Clinical manifestation of Campylobacter enteritis in children
Article information
Abstract
Purpose
Timely antibiotic therapy in selected cases of diarrhea associated with bacterial infections can reduce the duration and severity of illness and prevent complications. The availability of a predictive index before identification of causative bacteria would aid in the choice of a therapeutic agent.
Methods
The study included patients admitted to the pediatrics unit at Konyang University Hospital for acute inflammatory diarrhea from August 1, 2015 to July 31, 2016 who underwent multiplex polymerase chain reaction testing. Of 248 patients, 83 had positive results. The clinical symptoms and blood test results were examined in 61 patients with Campylobacter spp. (25 patients), Salmonella spp. (18 patients), and Clostridium perfringens (18 patients) infections. The mean age of the 61 patients (male:femal=31:30) was 84.0±54.8 months, and the mean hospital stay was 4.6±1.7 days.
Results
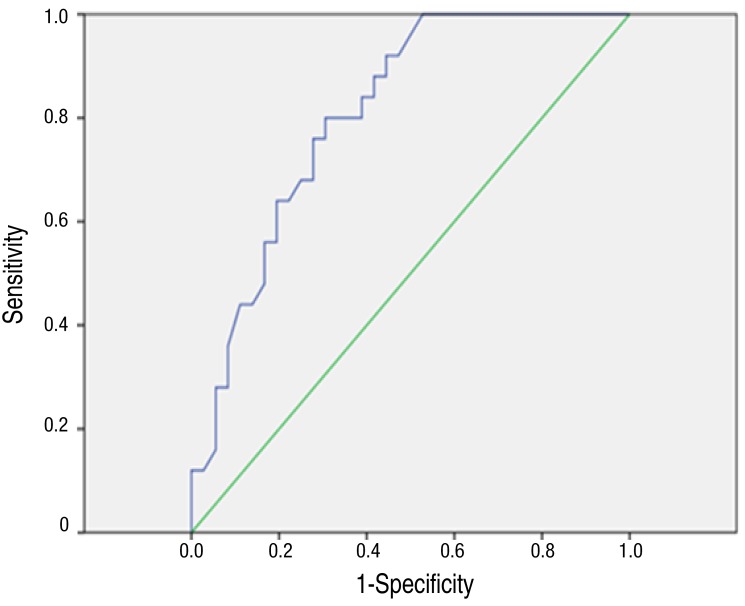
There were no statistical differences in sex, age, clinical symptoms, or signs. Patients with Campylobacter infection were significantly older (P=0.00). C-reactive protein (CRP) levels in patients with Campylobacter infection were higher than those in the other 2 groups, at 9.6±6.1 mg/dL. The results of receiver-operating characteristic curve analysis showed that the cutoff age was ≥103.5 months (sensitivity, 72%; specificity, 86%) and the CRP cutoff level was ≥4.55 mg/dL (sensitivity, 80%; specificity, 69%).
Conclusion
Age (≥103.5 months) and higher CRP level (≥4.55 mg/dL) were good predictors of Campylobacter enterocolitis. If neither criterion was met, Campylobacter enterocolitis was unlikely (negative predictive value 97.2%). When both criteria were met, Campylobacter enterocolitis was highly likely.
Introduction
Gastroenteritis, the most common cause of acute diarrhea, is caused by bacteria, viruses, and parasites. Suspected causes may be inferred based on epidemiologic clues, clinical features, findings from a physical examination, and pathophysiological mechanisms of the pathogen involved. Examinations for identifying the cause may not be needed since most patients recover naturally, but all patients do require a supply of fluids and electrolytes, while a small percentage of patients may benefit from antibiotics. Acute infectious diarrhea can be divided largely into noninflammatory and inflammatory types. When bacteria directly infiltrate the intestines or produce cytotoxins, inflammatory diarrhea can be caused. Foodborne outbreaks in the United States are mostly caused by norovirus, accounting for 58% of all episodes, and by bacterial causes, which are most commonly nontyphoidal Salmonella spp. (11%), Clostridium perfringens (10%), Campylobacter spp (9%).1) However, it is very difficult to distinguish the causative bacteria based on clinical features, physical examination, and laboratory findings in cases of inflammatory diarrhea caused by such bacteria.
In most cases, treatment against the bacteria that causes inflammatory diarrhea does not require antibiotic therapy, but some cases do require treatment by antibiotics. Symptoms caused by Campylobacter spp. can be improved through supportive care, but in severe cases, macrolide class antibiotics, such as erythromycin or azithromycin, must be administered.2) In cases of Salmonella spp., improvements can be achieved without antibiotics for enteritis without complications, but antibiotics must be administered in cases involving infants less than 3 months of age, immunosuppressed patients, and patients with underlying diseases.2) Meanwhile, cases of C. perfringens can be treated via supportive care without using antibiotics.2)
Previous studies have reported that antibiotic use was associated with symptom improvement and a faster recovery in cases of bacterial enteritis, and in cases that meet the indications for antibiotics administration. Although few in number, administering antibiotics can reduce the duration of disease, reduce transmission, and prevent complications.34) Accordingly, although cases that require the use of antibiotics for bacterial enteritis in children are limited, if clinical administration of antibiotics is required, then deciding which antibiotic to use would ultimately have an effect on the progression of the disease. However, it is difficult to distinguish the causative bacteria based on clinical features, physical examination, and laboratory findings, while identification through culture test can be time consuming, it is not useful when antibiotics must be administered in a timely manner. Therefore, the present study was conducted under the premise that if certain predictive indices were available to use before we actually knew the causative bacteria of acute inflammatory diarrhea, this would be helpful in selecting the appropriate antibiotic.
Materials and methods
Among a total of 248 pediatric patients who were admitted to the pediatric unit at Konyang University Hospital for a chief complaint of acute diarrhea between August 1, 2015 and July 31, 2016 and underwent subsequent fecal examination via multiplex polymerase chain reaction (PCR), 165 patients with no detection of bacteria and 22 patients with detection of pathogens other than Campylobacter spp., Salmonella spp. and C. perfringens were excluded. As a result, a total of 61 pediatric patients were selected for the present study.
All blood tests and fecal examinations were performed on the day of admission, with all fecal samples sent to the lab immediately. Nucleic acid was extracted using SeePrep12 Automated Magnetic Bead-based Nucleic Acid Extraction kit (Seegene, Seoul, Korea), which was used for performing multiplex PCR with Seeplex Diarrhea ACE detection kits (Seegene). All procedures were performed in accordance with the manufacturer's recommended protocol. The number of possible causative bacteria types being tested was 8: Campylobacter spp., Clostridium difficile toxin B, Salmonella spp., Shigella spp., enteroinvasive Escherichia coli, Vibrio spp., Yersinia enterocolitica, and Aeromonas spp.
Patients who were infected with Campylobacter spp., Salmonella spp., and C. perfringens which were the bacteria most commonly detected in the fecal examination via multiplex PCR performed at Konyang University Hospital, were investigated for their sex, age, season, length of hospital stay, extragastrointestinal symptoms, peak body temperature (PBT), presence of bloody stool, suspected surgical abdomen, area with tenderness during physical examination, and laboratory findings. The results were compared and analyzed, after which, 2 groups were created as the Campylobacter spp. group and other groups, and the same items mentioned above were compared and analyzed between the 2 groups. Based on this, the receiver-operating characteristics (ROC) curve and predictive indices of Campylobacter spp. were derived (Fig. 1).
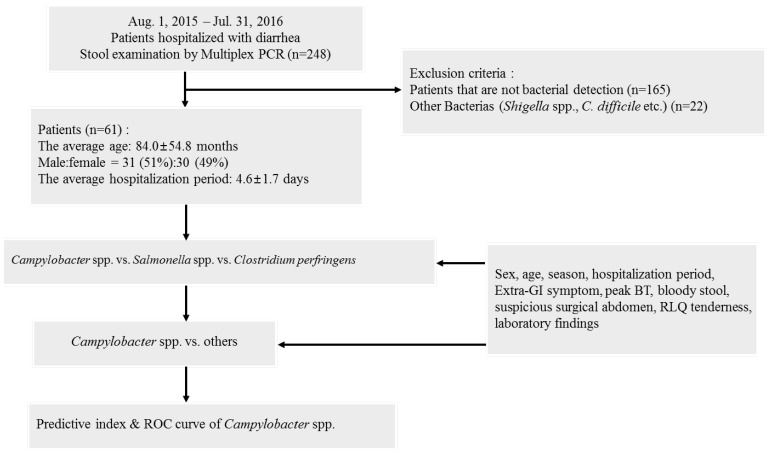
A flow chart of the protocol of the present study. PCR, polymerase chain reaction; RLQ, right lower quadrant; GI, gastrointestinal; BT, body temperature; ROC, receiver-operating characteristics.
Continuous variables were analyzed using analysis of variance, and categorical data were analyzed by using the chi-square test or Fisher exact test. A P value of <0.05 was considered statistically significant. Two variables (age and C-reactive protein [CRP]) with statistical significance were used as a predictive index of Campylobacter spp. ROC analysis was performed on this predictive index to assess the ability to discriminate Campylobacter spp. from Others (Salmonella spp., C. perfringens). Statistical analyses were performed using IBM SPSS ver. 18.0 (IBM Co., Armonk, NY, USA).
This study was approved by the Institutional Review Board of Konyang University Hospital (approval number: KYUH 2017-10-003).
Results
Among the 61 patients included in the study, 31 (51%) were boys and 30 (49%) were girls. Their mean age was 84.0±54.8 months, and the mean length of hospital stay was 4.6±1.7 days. In the fecal examination via multiplex PCR, 25 patients (41%) were detected with Campylobacter spp., which was the most common, followed by 18 patients (29.5%) each detected with Salmonella spp. and C. perfringens.
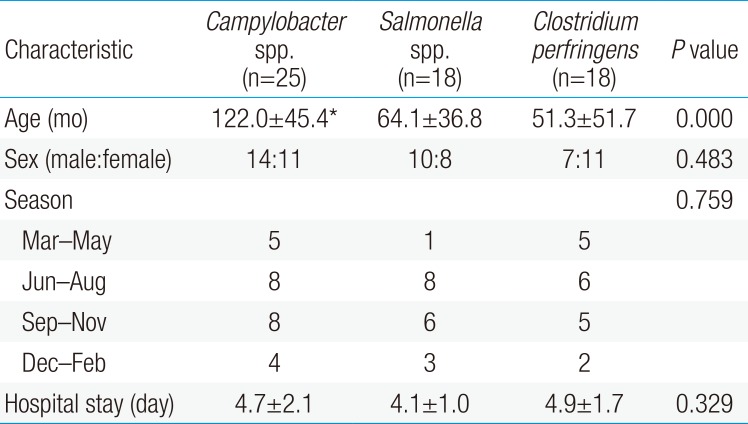
The demographic characteristics of these 3 groups are shown in Table 1. The age of the Campylobacter-infected group was 122.0±45.4 months, which showed a statistically higher difference than the other groups (P=0.000). There were no statistically significant differences in sex, season of onset, and length of hospital stay among the 3 groups.
There were no statistically significant differences among the 3 groups with respect to PBT and presence of bloody stool in the clinical manifestations at the time of diagnosis. Extragastrointestinal symptoms were investigated as having no extragastrointestinal symptoms, upper respiratory infection symptoms (coughing, phlegm, and runny nose), and other symptoms. The results did not show significant differences. Among other symptoms, there were 2 patients, each from the Campylobacter spp. and Salmonella spp. groups, who showed nonspecific rashes. With respect to radiologic examinations, such as abdominal computed tomography or ultrasonography, performed for right lower quadrant (RLQ) tenderness and suspected surgical abdomen, there were no statistically significant differences among the three groups (Table 2).
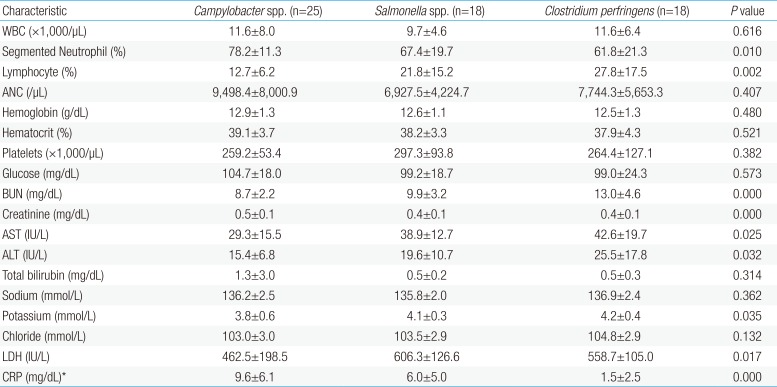
Laboratory findings are shown in Table 3. Segmented neutrophil and lymphocyte results showed statistically significant differences, but the differences were within their normal range (P=0.010, P=0.002). There were no significant differences in white blood cell (WBC), absolute neutrophil count, hemoglobin, hematocrit, and platelet count. Although there were statistically significant differences in blood urea nitrogen, creatinine, aspartate transaminase, alanine transaminase, potassium, and lactate dehydrogenase levels, the differences were within their normal range, and thus, the results did not bear much significance. Meanwhile, there was a significant difference in the CRP among the three groups (P=0.000), with the Campylobacter spp.-infected group showing a higher value than the other 2 groups at 9.6±6.1 mg/dL.
Items that showed significant differences among the 3 groups were age and CRP. The results from comparing these 2 items by 2 groups (Campylobacter spp. vs. others) are shown in Table 4. Similar to when they were divided into 2 groups, both age and CRP showed statistically significant differences. In the Campylobacter spp. group, age was higher at 122.0±45.4 months, and CRP was also higher with 9.6±6.1 mg/dL (P=0.000, P=0.017). ROC curve analysis of age and CRP showed that the area under the curve (AUC) of the ROC curve for age was 0.832, with a cut off value of ≥103.5 months, and sensitivity was 72%, and specificity was 86% (Fig. 2). The AUC of the ROC curve for CRP was 0.808, with a cutoff value of ≥4.55 mg/dL, and sensitivity was 80%, and specificity was 69% (Fig. 3). For Campylobacter spp., the positive and negative predictive values for satisfying both conditions of age cutoff value of ≥103.5 months and CRP cutoff value of ≥4.55 mg/dL were 56% and 97.2%, respectively.
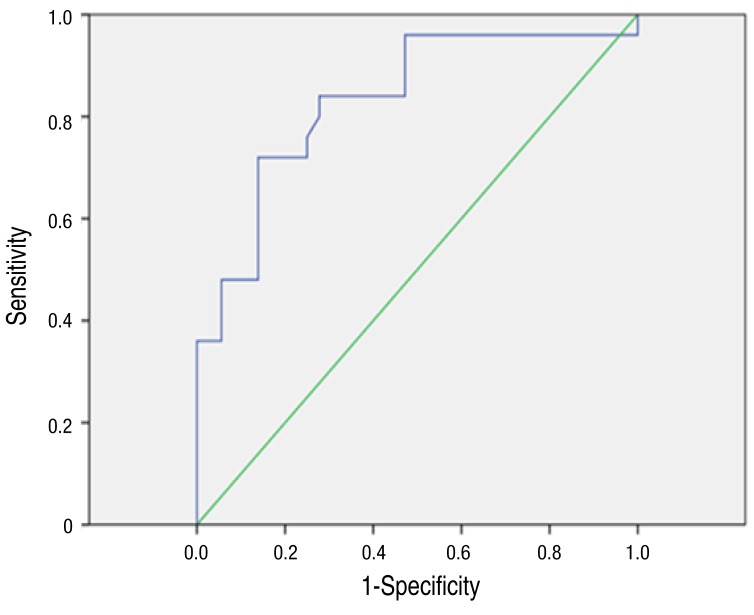
Receiver operating characteristic (ROC) curves of age for the prediction of Campylobacter enteritis. In the ROC curve for age, the area under the curve was 0.832, and when the cutoff value was set to ≥103.5 months, sensitivity was 72% and specificity was 86%.
Discussion
According to statistics on bacterial gastrointestinal infections, regardless of age, gathered from tertiary general hospitals and medical institutions with 300 or more beds throughout during the 2015 calendar year by the Korea Centers for Disease Control and Prevention (KCDC), among a total of 7,001 patients who had reported bacterial gastrointestinal infection, Salmonella spp. was most prevalent (n=625), followed in order by Campylobacter spp. (n=539) and C. perfringens (n=396).5) The present study showed slightly different results with incidence of Campylobacter spp. being the highest, followed in order by Salmonella spp. and C. perfringens. It is believed that the difference may be because the results in the present study represented findings from a single university hospital with shorter study period, whereas the results reported by KCDC represented nationwide results with longer study period. According to studies from other countries, a study conducted in India between June and July 2012 on 280 diarrheal patients 12 years or younger, reported that among 280 patients with detection of bacteria in their stool culture, E. coli was most common (n=124), followed by Shigella (n=79) and Salmonella (n=38).6) A study conducted in China between August 2014 and September 2015 on diarrheal patients, ranging in age from 1 month to 78 years reported that among 107 patients with bacteria identified from stool culture or multiplex PCR, 37, 20, and 18 patients were identified as having Salmonella spp., enterotoxigenic E. coli, and Campylobacter jejuni, respectively.7) Such differences may be attributed to differences in the age of the subjects and bacteria detection methods, as well as differences in the lifestyle and level of hygiene in those countries and regions. Therefore, additional studies focusing on children and adolescents are needed, while it is also necessary to renew the trends on an annual basis. Unlike the past, when fecal examinations were conducted through stool culture, the introduction and more universal acceptance of multiplex PCR have led to an increased frequency of bacterial causes being identified in children and adolescents, and as such, studies on this topic are expected to increase in the future. In particular, studies on bacterial enteritis in children and adolescents are especially lacking in Korea, which makes the present study more significant.
In the present study, the mean age of the C. jejuni-infected group was significantly higher than the other infection groups at 122±45.4 months, which was similar to a previous study by Lee et al.8) However, in the previous study, the C. jejuni-infected group showed significantly higher incidences of abdominal pain and watery diarrhea, whereas the present study did not show differences in clinical manifestations based on the causative bacteria. In the previous study, laboratory findings showed that C. jejuni had significantly higher leukocytosis and predominance of segmented neutrophil than non-typhoid Salmonella, while in the present study, the Campylobacter spp.-infected group a showed significantly higher WBC and segmented neutrophil count, although they were all within the normal range. Moreover, although the C. jejuni-infected group did not show a statistically significant difference in CRP levels compared to the non-typhoid Salmonella-infected group, in the present study, pediatric patients infected with Campylobacter spp. showed a higher CRP value of 9.6±6.1 mg/dL than those infected with Salmonella spp. or C. perfringens.8) The previous study of Shim et al.9), it was similar to the present study that the incidence and age were significantly higher in patients infected with Campylobacter spp. However, the lack of statistical significance of CRP was different from the present study. Therefore, the present study is meaningful in that CRP is statistically significant and can be effective value for diagnosis of Campylobacter enteritis.
Although there have been reports of neurologic complications such as grand mal seizures, meningismus, and encephalopathy being found in children and adolescents with Campylobacter enteritis, in the present study, there were no extragastrointestinal symptoms, except nonspecific rashes.1011)
Although there were no significant differences, Campylobacter spp. cases in the present study showed 3 children with RLQ tenderness and 5 children who required additional radiologic examination (abdominal ultrasonography and computed tomography) for suspected abdomen surgery due to severe cramping pain or RLQ tenderness. A previous study reported that if there is severe abdominal pain prior to the diarrhea episode in C. jejuni enteritis patients, it can appear similar to an acute appendicitis due to RLQ tenderness, while another study suggested that acute appendicitis might be induced by C. jejuni.81213)
With respect to changes in antibiotic resistance by Campylobacter spp. from 1994 to 2010, antibiotic resistance rates for fluoroquinolones, gentamicin, and cefuroxime have shown large increases, whereas the increase in the resistance rate for erythromycin has been lower than that for other antibiotics with an increase of only 3%–6.4%.14) Even in another study, the resistance rate for erythromycin was low at only about 6%.15) A study in Poland analyzed children 4 years or younger between 2011 and 2013 and found that there were no children that displayed resistance to azithromycin or erythromycin.16) Fluoroquinolones are not commonly used on children, but its increase in resistance rate may be meaningful. Fluoroquinolones are the first-line antibiotics used on adult cases of acute diarrhea, and their overuse may have led to the current high resistance rate. Therefore, although the current resistance rates for macrolide-class antibiotics are low, they may increase similarly if used indiscriminately.
Limitations in the present study included that while testing for bacteria via multiplex PCR, coinfections with viruses were not considered. Therefore, the clinical manifestations and laboratory findings being affected by viruses that can cause enteritis cannot be eliminated. In addition, the present study did not analyze each type of bacteria by its subspecies. Therefore, comparisons of characteristics between bacteria could not be made.
In summary, the Campylobacter spp.-infected group had age and CRP cutoff values of ≥103.5 months and ≥4.55 mg/dL, respectively. These criteria would indicate that they are useful predictors of Campylobacter enterocolitis. Cases that do not satisfy both age and CRP cut off values have a low probability of experiencing Campylobacter enteritis, and antibiotics other than macrolide-class antibiotics should be considered. If both conditions are satisfied, then Campylobacter enteritis can be strongly suspected, which would be very helpful in selecting the applicable antibiotics. Therefore, it is believed that using antibiotics in an appropriate and limited manner can be helpful in reducing the duration of the disease and complications, while also reducing antibiotics resistance rates.
Notes
Conflicts of interest: No potential conflict of interest relevant to this article was reported.