Neonatal hypertension: concerns within and beyond the neonatal intensive care unit
Article information
Abstract
Neonatal hypertension occurs in 1%–2% of neonates in the neonatal intensive care unit (NICU) although may be underdiagnosed. Blood pressure values in premature neonates change rapidly in the first days and weeks of life which may make it more difficult to recognize abnormal blood pressure values. In addition, the proper blood pressure measurement technique must be used to ensure the accuracy of the measured values as most blood pressure devices are not manufactured specifically for this population. In premature neonates, the cause of the hypertension is most commonly related to prematurity-associated complications or management while in term neonates is more likely to be due to an underlying condition. Both oral and intravenous antihypertensive medications can be used in neonates to treat high blood pressure although none are approved for use in this population by regulatory agencies. The natural history of most neonatal hypertension is that it resolves over the first year or two of life. Of concern are the various neonatal risk factors for later cardiovascular and kidney disease that are present in most NICU graduates. Prematurity increases the risk of adulthood hypertension while intrauterine growth restriction may even lead to hypertension during childhood. From neonates through to adulthood NICU graduates, this review will cover each of these topics in more detail and highlight the aspects of blood pressure management that are established while also highlighting where knowledge gaps exist.
Key message
Some neonates, especially those who are premature, may experience hypertension while in the neonatal intensive care unit (NICU). The most common causes are prematurity-related and the hypertension usually resolves over the first 1–2 years of life. Unfortunately, the increasing population of NICU graduates is at risk for later cardiovascular and kidney disease in childhood and adulthood. This population requires careful attention to blood pressure and weight throughout their life course.
Graphical abstract
Introduction
Hypertension in the neonatal period and neonatal intensive care unit (NICU) has become increasingly recognized. Historically, estimates of incidence have ranged from 0.2% to 3% [1-6]. More recently, results from the Assessment of Worldwide Acute Kidney Injury Epidemiology in Neonates study identified hypertension in 1.8% of the cohort [7]. Undiagnosed hypertension was found in an additional 3.7% of neonates. Management of hypertension in the neonate is not only important in the acute or critical care setting, but it is now becoming apparent that hypertensive neonates require ongoing follow-up well beyond the neonatal period. In this review, identification and management of neonatal hypertension will be discussed, with emphasis on follow-up of not only hypertensive neonates, but also all NICU graduates, into childhood and beyond.
Definition of hypertension in the neonate
Defining hypertension in a neonate can be difficult, as there are multiple changes that occur in blood pressure during the first weeks of life and are especially pronounced in the preterm population. Blood pressure can also be variable in this population based on a variety of factors. During the first month of life, blood pressure increases more rapidly in preterm than full-term infants and the highest rates of rise occur in infants ≤32 weeks’ gestation and those with very low birth weight (VLBW, ≤1,500 g) [8]. Mean blood pressure increases with gestational age at birth as well as postnatal age.
The reason for the rapid changes in blood pressure in the first month of life may be due to decreased activity and synthesis of vasodilatory metabolites [9] and due to more pronounced hemodynamic changes in preterm infants due to decreased maturity of their cardiovascular systems compared to term infants [8]. Decreased nephron mass may also play a role for these premature, low birth weight and also for small for gestational age (SGA) infants [10,11].
Other factors affecting blood pressure can include medication exposures in both mother and infant. Perinatal treatment with steroids including dexamethasone has been potentially linked to neonatal hypertension as well as cardiovascular disease later in life [12-14]. Antenatal exposure to tocolytic nonsteroidal antiinflammatory drugs has been associated with greater odds of neonatal hypertension, even after adjustment for other factors associated with neonatal hypertension such as critical illness, lung disease, ventilation, and umbilical artery catheterization [15]. Prenatal calcium channel blocker exposure has been suggested to be protective against development of neonatal hypertension while maternal labetalol has been associated with neonatal hypotension [15,16].
Large-scale studies on normal blood pressures of a noncritically ill neonatal population are limited (Table 1). Previously, Dionne et al. [17] used existing data to derive a table of systolic, diastolic, and mean arterial blood pressure percentiles by postmenstrual age. This table is generally used as a reference for normative blood pressure values in this population although better normative references are needed. In addition, the correct definition of neonatal hypertension is not known but it has been recommended that neonates have close monitoring for consistent blood pressure readings >95th percentile, and that evaluation and treatment be considered for blood pressure values >99th percentile [18].
Measurement of blood pressure
In critically ill neonates, intra-arterial measurement for direct monitoring via indwelling radial or umbilical arterial catheter is considered the gold standard. Yet, intra-arterial catheters come with risks of thrombosis and ischemia so are limited to the most critically ill neonates. Nondirect measurement in neonates without arterial access is more common but can be more challenging. Oscillometric devices are widely used in the NICU setting but have some limitations. Oscillometric device values are most accurate for mean arterial pressure while systolic blood pressure values tend to overestimate directly measured values [19]. These devices are also less accurate when the mean arterial pressure is less than 30 mmHg (Table 1). Ultrasonic Doppler measurement is less commonly used, and manual auscultation and palpation methods tend to be less practical in the intensive care setting [6].
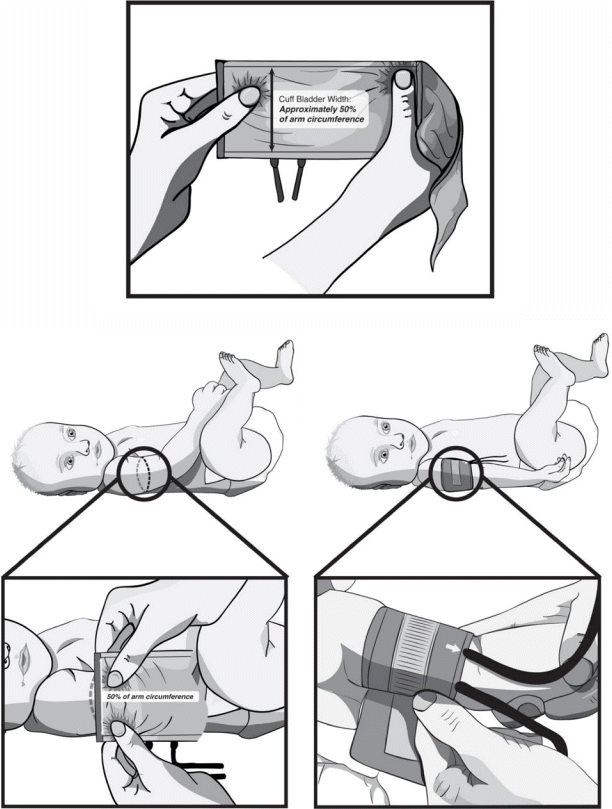
An appropriate blood pressure cuff size is also important in the measurement of neonatal blood pressure. It is recommended that the bladder of the cuff encircle > or = 75% of the upper arm circumference [20], and more importantly, the cuff width to arm circumference ratio should be approximately 0.5 (Fig. 1) [19]. In infants up to 6 months of age, measurements on the calf are equivalent to values obtained from the upper arm [18], although International Neonatal Consortium recommendations for measurement of blood pressure in neonates suggest that calf blood pressures be used only in the first few days of life or if there is a contraindication to using the upper arm for blood pressure measurements [19].
Neonatal blood pressure can also vary based upon the level of activity of the infant and multiple readings may be needed for an accurate assessment. A protocol that has been suggested for neonatal blood pressure measurement includes using an oscillometric device on the infant’s right upper arm, waiting 15 minutes after cuff placement and at least 1.5 hours after feeding, while the infant is in a restful state. It is recommended that 3 readings be taken, at 2-minute intervals [21]. This standardized blood pressure measurement technique resulted in lower and less variable blood pressure values than by routine nursing care.
Etiology
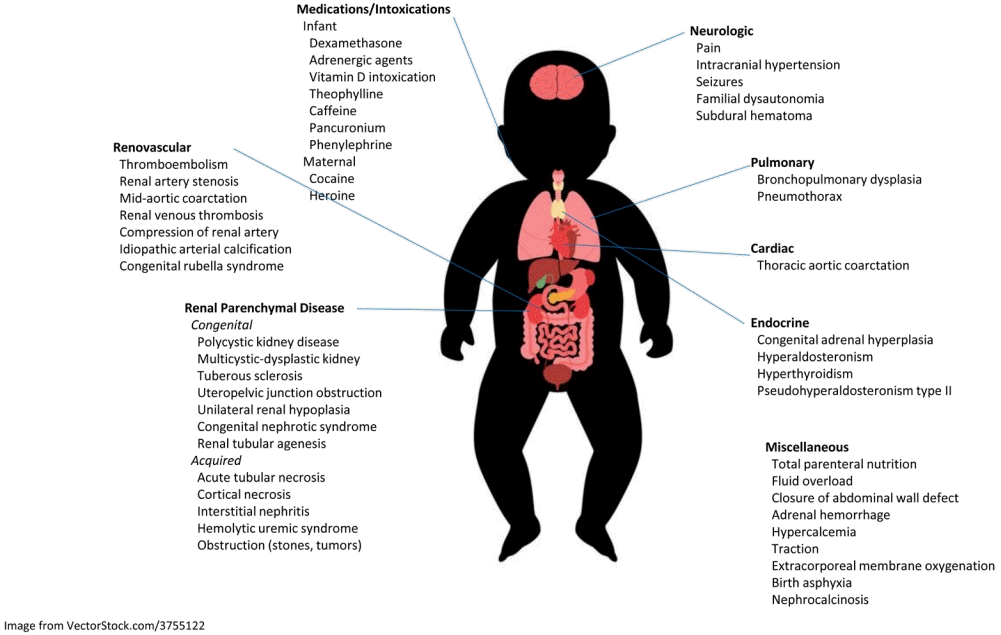
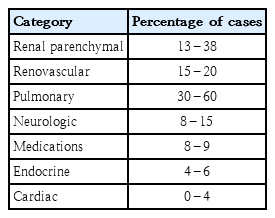
The most common causes of hypertension in the neonatal population fall into 3 categories: renovascular disease (most commonly catheter-associated thrombus), renal parenchymal disease, which can be acute or chronic, and chronic lung disease (Fig. 2, Table 2) [3,22-26]. However, a 2017 study reported “unexplained hypertension” with no underlying cause in approximately half of the hypertensive neonate population [27]. The etiology of neonatal hypertension in premature infants is primarily iatrogenic causes and complications or risk factors related to prematurity [22]. These neonates often have a long and complicated neonatal course with several perinatal risk factors for systemic hypertension [23] (Table 1). In term neonates with hypertension, an underlying cause or condition is often found [22].
Umbilical artery catheter (UAC)-associated thrombi have been reported to occur in about 25% of neonates with these types of lines, although UAC-associated hypertension can occur even without a current thrombus on imaging. This suggests that vascular endothelial disruption may play a role in the development of hypertension in this population [28]. Other renovascular etiologies include renal vein thrombosis (RVT), which is the most common noncatheter-associated thrombosis of infancy. It may not always present with the classical signs of gross hematuria, palpable flank mass and thrombocytopenia, and risk factors for RVT include male sex and prematurity <36 weeks' gestation [29]. One study noted a reported incidence of symptomatic RVT in 2.2 per 100,000 live births [30]. Persistent hypertension requiring ongoing follow-up has previously been identified in 1/5 of neonates with diagnosed RVT [29]. Other less common causes of renovascular hypertension in a neonate can include noncatheter-related renal arterial thrombosis, fibromuscular dysplasia leading to renal artery stenosis, external compression of the renal arteries, and syndromic causes such as Neurofibromatosis type I, Williams’ syndrome or Marfan’s syndrome [31].
Renal parenchymal disease leading to neonatal hypertension can be caused by cystic kidney disease, congenital anomalies of the kidney and urinary tract, and tubulointerstitial disease. Either autosomal dominant or recessive types of polycystic kidney disease may be associated with hypertension, with autosomal recessive disease more commonly associated with hypertension in infants. Congenital causes of renal parenchymal disease include obstructive uropathy (such as ureteropelvic junction obstruction), renal hypoplasia, tuberous sclerosis, congenital nephrotic syndrome, and renal tubular agenesis. Acquired renal parenchymal disease can result from acute kidney injury secondary to acute tubular necrosis, cortical necrosis, interstitial nephritis, hemolytic uremic syndrome, and obstruction resulting from nephrolithiasis or tumors (Fig. 2).
Chronic lung disease has been increasingly recognized in the development of neonatal hypertension over the past few decades. The incidence of hypertension in infants with bronchopulmonary dysplasia has been reported at 43% versus 4.5% in infants who do not have chronic lung disease [32]. It has been noted to present around 40 weeks’ postmenstrual age and to resolve within another 25 weeks (or the approximately within the first year of life) within all patients. Some patients with chronic lung disease develop their hypertension after discharge from the NICU so studies on the prevalence of pulmonary causes of hypertension in infants can vary widely (Table 2) [33]. The mechanism is felt to be related to enhanced sodium retention leading to volume expansion and is likely related to activation of the mineralocorticoid receptor and a low-renin state [27]. In addition to being at risk for systemic hypertension, infants with chronic lung disease are at risk for other cardiovascular complications such as pulmonary hypertension and left ventricular hypertrophy, and so there may be a role for intermittent echocardiogram monitoring in severe cases [32].
Other less common causes of neonatal hypertension may be neurologic, cardiac, endocrine, or medication related (Fig. 2, Table 2). Neurologic causes can include pain, seizures, and intraventricular hemorrhage. The most common cardiac cause is coarctation of the thoracic aortic which is often identified in the newborn period. Endocrine causes of neonatal hypertension are rare but can include dysfunction of the thyroid and adrenal glands. Medication causes include drugs given to the mother perinataly as well as directly to the infant. Fortunately, most causes are easily identified by review of the patient’s pre- and postnatal course, physical examination, and basic investigations.
Evaluation
In addition to a careful history including medication review of both infant and mother, all hypertensive infants should have a comprehensive physical examination to screen for dysmorphic features which might be associated with genetic syndromes, abdominal distension which could suggest abdominal masses, signs of heart failure, and potentially an ophthalmologic exam to evaluate for retinal abnormalities [18].
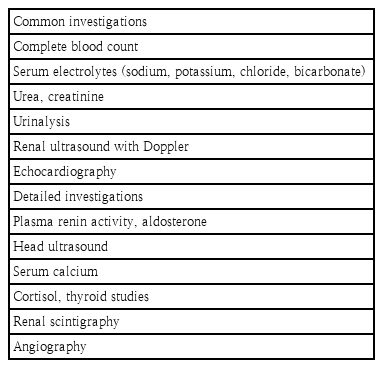
The laboratory and imaging evaluation of a neonate with hypertension can be minimal or more extensive (Table 3). It is generally recommended to obtain serum laboratory studies including a metabolic panel to evaluate serum electrolytes, urea, and creatinine. A complete blood count can screen for thrombocytopenia. A urinalysis is also generally recommended to screen for hematuria, proteinuria, or indicators of infection. A renal ultrasound is recommended to screen for structural kidney abnormalities or size discrepancies. A Doppler study is also recommended to screen for potential signs of abnormal renal vascular flow, although this study is not the gold standard study for diagnosing renal vascular abnormalities. At different medical institutions, the renal ultrasound may or may not standardly come with a Doppler study as part of the order and sometimes these studies must be ordered separately.
Other laboratory and imaging studies may be needed if the initial investigations are normal or a specific diagnosis is suspected based on the evaluation (Table 3). Other investigations that can be considered include plasma renin activity, plasma aldosterone level, cortisol level, thyroid studies (thyroid-stimulating hormone and free T4), and a head ultrasound [34]. Angiography such as computed tomography, magnetic resonance imaging, or digital subtraction angiography can be less accurate and technically challenging in small infants who may need to be medically managed until a size where the procedures are more feasible.
Treatment
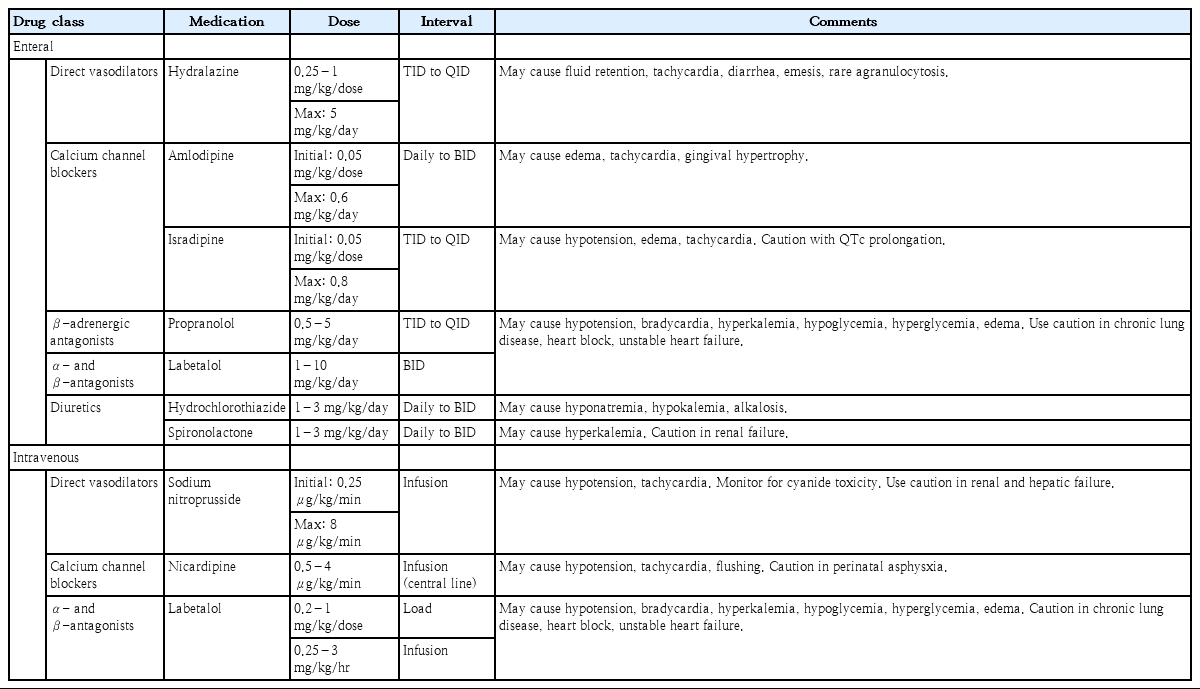
For neonates with mild to moderate or chronic hypertension who have no contraindication to enteral therapy, oral medications can be used (Table 4). Typically, calcium channel blockers are used as first-line oral medications for unspecified hypertension. Isradipine is generally given in the acute setting due to its quick onset of action and relatively low-risk side effect profile. Amlodipine has slower onset of action and is generally initiated in a less severe or more chronic setting. Other oral medication options include vasodilators such as hydralazine, or diuretics such as thiazides or spironolactone. Loop diuretics can be used but have a higher risk of electrolyte abnormalities, worsening acute kidney injury, and also development of hypercalciuria. Beta-blockers such as labetalol or propranolol are generally avoided in infants with chronic lung disease but can be used in chronic management of neonatal hypertension especially of cardiac origin.
The angiotensin-converting enzyme inhibitor and angiotensin receptor blocker classes of medications are generally avoided in neonates due to the risk of adverse effects and the importance of the renin-angiotensin-aldosterone system (RAAS) in renal development. In one study, over 20% of neonates given an angiotensin-converting enzyme inhibitor had adverse effects with the most common complications including hyperkalemia, acute kidney injury, hypotension, and death [35]. Maternal exposure to RAAS blockers is also avoided in pregnancy as these drugs have been associated with oligohydramnios, intrauterine growth restriction, limb and boney defects, and neonatal acute renal failure, anuria, hypotension, respiratory distress, cerebral complications, and death [36]. Significant interruption of the RAAS during fetal development leads to renal tubular dysgenesis, calvarial defects, oligohydramnios, and the Potter sequence (facial malformation, arthrogryposis, pulmonary hypoplasia, and often death) [37]. Given normal glomerular maturation occurs until 36 weeks gestational age with some individual variation, use of RAAS blockers are generally avoided until 40–42 weeks postnatal age when possible.
In the acute and/or acute severe hypertension setting, intravenous (IV) medications are typically recommended (Table 4). First-line agents include intermittent dosing of IV labetalol or hydralazine. However, for a neonatal patient with signs of end-organ damage and thus hypertensive emergency, a continuous infusion of medication is recommended due to the ability to easily titrate dosing to achieve desired blood pressure goals [34]. Recommended medications include nicardipine or sodium nitroprusside, although caution must be used with sodium nitroprusside due to risk of thiocyanate toxicity (Table 1). It is recommended that infants have arterial lines placed for continuous blood pressure monitoring if they are going to be managed with a continuous infusion. If oscillometric cuffs must be used, then it is recommended that the blood pressure frequently be assessed (q5-15 minutes) [18].
Gradual lowering of blood pressure is recommended in infants with hypertensive emergency, to avoid complications such as cerebral ischemia and/or hemorrhage. It is generally recommended that the blood pressure be lowered by one-third of the way to normal over the first 6 hours, by another one-third of the way to normal over the next 24–36 hours, and then corrected the rest of the way to normal over the next 48–72 hours [38]. Goal blood pressure is ultimately less than 95th percentile for postmenstrual age [34].
For neonates on extracorporeal membrane oxygenation (ECMO), a nicardipine infusion is recommended due to its rapid onset, titratability, low side effect profile, and low risk of hypotension [39]. However, the ECMO circuit’s expanded volume of distribution may result in a reduced antihypertensive effect that requires aggressive titration [40]. Nicardipine has also been found to be safe and effective without any significant change in mean heart rate or any adverse effects in children postcoartectomy [41].
Follow-up of neonatal hypertension
Infants who have been identified with neonatal hypertension should be monitored following discharge from the NICU as most neonatal hypertension improves over time. The majority of NICU hypertension resolves within the first one to 2 years of life [23]. In an Australian NICU cohort of neonates with hypertension, 41% were prescribed antihypertensive medications at discharge and only 15% were still on treatment at 3 to 6 months of age [24]. In an earlier American study, researchers found 56% of infants with neonatal hypertension were normotensive at 1 month, 67% by 6 months, and 81% by 1 year [25]. It is reassuring that most prematurity-related neonatal hypertension resolves but the compensation mechanisms responsible for the improvement may leave them at risk of later cardiovascular and kidney disease (Table 1).
Occasionally, hypertension may be diagnosed in follow-up clinics of NICU graduates who were not hypertensive during their NICU course. An early study that assessed blood pressure at 3 months following discharge from the NICU found that 2.6% were newly identified as hypertensive with causes including ureteropelvic junction obstruction, renal artery thrombosis, coarctation of the aorta, and neuroblastoma although no cause was identified in the majority of cases [42]. Eighty percent of the infants with identified causes were managed with surgical correction while the remainder were treated with antihypertensive medications. All were able to be weaned off the antihypertensives within 1 to 2 years [42]. For infants with chronic lung disease, the development and diagnosis of hypertension may occur following NICU discharge [33]. In other neonatal conditions, such as autosomal recessive polycystic kidney disease and renal venous thrombosis, the probability of requiring antihypertensive medication increases with age and therefore many patients are likely to be diagnosed with hypertension following NICU discharge [43,44].
The American Academy of Pediatrics Clinical Practice Guideline for Screening and Management of High Blood Pressure in Children recommends that blood pressure should be measured at every healthcare encounter in children, including those less than 3 years of age, if they have a risk factor for hypertension [45]. Included in this high-risk category are children with a history of prematurity, intrauterine growth restriction, VLBW, UAC, or any condition requiring admission to the NICU. Unfortunately, adherence to this recommendation seems to be less than optimal with a recent healthcare system analysis showing only 38% of premature, VLBW, or former NICU children having a blood pressure measurement in the first 3 years of life [46]. Of the group with blood pressure measurements, 45% had a high screening systolic blood pressure needing repeat assessment suggesting this is a high-risk group that needs specific attention.
Neonatal risk factors for later development of cardiovascular and kidney disease
1. Hypertension in childhood and adolescence
Children who were born premature have higher blood pressure than those born at term but do not seem to have more hypertension during childhood. A study of extremely preterm (<27 weeks’ gestation) children at 6 years of age compared to term-born children found the premature children had higher height-adjusted blood pressure z scores but not more hypertension [47]. The researchers found that for each week longer gestational age at birth, the blood pressure at 6 years was 0.1 standard deviations lower. In a study of adolescents at 14 years of age who were born VLBW (<1.500 g), the low birth weight teens had higher blood pressure than term controls and more elevated blood pressure [48]. These low birth weight adolescents also had lower estimated glomerular filtration rate and higher albumin to creatinine ratios than term-born adolescents although the abnormalities were clinically minor. Using 24-hour ambulatory blood pressure monitoring (ABPM) to assess blood pressure patterns, Bayrakci et al. [49] found preterm children and adolescents had similar 24-hour and daytime blood pressure but higher nighttime blood pressure and less nocturnal dipping than term controls. The elevated nighttime blood pressure reached the threshold of hypertension predominantly only in the premature children who were also born growth restricted.
While prematurity alone does not seem to lead to hypertension during childhood and adolescence, being born intrauterine growth-restricted or SGA has been associated with higher rates of hypertension. Children born SGA have been shown to have higher 24 hours, daytime, and nighttime blood pressure in addition to reduced nocturnal blood pressure decline compared to those born appropriate size for gestational age (AGA) [50]. An ABPM study of 6- to 10-year-old children born SGA showed 18% had hypertension while none of the children born AGA were hypertensive [51]. Elevated blood pressure was found in 50% of the SGA children compared to only 16% in those born AGA. In addition, children born SGA have been shown to have altered circadian and ultradian blood pressure rhythmicity [50]. Vascular alterations in those born SGA may be responsible for these differences with SGA children and adolescents having more arterial stiffness and endothelial dysfunction than children born at an appropriate size [52].
The role of childhood growth trajectories on blood pressure levels of children born premature and/or SGA has been an evolving area of research (Table 1). Studies have shown higher blood pressures in children born premature who had rapid weight gain for length over the first months and years of life, most significantly for those also born intrauterine growth restricted [53]. Belfort et al. [54] found that children at 6 years of age who were born preterm and low birth weight and then had more rapid weight gain in the first year of life had slightly higher blood pressure but they also had higher cognitive scores. It also seems to be important what the infants are feeding with a systematic review showing lower blood pressure at 6 to 7 years of age for infants “ever breastfed.” [55] In older children and adolescents, the effect of rapid early weight gain remains but the effect of current weight or body mass index (BMI) becomes stronger. Lurbe et al. [56] found at 5 years of age, blood pressure and metabolic abnormalities were associated with birth weight and current BMI while at 10 years of age, current BMI became the strongest predictive factor. Incorporation of counseling on avoidance of obesity in these high-risk children should become part of routine primary prevention of early cardiovascular disease.
2. Cardiovascular disease risk in adulthood
Adults born preterm have higher office and ambulatory blood pressure and a higher risk (1.2X to 2.5X) of hypertension than those born at term [57]. In the Cardiovascular Risk in Young Finns Study of adults 41 years of age, those born preterm and SGA had higher SBP than preterm born AGA and more hypertension than those born at term [58]. Some of the adulthood studies have not differentiated between those born premature only from those born growth restricted which is unfortunate as there seems to be a difference in vascular phenotype between these groups. Lazdam et al. [59] showed that at 25 years of age, adults who were born premature after a normotensive pregnancy had more arterial stiffness while those born premature after a hypertensive pregnancy (often associated with intrauterine growth restriction) had higher carotid intima media thickness and impaired endothelial dysfunction. The researchers also showed that all adults born preterm also had higher central and peripheral BP, and higher glucose, cholesterol and low-density lipoprotein compared to term-born adults. Based on both childhood and adulthood vascular studies, it seems that those born premature have impaired or immature vascular development leading to arterial stiffness with age while in those with intrauterine growth restriction there is more endothelial dysfunction and differences in vascular reactivity [52,59].
The adulthood blood pressure level and risk of hypertension are also associated with lower birth weight and rapid early weight gain but the effect is often attenuated when adjusted for adulthood BMI (Table 1). Early rapid weight gain effects on blood pressure are most pronounced in those born SGA. Law et al. [60] found that systolic blood pressure increased for each decrease in birth weight standard deviation score and for each increase in early weight gain standard deviation score. The interaction is complex with evidence from the Fels Longitudinal Study showing that a higher increase in weight-for-length z score in the first 2 years of life was associated with higher adulthood BMI and systolic blood pressure with the strongest effect in the most recent cohort during the obesity epidemic [61]. This brings into question environmental or food quality factors potentially mediating some of these effects. Yet even in adults, a systematic review has shown that rapid growth in those born SGA may be associated with higher blood pressure, higher BMI and more insulin resistance but is also associated with better cognition at 17–26 years of age [62]. Unfortunately there is no evidence to suggest the optimal amount of catch-up growth to protect neurocognition while limiting cardiovascular disease risks.
3. Renal consequences of prematurity
There are not only cardiovascular abnormalities but also renal consequences of prematurity and early life events. A systematic review of preterm birth effects on kidneys found those born preterm had a lower glomerular filtration rate, lower effective renal plasma flow, and a higher urine albumin to creatinine ratio compared to those born at term [63]. They also found smaller kidney length and kidney volume in addition to higher blood pressure in those born preterm. In a group of children with proteinuric kidney disease, Abitbol et al. [64] demonstrated the compounding effect of obesity on reducing kidney survival in those born preterm. The children who were born preterm and became obese experienced significant loss of kidney function while all nonobese preterm children had preserved kidney function in this study. There is also evidence that children who are born preterm and experience neonatal acute kidney injury have more kidney dysfunction, as determined by reduced glomerular filtration rate, proteinuria, or hypertension, than those without acute kidney injury events [65]. Given the close association between kidney disease, hypertension, and cardiovascular disease, kidney health should also be checked periodically along with blood pressure during long-term health surveillance in the premature population.
4. Pathological evidence for a mechanism
Antenatal kidney development is not complete until around 36 weeks’ gestational age so most premature infants are born prior to renal maturation. There is a natural variability in the number of glomerular generations (layers of glomeruli) but the earlier in gestation that a neonate is born the fewer the layers of viable glomeruli [66]. When birth happens prior to completion of nephrogenesis, there is accelerated postnatal glomerular maturation but the consequence can be development of larger and/or abnormal glomeruli [67]. It seems that the rapid maturation period only occurs over the first several weeks of life regardless of gestational age at delivery and then further maturation ceases [68]. In addition, neonates that experience acute kidney injury have further reduced glomerulogenesis. Early work by Brenner et al. [69] postulated that a reduced nephron endowment, whether congenital or acquired, can lead to hypertension due to a reduced ability to excrete salt. Barker also recognized the association of low birth weight with adulthood cardiovascular disease and hypothesized an association with inadequate fetal nutrition and growth leading to abnormal vascular programming [70]. Putting the hypotheses together, Mackenzie et al. [71] proposed that nephron and pancreatic beta-cell endowment may be determined by genetic and in utero factors, and if offspring are born with a low complement of nephrons into an environment of nutritional plenty that the kidney would develop glomerular hyperfiltration, glomerulosclerosis, and the person experience hypertension, diabetes, and hyperlipidemia. Pathological and clinical studies since these early papers continue to support the nephron endowment hypotheses.
Conclusions
Early life exposures can have an impact on neonatal, childhood, and adulthood cardiovascular and kidney disease risks. Neonatal hypertension is not common but needs to be diagnosed and managed appropriately to avoid complications. In the infants with prematurity-related hypertension, the abnormal blood pressure usually resolves in the first year or two but all premature infants, especially those born growth restricted, carry a lifetime risk for later cardiovascular and kidney disease. While significant knowledge has been gained in the area, there are still many outstanding questions including how to best prevent, identify and manage the early and later cardiovascular risks. In addition, primary care providers need to be aware of the longer term risks associated with prematurity and early life events so that they can screen for and manage these complications in the aging premature population.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.