malignancy or immune deficiency. A skin biopsy of the initial lesion showed a perivascular lymphocytic infiltration, consistent with drug reaction. We then suspected DRESS syndrome resulting from the vancomycin, so to prevent further aggravation of symptoms, corticosteroids (1 mg/kg/day) was initiated on the fourth day. With steroid treatment, systemic symptoms such as fever, rash, and generalized edema had improved. His renal function and eosinophilia also improved with the exception of proteinuria. Subsequent investigations of proteinuria revealed hypocomplementemia (C3 30 mg/dL, C4 8.3 mg/dL) without antineutrophil antibody and antineutrophil cytoplasmic antibodies. Urinary protein electrophoresis demonstrated glomerular proteinuria, and an elevated urinary beta 2 microglobulin level. A kidney biopsy (Fig. 1) was performed on the 20th day because of persistent nephrotic range proteinuria (2,900 mg/day) and hypocomplementemia, which displayed hypercellular glomeruli with infiltration of polymorphonuclear leukocytes concomitant with focally severe necrotic tubular lesion. There was regenerating activity of tubular epithelial cells with hyperchromatic nuclei. Immunofluorescence findings were consistent with immune-mediated nephropathy and suggestive of PIGN having a diffuse mesangial staining of C3. We thought that his glomerular lesions could be related to previous infection of supurative parotitis, while his acute tubular necrosis was probably attributed to vancomycin.
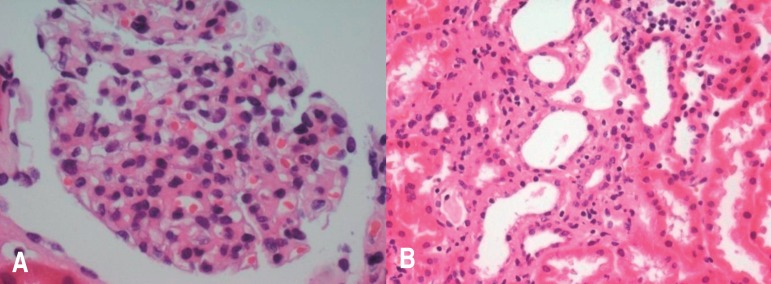
Glomerular and tubulointerstitial structures on light microscopy. (A) The glomeruli show a moderate increase in size and are hypercellular, with an infiltration of polymorphonuclear leukocytes and mononuclear cells (Periodic acid.Schiff stain, ×400). (B) Tubules indicate focally severe necrosis with denudation and sloughing of epithelial cells (Periodic acid.Schiff stain, ×200).
His massive proteinuria had slowly improved with supportive therapy, so he was discharged with a steroid tapering on the 30th day. Now, 1.5 years after discharge, he is doing well without recurrence of previous symptoms and eosinophilia. His renal function remains within normal range without proteinuria and hematuria.
Discussion
DRESS syndrome a severe cutaneous adverse reaction which presents with rash, hematological abnormalities and systemic illness. The syndrome has been known as a variety of names including drug-induced pseudolymphoma, anticonvulsant hypersensitivity, and drug-induced hypersensitivity syndrome, and the currently popular name of DRESS syndrome, as first used by Bocquet et al.5) in 1996. It has been reported to be caused by a variety of drugs, most commonly anticonvulsants, allopurinol, and sulfonamides. The estimated incidence ranges from 1 in 1,000 to 1 in 10,000 drug exposures67). Clinical symptoms can be diverse, the typical findings include fever, maculopapular eruptions, lymphadenopathy, multiorgan failure and eosinophilia or atypical lymphocytosis. Facial erythematosus edema, which our patient experienced, is sometimes mistaken for angioedema, this is typical of DRESS syndrome4). Diagnosis of this syndrome is challenging because of various skin manifestation and organ involvement. However, recognizing this syndrome is essential, as the mortality rate is up to 10%, which is due to the degree of hepatic and renal involvement12). Moreover, Kimmoun et al.8) recently reported that DRESS patients admitted to intensive care unit have a higher mortality rate (47%) compared with previous publish data (10%).
Although no formal diagnostic criteria have been accepted, two diagnostic criteria have been proposed. The RegiSCAR group has suggested a series of criteria and scoring system9). And another set of diagnostic guidelines generated by a Japanese working group in 2007, has included HHV6 activation as a diagnostic feature10). Our patient fulfilled the inclusion criteria and would be a definite DRESS case scoring 7 points on the RegiSCAR scoring system. However, our patients had 4 of Japanese group criteria which could not fulfill the criteria. We consider this is because we treated him early before further aggravation of disease.
The pathophysiology of DRESS syndrome is unclear. Failure of drug detoxification pathways, leading to an accumulation of toxic metabolites, has been hypothesized to explain the pathophysiology of DRESS syndrome3). It has been suggested that concomitant viral reactivation of herpes simplex virus-6 predisposes the patient to development of this syndrome10), however we were unable to check the viral status of herpes simplex virus-6.
Renal involvement of DRESS syndrome was seen in 8% of cases and usually presents as acute kidney injury due to interstitial nephritis, which may be granulomatous11). We could not find any reports about ATN as a part of DRESS syndrome, although there were a few reports about vancomycin induced ATN without DRESS syndrome. We suspect that our patient suffered from acute kidney injury due to ATN and his massive proteinuria was due to ATN with coincident PIGN. To our knowledge this is the first case of biopsy proven acute tubular necrosis as a part of DRESS syndrome, moreover with coincident PIGN. Vancomycin, which was suggested as causative agent in our case, is a nephrotoxic drug with acute kidney injury typically attributed to acute interstitial nephritis, and less commonly has been associated with acute tubular injury12). His renal pathology matched the criteria for acute tubular necrosis, however there was no extensive interstitial inflammation as should be found an acute interstitial nephritis Drug induced ATN are characterized by proximal tubular widening and epithelial cell attenuation. An interstitial component is not typically prominent in ATN while tubulointerstital compartment of PIGN shows edema and collection of inflammatory cells. Therefore we suspected that ATN in this patient could be caused by drug rather than PIGN itself.
As far as we know, there have been 2 reports of DRESS syndrome induced by ceftriaxone1314) and there was no renal involvement in those two cases. So, we believe that potential causative drug in our patient may be vancomycin rather than ceftriaxone. Although, until now there are few reports of vancomycin induced DRESS syndrome, vancomycin is increasingly being recognized as a causative drug in DRESS syndrome2).
Treatment consists of stopping the offending medication and providing supportive care. The use of steroids in DRESS syndrome is controversial because the etiology of the rash is not always obvious and the use of systemic corticosteroids has associated risks. Corticosteroids may make the disease worse by prolonging wound healing, increasing the risk of infection, masking early signs of sepsis and precipitating gastrointestinal bleeding, thus increasing mortality2). But, it is recommended to start systemic corticosteroids when internal organ involvement is present. Despite the lack of randomized controlled trials comparing supportive care alone to systemic steroids in the treatment of DRESS, experience has dictated their use, and they are recommended by experts. Relapses may occur when corticosteroids are tapered and if there is re-exposure to the same drug1). If a patient's symptoms deteriorate despite systemic corticosteroids, other options used successfully in patients have included pulsed intravenous methylprednisolone, cyclosporine, and plasmapheresis1). We had to start systemic corticosteroid because of aggravation of his systemic symptoms including sustained fever, generalized edema, and rash with hematologic deteriorations in spite of stopping the previous drug. Fortunately, he is doing well without recurrence after steroid tapering and his renal function have recovered completely. We still monitor him regularly in case of recurrence.
We present a biopsy proven case of severe ATN as a part of vancomycin induced DRESS syndrome with coincident PIGN. Renal involvement of DRESS syndrome usually presents as acute kidney injury due to interstitial nephritis but our case showed severe acute tubular necrosis which supposed to be induced by vancomycin. DRESS syndrome, although rare, should be considered as a differential diagnosis in children as well as adults with a severe cutaneous eruption and systemic symptoms. It is important for clinician to be aware of this syndrome due to the severity and life-threatening potential of this syndrome. Also we need to notice that vancomycin is increasingly being recognized as a causative drug in DRESS syndrome.
Notes
Conflict of interest: No potential conflict of interest relevant to this article was reported.
Acknowledgments
This study was supported by research grant from Pusan National University (2 years).

