Efficacies of different treatment strategies for infants hospitalized with acute bronchiolitis
Article information
Abstract
Background
Acute bronchiolitis is a common cause of hospitalization during infancy that carries significant morbidity and mortality rates.
Purpose
This study compared the efficacy of different treatment modalities for infants with bronchiolitis in terms of hospital stay and clinical severity scores.
Methods
The PubMed database was searched for relevant studies. Eligibility criteria included double-blind randomized controlled trial design, assessment of the effect of treatment on bronchiolitis in infants under 2 years of age, and publication in English from inception through July 31, 2020. The primary efficacy outcome was the length of hospital stay, while the secondary outcome was the clinical severity score. The standardized treatment effect and standard error of the effect size were calculated.
Results
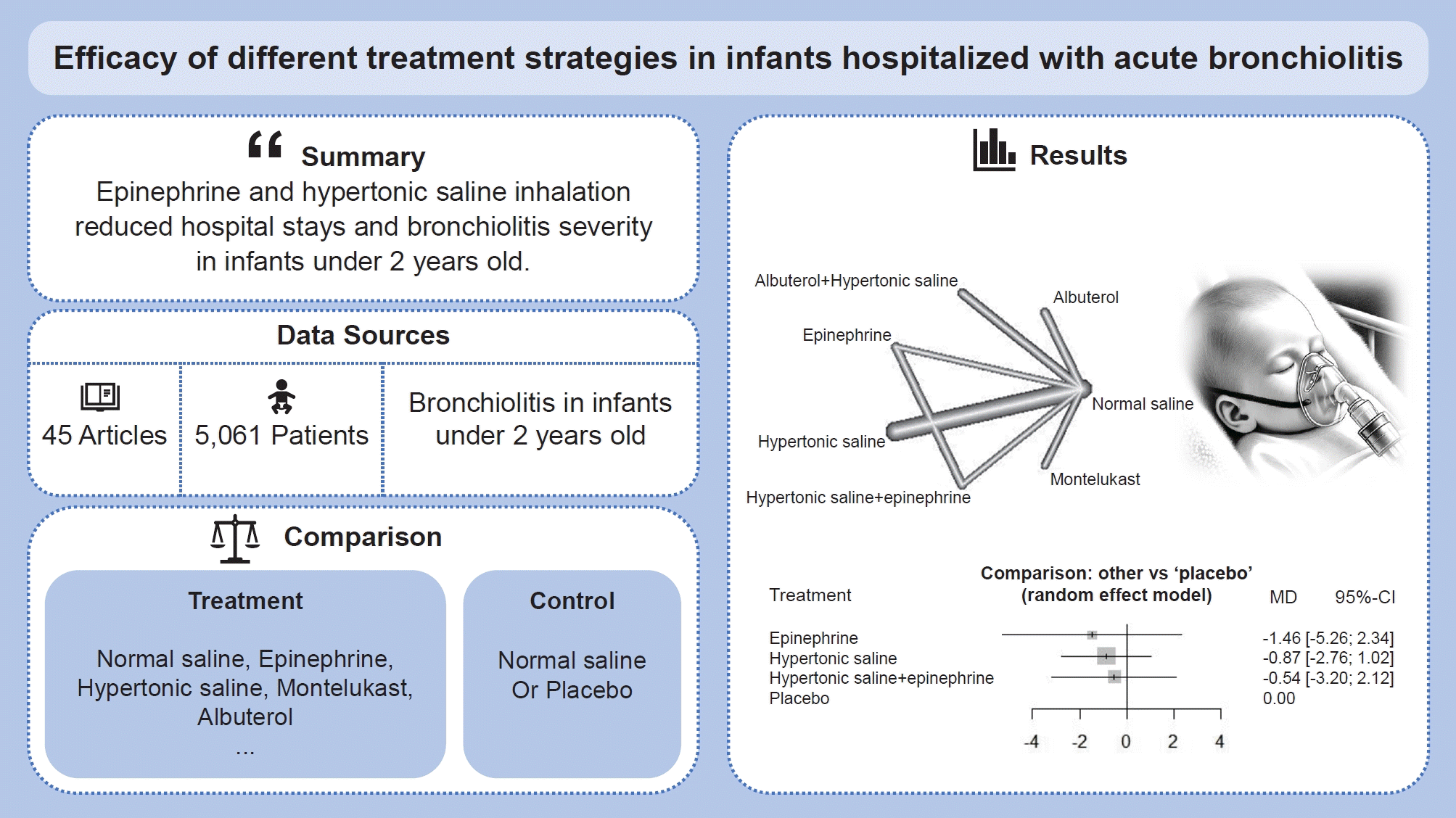
We identified 45 randomized controlled trials of 24 pairwise comparisons. These 45 trials included 5,061 participants and investigated 13 types of interventions (12 active, 1 placebo). Inhalation therapy with epinephrine (standard mean difference [SMD], -0.41; 95% confidence interval [CI], -0.8 to -0.03) and hypertonic saline (SMD, -0.29; 95% CI, -0.55 to -0.03) reduced the length of hospital stay compared with normal saline. Hypertonic saline was the most effective at improving the clinical severity score (SMD, -0.52; 95% CI, -0.95 to -0.10).
Conclusion
Inhalation therapy with epinephrine and hypertonic saline reduced the length of hospital stay and the clinical severity of bronchiolitis among infants under 2 years of age.
Key message
· This study analyzed 45 randomized controlled trials (5,061 participants, 13 interventions) of the comparative efficacies of treatments for acute bronchiolitis in infants.
· Inhalation therapy with epinephrine and hypertonic saline significantly reduced the length of hospital stay compared with normal saline.
· Hypertonic saline had the greatest ability to improve the clinical severity score of bronchiolitis in infants younger than 2 years of age.
Graphical abstract. MD, mean difference; CI, confidence interval.
Introduction
Acute bronchiolitis is the leading cause of morbidity among young children and infants under 2 years old world wide [1-4]. Respiratory syncytial virus (RSV) is the major etiologic agent for bronchiolitis [1,2]. Despite bronchiolitis having a significant impact on public health, its clinical management strategies vary considerably [3]. Supportive care including fluid intake, ensuring oxygen exchange, and feeding, has been the standard treatment for acute bronchiolitis [4]. Nevertheless, several studies have sought to find the most appropriate treatment for this condition [5].
Randomized controlled trials (RCTs) conducted to examine the efficacy of corticosteroids, bronchodilators, and other treatments, such as epinephrine, hypertonic saline, ribavirin, and rhDNase [6-10]. None of these, however, was found to significantly affect the rate of clinical improvement. A meta-analysis of studies assessing bronchiolitis treatment modalities might enable more practical and methodological approaches. To date, more than 20 meta-analyses have been conducted to evaluate the efficacy of epinephrine, bronchodilators, and hypertonic saline in the treatment of bronchiolitis [11,12]. These conventional meta-analyses involved only pairwise comparisons, making it hard to compare multiple treatment modalities or to assess the interactions of multiple therapies [13]. However, meta-analyses based on both direct and indirect comparisons for the same outcome may enable assessment of the relative efficacy of several treatments [13,14].
The present study was designed to integrate evidence of interventions currently used to manage bronchiolitis in infants, by comparing the effectiveness of these modalities on length of hospital stay and Wang score representing clinical severity [15]. The PubMed database was searched to identify RCTs comparing therapeutic regimens in infants with bronchiolitis, followed by a network meta-analysis to determine effective treatments for this condition.
Methods
1. Information sources and search strategy
PubMed was searched for relevant articles in English published through 31 July 2020. The main search terms were “bronchiolitis,” “wheezing,” “respiratory syncytial virus,” and “RSV.”
Included studies were restricted to “clinical trials” and “randomized controlled trials.” The titles and abstracts of studies identified by the initial search were reviewed to determine their relevance. The bibliographies of selected studies were also reviewed.
2. Eligibility criteria
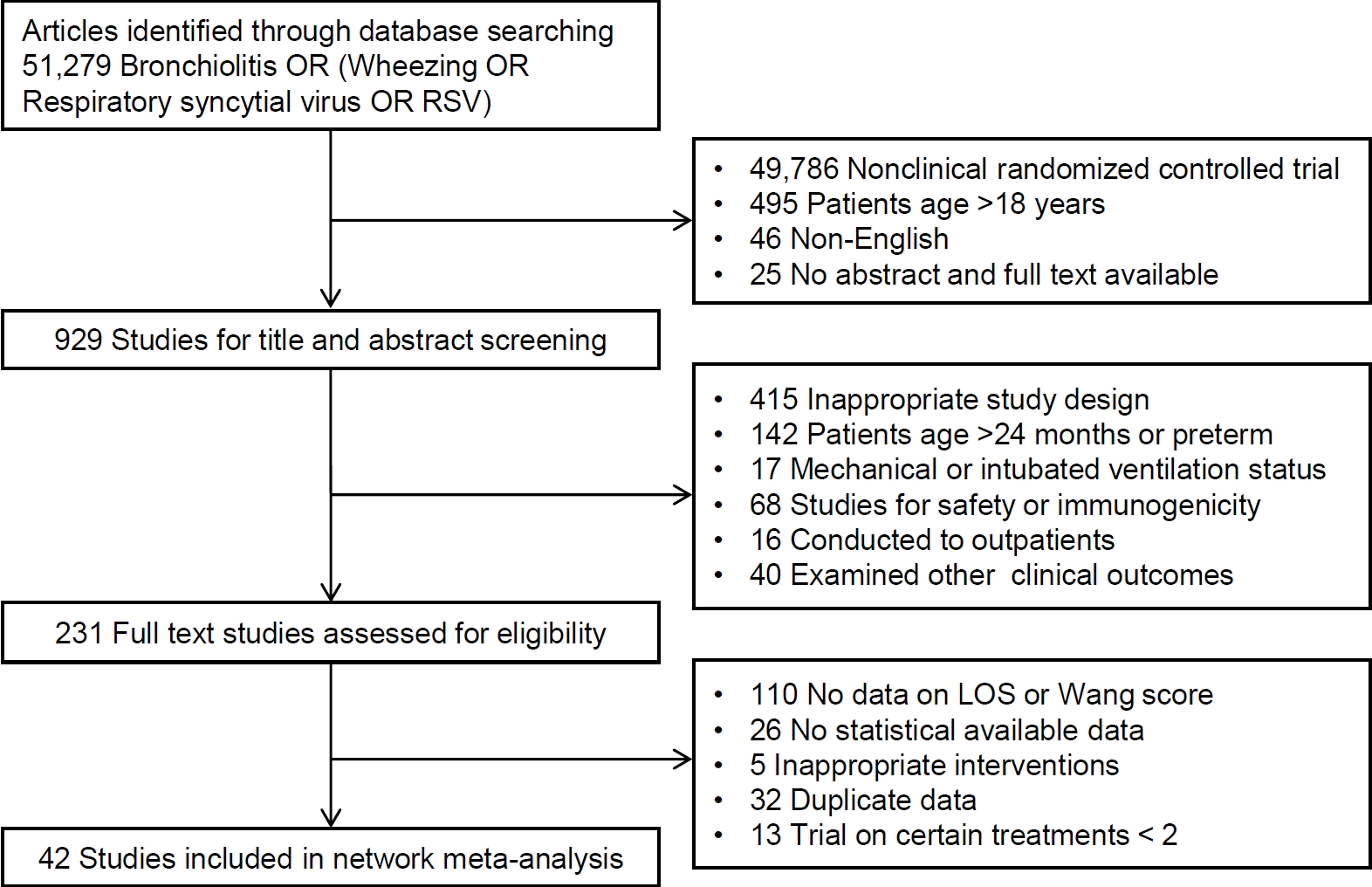
Studies were included if their populations consisted of inpatients aged <24 months who were hospitalized with bronchiolitis, wheezing, and/or RSV infection; if these studies compared 2 or more types of treatment; and if they reported Wang scores and/or length of hospital stay. Retrospective and long/short term follow-up studies were excluded, as were studies in populations with other underlying diseases, such as cystic fibrosis, chronic pulmonary diseases, congenital heart disease, and immunodeficiency. Also excluded were trials that examined other outcomes, including gastrointestinal symptoms, pulmonary function, oxygen saturation, hospitalization rate, hypothalamic-pituitary-adrenal axis function, serum cytokine and chemokine concentrations, and heart rate. In addition, trials that examined the effects of herbs and Chinese traditional medicines were excluded.
3. Study selection
Two investigators (MYH and HJ)independently screened articles by title and abstract according to the inclusion criteria. These investigators subsequently read the full text of selected articles. Studies were excluded if they (1) did not report data on length of hospital stay or average Wang score after hospitalization, (2) did not include statistical comparisons, or (3) were performed to assess chest physiotherapy. In addition, studies were excluded if they were the only studies to assess certain treatments. The mean and standard deviation for the 2 primary outcomes; length of hospital stay and Wang scores, were determined. Data unavailable from the original publications were calculated using presented results. Trial details (e.g., study ID, first author, journal, publication year, countries, patient characteristics, treatments, and outcomes) were recorded on a spreadsheet.
4. Quality assessment and risk of bias
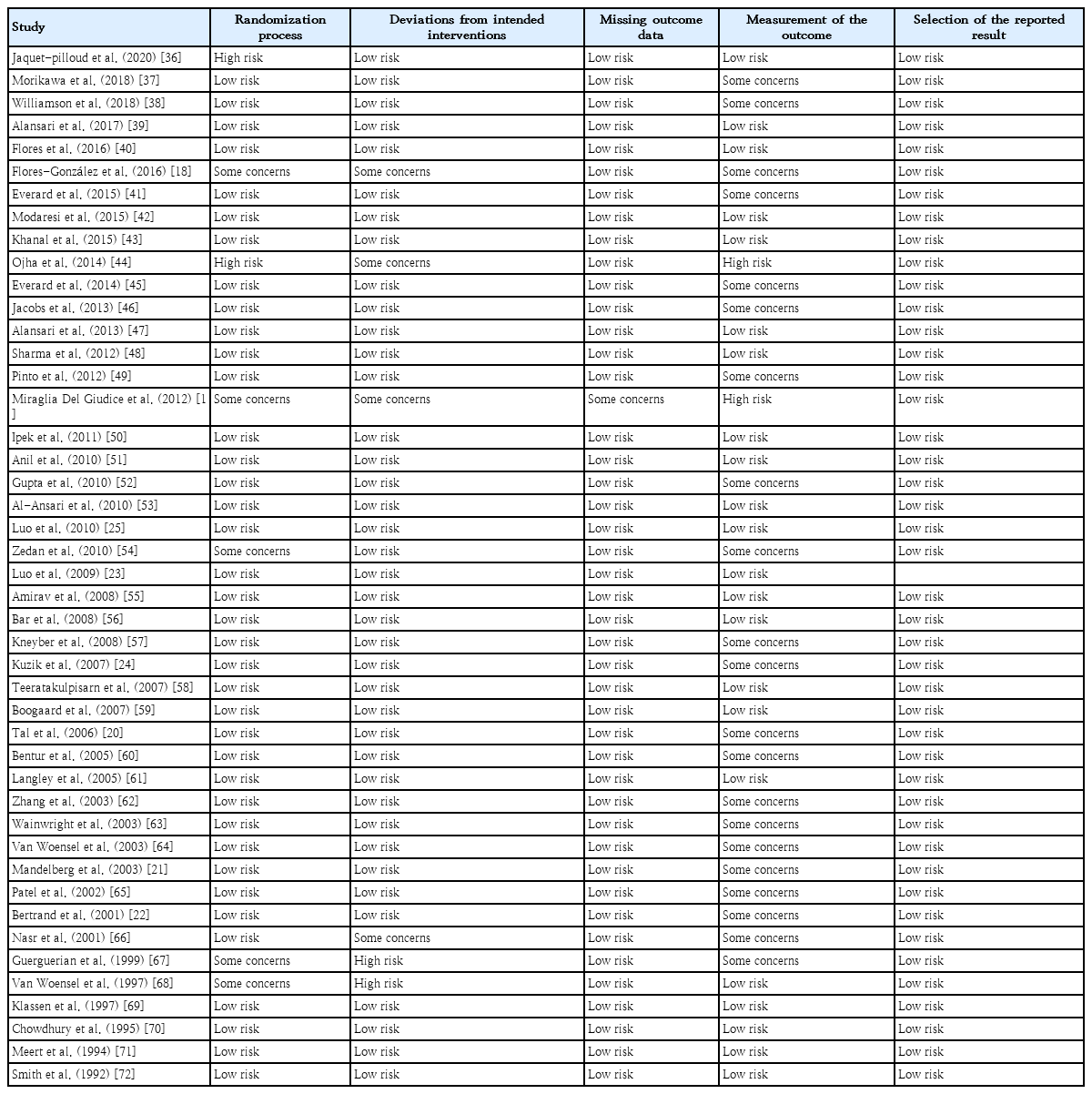
Overall risk of bias was evaluated on each included study using the revised Cochrane risk of bias tool for randomized clinical trials [16]. This tool consists of 5 domains: randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome and selection of the reported result. Results were scored as low, high, or some concerns.
5. Data synthesis and statistical analysis
The network meta-analysis was performed using a random effects model with a frequentist approach.All statistical analyses were conducted using the netmeta package (version 1.2-1) R version 4.0.2. For data synthesis in the analysis, the standardized treatment effect (TE) and standard error of effect size were calculated using EffectSizeCalculator (CEM, Ushaw college, Durham, England). Normal saline was distinguished from placebo because normal saline may have active effects on bronchiolitis [9]. Normal saline was defined as the common comparator in this study. Heterogeneity was assessed using the τ2 measure. I2 statistics are commonly used to measure heterogeneity in meta-analyses, with I2 ranging from 0% to 100%. These statistics are easy to interpret and do not vary with the number of studies [17]. However, the I2 value can increase with the number of patients included in the meta-analysis. Because I2 statistics for heterogeneity can increase with the number of patients included in the meta-analysis, global statistical heterogeneity was evaluated across all comparisons with the τ2 measure using the netmeta statistical package. Each treatment was ranked by P score, which is analogous to the surface under the cumulative ranking curve in Bayesian network meta-analysis with a higher score indicating a greater probability of being beneficial. Forest plots were calculated to visualize the directionality and confidence intervals (CIs) of each TE. Network diagrams were plotted to show the overall structure of the treatment network for each outcome.
Results
1. Study selection
Fig. 1 shows the flow of study selection. The initial database search identified 51,279 articles. Of the 929 studies eligible for title and abstract screening, 698 did not meet our inclusion criteria and were excluded, as were 25 articles not available in full text version. Overall, 45 articles with a total of 5,061 patients were included in the network meta-analysis.
2. Study characteristics
Table 1 shows the number of included studies by treatment comparison, and Table 2 shows the detailed characteristics of the included studies. The included studies reported 24 treatment comparisons. Most involved hypertonic saline as an intervention, with 10 studies comparing hypertonic saline with normal saline. These 45 trials were performed in numerous countries and evaluated the effects of 12 active treatments, including normal saline, epinephrine, magnesium sulfate, hypertonic saline, montelukast, ribavirin, systemic steroids (dexamethasone and prednisolone), furosemide, antibiotics, ipratropium bromide, albuterol, and rhDNase. All included studies were conducted in the inpatient setting. Ten studies reported Wang scores.
3. Risk of bias
Eighteen studies were categorized as possibly having risk of overall bias based on outcome measurements (Table 3). These 18 studies did not clearly describe the criteria for measuring outcomes. Five studies were categorized as having a high risk for overall bias, notably for the randomization process and outcome measurements. Blinding of participants and staff was not feasible owing to the nature of the intervention, thus limiting protection against performance and detection bias. The funnel plots are shown in Supplementary Fig. 1.
4. Network meta-analysis
Figs. 2A and 3A display the efficacy estimated using network meta-analysis of indirect comparisons of each treatment with that of normal saline, as measured by duration of hospitalization and averageWang score, respectively. Epinephrine (standardized mean difference [SMD], -0.41; 95% CI, -0.8 to -0.03) and hypertonic saline (SMD, -0.29; 95% CI -0.55 to -0.03) were the most effective treatments for decreasing duration of hospital stay. Compared with normal saline, epinephrine and hypertonic saline reduce 0.41 days and 0.29 days of hospitalization, respectively.
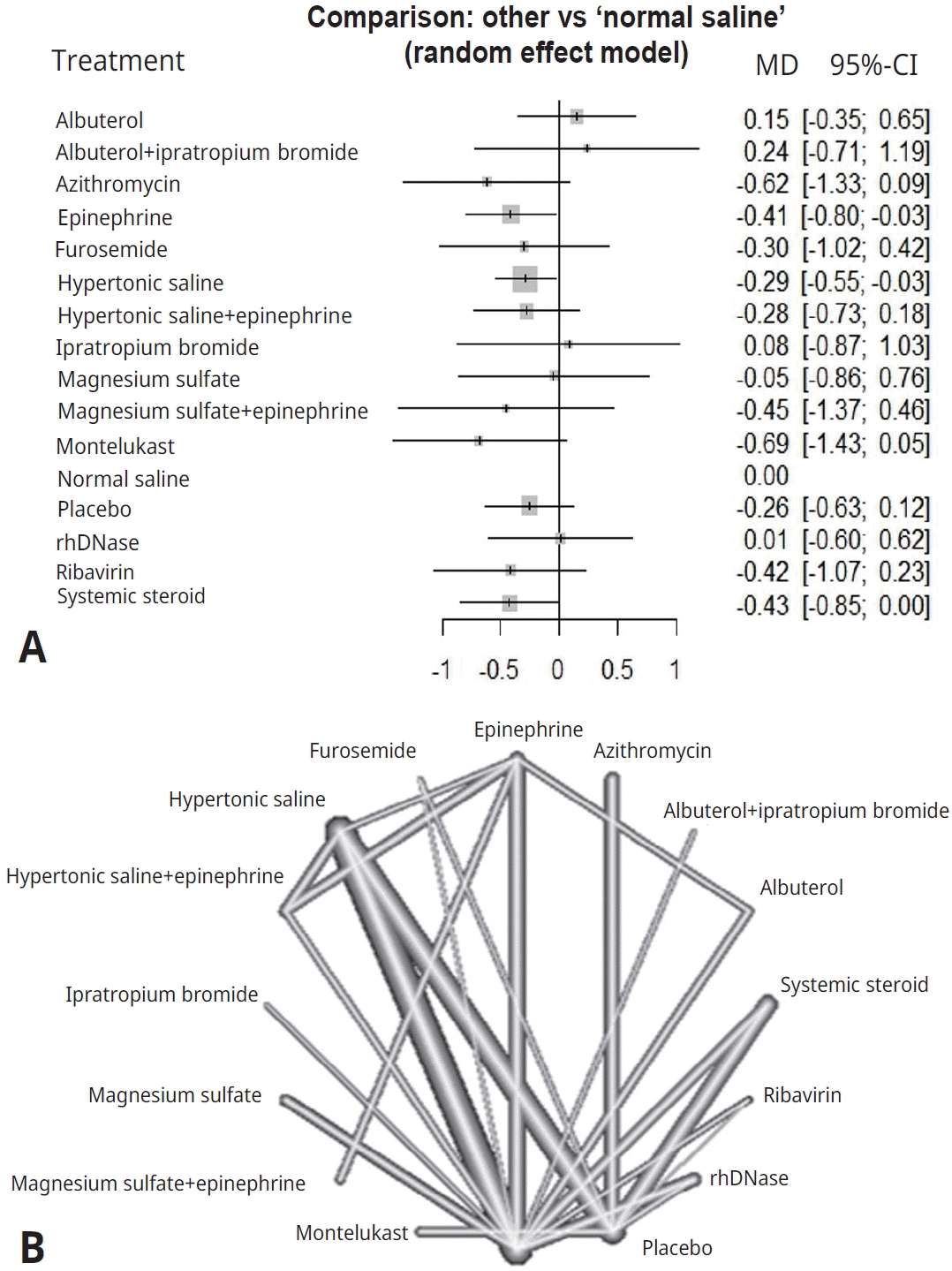
(A) Forest plot of lengths of hospital stay. (B) Network graph of lengths of hospital stay. The nodes indicate treatments, while the interconnecting lines (edges) indicate direct comparisons. The thickness of each edge is proportional to the precision of the compared estimate. MD, mean difference; CI, confidence interval.
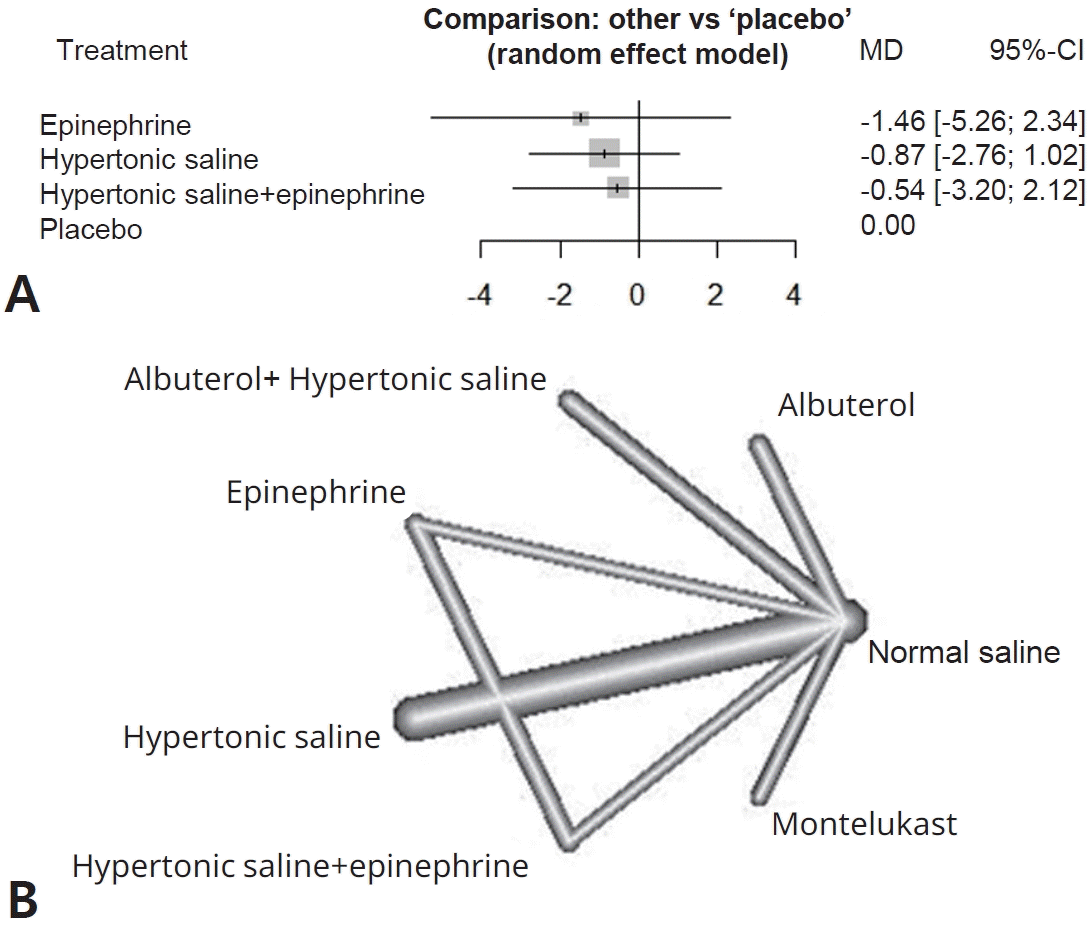
(A) Forest plot of Wang scores representing clinical severity. (B) Network graph of Wang scores representing clinical severity. MD, mean difference; CI, confidence interval.
The estimated heterogeneity (τ2) was 0.147 for duration of hospital stay and 0.233 for Wang score, which represented a moderate degree of heterogeneity. Inconsistencies of comparisons are presented in Supplementary Fig. 2.
For clinical scores, 10 studies had useable data. Hypertonic saline is considered to have an effect on improving Wang score (SMD, -0.52; 95% CI-0.95 to -0.1), which means reducing clinical severity. Forest plots presenting all comparisons based on both direct and indirect evidence for duration of hospitalization and Wang scores are shown in Supplementary Fig. 3A and B, respectively. The area of each box surrounding the estimate was proportional to the weighting in the meta-analysis.
A network graph (Figs. 2B and 3B) showed that the network is a sparsely connected intersection of 2 ‘star’ networks, the principal comparators being placebo and normal saline for length of hospital stay, and normal saline for Wang score. The thickness of each edge corresponds to the number of trials and represents the precision of the estimate. Montelukast had the highest probability of being the best treatment for bronchiolitis (P score=0.833), followed by azithromycin (P score=0.792), systemic steroids (P score=0.682), and epinephrine (P score=0.675) (Table 4).
Discussion
This systematic review and meta-analysis comparing different therapy strategies in infants hospitalized for bronchiolitis included 45 RCTs and 5,061 patients. Based on both direct and indirect evidence, this study compared the therapeutic effects of 12 types of intervention on length of hospital stay and Wang score. Of these 12 treatments, epinephrine and hypertonic saline were found to be most beneficial in reducing length of hospital stay, and hypertonic saline was found to help improve Wang score. Twenty RCTs compared epinephrine, hypertonic saline, normal saline and placebo, with 8 of these studies yielding results similar to ours [18-25].
Among the infants with bronchiolitis, approximately 1%–2% of whom require hospitalization [26]. In the United States, it is reported at 18% of all hospitalizations in children under 2 years old in 2016. The proportion of infants with bronchiolitis requiring intensive care unit admission has previously been accounted for 6% to 22% [27]. Based on our results, epinephrine and hypertonic saline can be considered as effective treatment options.
Nebulized epinephrine is generally used to treat significant respiratory distress in children. Epinephrine possesses both alpha-adrenergic and beta-adrenergic properties. Its alpha-adrenergic properties reduce airway edema and vasoconstriction [28,29]. Thus, epinephrine can be theoretically possible to be a benefit of managing bronchiolitis. A Cochrane review [28] demonstrated that epinephrine significantly decrease the risk of hospitalization In addition,the combination of epinephrine and hypertonic saline has been reported to reduce the length of hospital stay [10]. Although several studies found that epinephrine improved clinical outcomes in outpatients [7,30,31]. other studies have yielded conflicting results [32]. Hypertonic saline also can improve mucociliary clearance and reduce edema of the airway through absorbing water from the mucosa and submucosa [4]. Two meta-analysis studies [23,33] derived that nebulized hypertonic saline has the potential to reduce the risk of hospitalization in infants with bronchiolitis. These properties would be helpful to infants with bronchiolitis. We also calculated the P score to identify the relative therapeutic superiority of interventions. Although montelukast had the highest P score, it should be noted that CIs must also be taken into account to determine the best treatment [34,35]. There are only 2 trials comparing the effect of montelukast and placebo for the length of hospital stay, and each trial suggested contradictory results. In this context, we considered both the P score and forest plot. Consequently, epinephrine and hypertonic saline are regarded to have an effect to reduce the length of hospital stay. The strength of our study includes comprehensive integration of individual RCT on effective treatment for bronchiolitis in infants. Contrary to standard meta-analysis which can only statistically combine 2 interventions, the methodology used in this study enabled indirect comparisons of all pairs of treatments for the same outcomes. Furthermore, we assessed relative superiority between treatments that have not been directly compared in RCT. This result could provide research insights to choose the best treatments in managing bronchiolitis.
There are several limitations in the present study. First, we selected 2 primary outcomes, the length of hospital stays and the Wang score, due to the lack of sufficient other outcome data. However, this approach is less precise because of high heterogeneity. Since the measurement times for the Wang score varied by trial, we decided to use the Wang score from the second day after enrollment. In this process, it was difficult to obtain sufficient data for our analysis, which may be the reason for the high heterogeneity. There are many different outcomes reflecting clinical improvement in bronchiolitis, such as the respiratory distress assessment instrument score, oxygen saturation, and respiratory rate. Thus, different results may be derived depending on the outcomes selected. Second, several included trials did not adequately describe the randomization process, outcome measurement sequence, or deviations from intended interventions. These limitations in the individual included trials could lead to potential bias in this study. Third, we did not account for variations within the same treatment—specifically, we were unable to thoroughly compare differences in concentrations of hypertonic saline or the effects of epinephrine versus racemic epinephrine. Such variations could result in outcome deviations across different doses.
In summary, epinephrine and hypertonic saline can be beneficial to reduce length of hospital stay and epinephrine is considered to improve Wang score in bronchiolitis in infants under 2 years old.Further studies need for validating the effectiveness of treatment listed in this study.
Supplementary materials
Supplementary Figs. 1-3 can be found via https://doi.org/10.3345/cep.2023.01676.
Supplementary Fig. 1. (A) Funnel plot: length of hospital stays. (B) Funnel plot: Wang score.
cep-2023-01676-Supplementary-Fig-1.pdfSupplementary Fig. 2. Heat plot based on random-effects model.
cep-2023-01676-Supplementary-Fig-2.pdfSupplementary Fig. 3. (A) Forest plot displaying all comparisons for which there is both direct and indirect evidence: length of hospital stays. (B) Forest plot displaying all comparisons for which there is both direct and indirect evidence: Wang Score. CI, confidence interval.
cep-2023-01676-Supplementary-Fig-3.pdfNotes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This study was supported by grant HR22C1605-C1 from the Korea Health Technology Research and Development Project through the Korea Health Industry Development Institute funded by the Ministry of Health and Welfare, Republic of Korea.
Author contribution
Conceptualization: MYH; Data curation: MYH; Formal analysis: HJ, HH; Funding acquisition: MYH; Methodology: MYH; Project administration: HJ, DP; Visualization: HJ; Writing- original draft: HJ; Writingreview & editing: HJ, EKH, JHK, JS, HB, YHS, HMJ