Prolonged cerebral oxygenation surveillance with algorithm-based management: a neurocritical care bundle for extremely preterm infants
Article information
Abstract
Background
Cerebral hypoxia-ischemia impairs brain development in extremely preterm infants and is associated with poor neurological outcomes. Near-infrared spectroscopy (NIRS) is a noninvasive continuous monitoring method for regional cerebral oxygen saturation (rcSO2).
Purpose
This study evaluated the clinical feasibility and neurological impact of a neurocritical care bundle that incorporates prolonged multidisciplinary hemodynamic monitoring and a stepwise management algorithm.
Methods
Preterm infants with a gestational age (GA) ≤28 weeks or birth weight (BW) ≤1,000 g were prospectively enrolled in a bundle group subjected to NIRS for rcSO2, electrical cardiometry for cardiac output, and daily brain and cardiac echography during the first 72 hours of life. Monitoring was repeated weekly in the first month and then monthly until discharge or the term-equivalent age (TEA) was reached. We implemented a stepwise management algorithm for treating cerebral hypoxia. The primary outcome was a composite of mortality and adverse neurological events (structural abnormalities or electroencephalogram-confirmed seizures) before discharge. The secondary outcomes were the physiological pattern of rcSO2 within the initial 72 hours and up to discharge or TEA.
Results
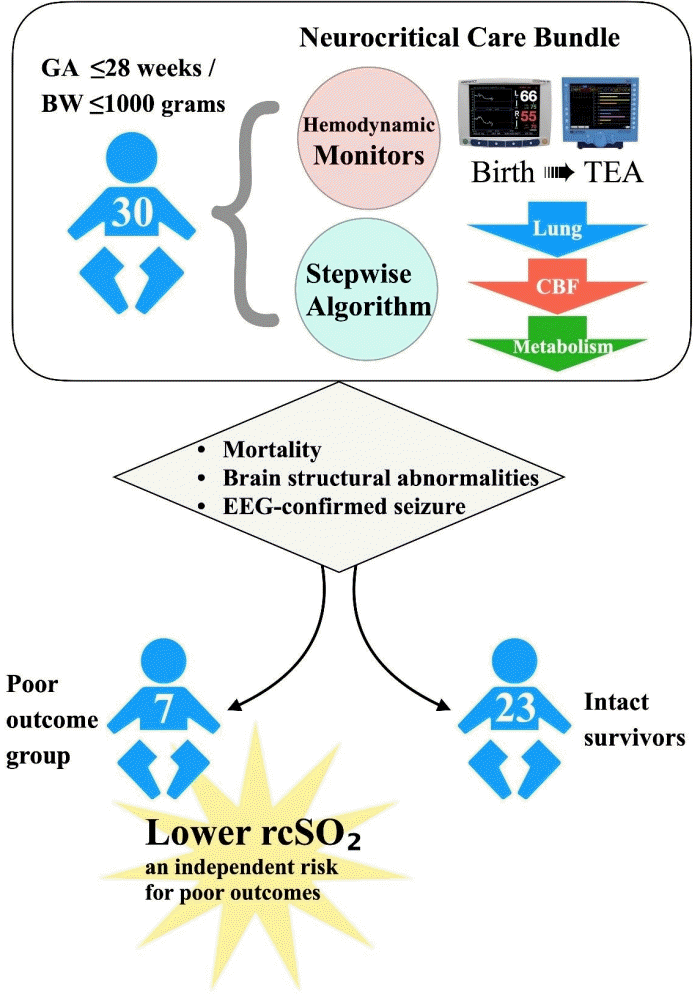
Thirty preterm infants (GA, 27.1±2.0 weeks; BW, 830±225 g) were enrolled in the bundle group. The mean time-averaged rcSO2 (66.8%±10.3%) was not associated with GA or BW. However, postnatal age appeared to influence physiological rcSO2 changes, given that rcSO2 values were higher during the initial 72 hours than at subsequent intervals. Seven infants (23.3%) had poor outcomes and significantly lower time-averaged rcSO2 (51.1% [50.0%–65.2%] vs. 71.8% [67.1%–73.1%], P=0.002). Multivariate regression indicated that a lower rcSO2 was an independent risk factor, and a 65% threshold showed an optimal predictive value for poor outcomes.
Conclusion
The neurocritical care bundle helped identify preterm infants at risk of cerebral hypoxia, and lower rcSO2 was an independent risk factor for composite mortality and adverse neurological outcomes.
Key message
Question: Is prolonged monitoring of regional cerebral oxygen saturation (rcSO2) and hemodynamic parameters a feasible approach? Can these measures predict the neurological outcomes in extremely preterm infants?
Finding: We used a neurocritical care bundle from birth to discharge or term-equivalent age. Infants with poor outcomes had significantly lower rcSO2 values.
Meaning: Understanding rcSO2 and hemodynamic parameters may help manage cerebral hypoxia and reduce neurological complications in extremely preterm infants.
Graphical abstract. GA, gestational age; BW, birth weight; TEA, term-equivalent age; CBF, cerebral blood flow; EEG, electroencephalogram; rcSO2, regional cerebral oxygen saturation.
Introduction
As the survival of extremely preterm infants improves, there is also an increase in the neurological disabilities in the survivors. Among those born with a birthweight lower than 1,000 grams, up to 50% would be diagnosed with neurodevelopmental impairment by 2 years of age [1,2]. Causative risk factors mainly point to cerebral hypoxia-ischemia related neuronal injury, and deranged brain growth afterwards [3-6]. To pursue the imperative goal of intact survival for these high-risk neonates, the concept of neurocritical bundle care has been increasingly emphasized and implemented. Strategies for neurocritical care provide an interface between the brain and other organ systems for comprehensive medical and specialized neurological support, so as to deliver sufficient oxygen to meet tissue metabolic demands. Patients in the neonatal intensive care unit (NICU) are among those who would benefit the most from neurocritical care. Yet in reality limitations usually come from reliable monitoring of the macro- or microcirculation in the tiny patients to ensure a normal physiology [7].
Near-infrared spectroscopy (NIRS) employs the distinct absorption properties of oxygenated and deoxygenated red blood cells when exposed to near-infrared light [8]. It serves as a monitoring device for measuring cerebral oxygenation. Regional cerebral oxygen saturation (rcSO2) represents tissue oxygen saturation of the brain, while fractional tissue oxygen extraction (FTOE), calculated as the formula: (SaO2–rcSO2)/SaO2, indicates oxygen utilization [9]. The balance between oxygen supply and utilization provides insight in neonatal cerebral pathophysiology. Normal rcSO2 is approximate 65%–85% in neonates, and a low rcSO2 <55% may cause adverse effects on the brain [5,10]. By integrating rcSO2 and FTOE, clinicians can explore potential causes of abnormal cerebral oxygenation and formulate appropriate responses [11,12].
To keep adequate tissue oxygenation also relies on an efficient cardiopulmonary function and an intact regional distributive mechanism. For brain tissue, delivery of oxygen consists of three major macro-circulatory components: cardiac output (CO), cerebral blood flow (CBF), and oxygen-saturated hemoglobin. Lately, the noninvasive impedance-based CO monitor has been increasingly applied and the reference values been validated in preterm infants [13-15]. In NICU, functional echography (fEcho) has become a mainstream to assess CBF while screening for intraventricular hemorrhage (IVH) or patent ductus arteriosus (PDA) [16]. These hemodynamic monitoring devices are currently available and clinically applied in preterm infants.
Previous study demonstrated that using NIRS to monitor rcSO2 and guide clinical management during the initial 72 hours of life could mitigate the burden of cerebral hypoxia in preterm infants [10]. However, the protective effect did not bring about an improved long-term outcome [17,18]. Some of the major critiques for not able to achieve the study goals include inadequate rcSO2 monitoring for all critical points of the hospital stay, the shortage of all-around multidisciplinary monitoring to assist diagnosis, and inconsistency of implementation of the proposed management guidelines.
In this study, we designed a neurocritical care bundle including NIRS, noninvasive CO monitor, and fEcho to monitor their cerebral hemodynamic status, and set up a step-wise triaging and treatment protocol for the remedy of its disturbances. By doing so, we aimed to secure better neurodevelopmental outcomes in the extremely low birth weight (BW) preterm infants.
Methods
1. Study population
This prospective study was conducted in the neonatal intensive care unit of Chang Gung Memorial Hospital Linkou Branch between September 2022 and July 2023. Preterm infants of gestational age (GA) no greater than 28 weeks or BW between 500–1,000 grams would be enrolled at birth, with the exclusion criteria of major congenital anomalies or evidence of severe perinatal asphyxia (5 minutes Apgar score <5) before enrollment.
An a priori power analysis was conducted for linear multiple regression using a random model. Assuming an effect size of R² 0.30, an alpha level of 0.05, and a statistical power of 0.80, the required sample size was calculated to be 28 subjects for detecting a significant association with one predictor variable. To ensure adequate power, a total of 30 preterm infants was planned for enrollment in the bundle care group. The Institutional Review Board of Chang Gung Medical Foundation approved this study (202102305B0C601) and written informed consents were obtained from study infants’ parents.
2. Monitoring devices
Hemodynamic monitoring devices included NIRS (INVOS 5100c, Medtronic, USA) with neonatal cerebral sensors for rcSO2 measurement, Aesculon electrical cardiometry (EC, Osypka Medical, Germany) for noninvasive CO assessment, and Acuson P300 ultrasound machine with a 4–11 MHz transducer (Siemens Healthcare, Germany) for cerebral and cardiac echographic evaluation. Target rcSO2 has been suggested to be ranging from 65% to 85% [10,11], even utilizing neonatal sensors [18], and cerebral hypoxia was defined as rcSO2 <55% [5,10]. The reference range of FTOE is suggestive 10%–40% [9], and normal CO was set at ≥0.15 L/kg/min based on our previous studies [15,19]. Appropriate blood pressure (BP) was defined as the value of mean BP greater than individual’s postmenstrual age in weeks.
Peak systolic velocity, end-diastolic velocity, and mean velocity of left middle cerebral artery (MCA) flow were measured using fEcho to assess CBF. Reference values for MCA flow in preterm infants were adapted from Romagnoli et al. [20], and the normal range was kept between 10th to 90th percentile [16,20]. A hemodynamically significant PDA (hsPDA) was defined as ductal diameter >2.0 mm or left atrium to aortic root (LA/Ao) ratio >1.4 which was also associated with abnormal organ blood flows [16].
3. Study protocol
For each eligible infant, a brain fEcho was performed prior to enrollment to screen for IVH. Once an infant was enrolled, a set of noninvasive hemodynamic monitors including NIRS+EC were applied within the 6 hours after birth and continuously measured rcSO2 and CO, as well as daily brain and cardiac fEcho, during the initial 72 hours of life. If the neonate remained stable, the NIRS+EC monitoring was scheduled weekly for at least 1-hour sessions during the first month and subsequently extended to monthly sessions until discharge or reaching term-equivalent age (TEA). Both brain and cardiac fEcho assessments were performed concurrently with each NIRS+EC monitoring session. The monitoring protocol is demonstrated in Fig. 1.
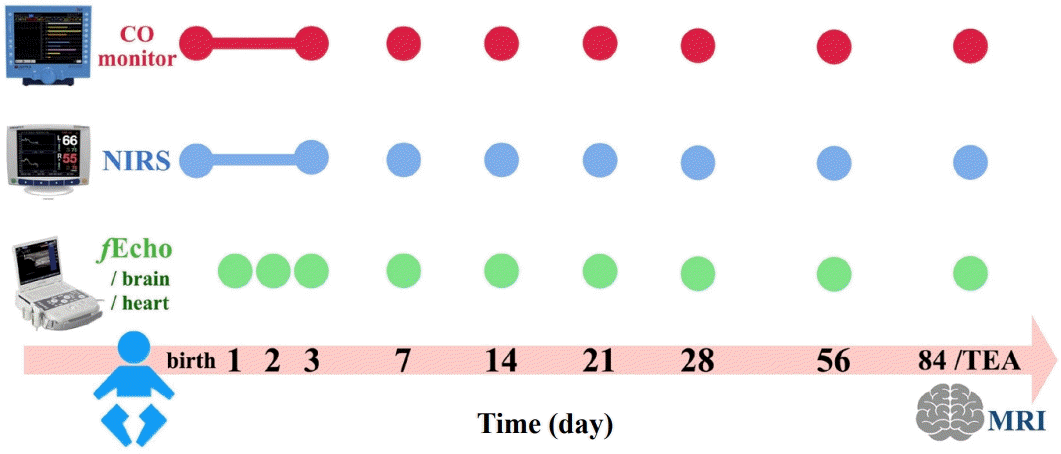
Study protocol. Continuous noninvasive CO monitoring and NIRS for rcSO2 were applied within 6 hours after birth and maintained during the initial 72 hours of life, followed by scheduled 1-hour sessions weekly during the first month and monthly thereafter until discharge or until TEA. fEcho of the brain and heart was performed daily for the first 3 days and then concurrently with each NIRS+CO monitoring session at later time points. Brain MRI was performed at TEA or before discharge to assess structural outcomes. CO, cardiac output; fEcho, functional echography; MRI, magnetic resonance imaging; NIRS, near-infrared spectroscopy; rcSO2, regional cerebral oxygen saturation; TEA, term-equivalent age.
4. Management algorithm
As previously published studies [10,11,18] and our own preliminary experiences, a management algorithm was designed to maintain infants’ rcSO2 and FTOE within target ranges. Since rcSO2 is correlated positively with SaO2 and negatively with abnormal CBF [19], our algorithm was following a rationale of “lung-CBF-metabolism” steps, as shown in Fig. 2.
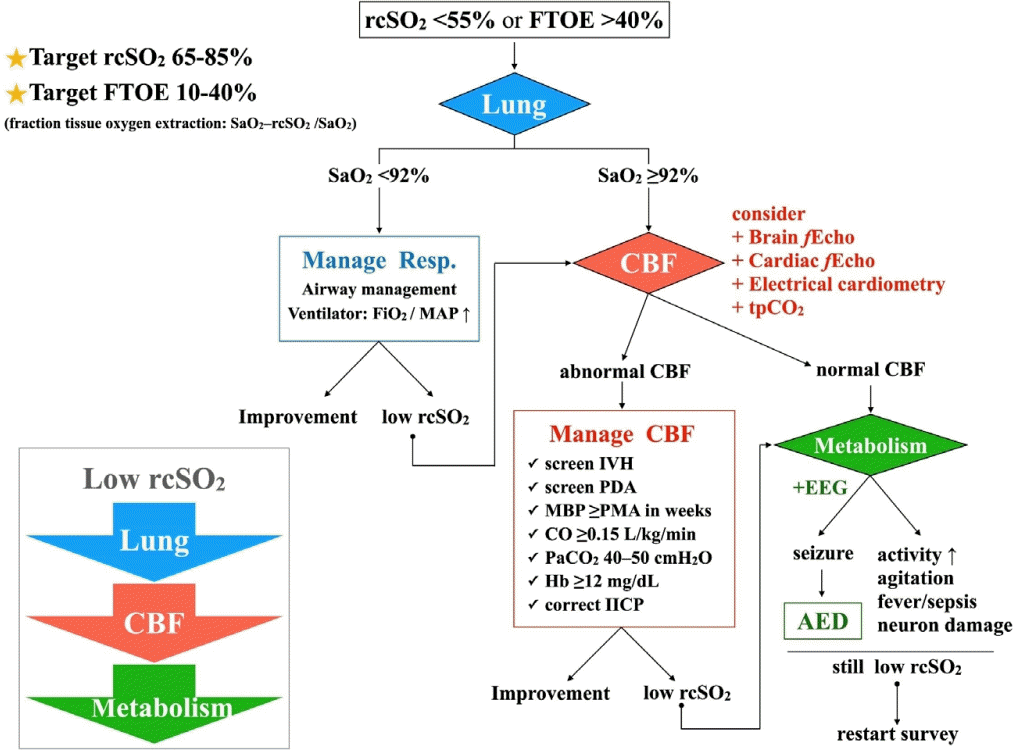
Management algorithm. A stepwise management algorithm for abnormally low cerebral saturation (rcSO2 <55% or FTOE >40%) based on the “lung-CBF-metabolism” rationale. AED, antiepileptic drug; CBF, cerebral blood flow; CO, cardiac output; EEG, electroencephalogram; fEcho, functional echography; FiO2, fraction of inspired oxygen; FTOE, fraction of tissue oxygen extraction; IICP, increased intracranial pressure; IVH, intraventricular hemorrhage; MAP, mean airway pressure; MBP, mean blood pressure; PDA, patent ductus arteriosus; PMA, postmenstrual age; rcSO2, regional cerebral oxygen saturation; Resp, respiration; SaO2, arterial oxygen saturation; tpCO2, transcutaneous carbon dioxide monitor.
5. Outcomes
Serial brain fEcho were conducted per study protocol, and IVH ≥3, periventricular leukomalacia (PVL) or ventriculomegaly confirmed by independent pediatric neurologists were considered abnormal. Additionally, brain magnetic resonance imaging (MRI) was obtained at TEA or before discharge, with abnormalities including IVH ≥3, PVL, abnormal white matter signals or cerebellar hemorrhage (CBH) identified by an independent radiologist. The primary outcome was the composite of mortality and adverse neurologic events including structural abnormality on brain fEcho or MRI, or electroencephalogram (EEG)-confirmed seizure, assessed at TEA or before discharge. The secondary outcomes were physiologic pattern of rcSO2 and FTOE during the early transitional period (within the first 72 hours), as well as their trajectories up to discharge or TEA. Additional analyses focused on rcSO2 and FTOE characteristics among preterm infants under specific conditions such as hsPDA or IVH. For mortality cases during hospitalization, continuous parameters (e.g., time-averaged rcSO2 and FTOE) were calculated based on each infant’s actual lifespan, and categorical outcomes were classified according to final clinical condition.
6. Statistics
Student t test or Mann-Whitney U test was used for parametric and nonparametric continuous data, respectively, whereas chi-square or Fisher exact test was used for categorical data. Time series variables were analyzed with repeated measure analysis of variance (RM-ANOVA) for intra- and between- variables difference. Time-averaged rcSO2 and FTOE were calculated based on mean value of respective specific time intervals to represent overall mean rcSO2 and FTOE (Supplementary Fig. 1). Receiver operating characteristic (ROC) curve was employed to determine the cerebral hypoxia cutoff for composite poor outcomes. Sample size estimation was performed using G*Power 3.1 (Heinrich Heine University, Germany) and statistical analyses were conducted with IBM SPSS Statistics ver. 24.0 (IBM Co., USA), with a significance level set at P value <0.05.
Results
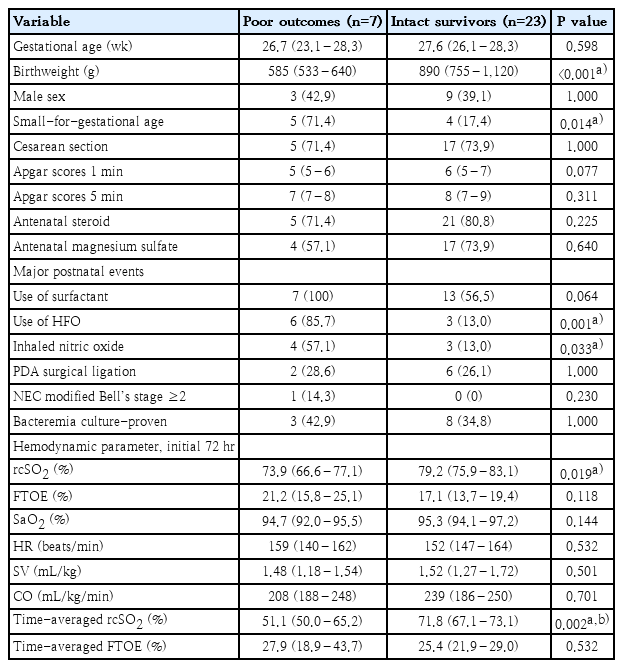
Eighty-four extremely preterm infants were admitted to our unit during the study period, and 8 infants who had received chest compressions or experienced perinatal asphyxia were excluded from enrollment. Because there was only one set of NIRS+EC monitors was available in situ, 44 of the remaining infants were unable to be enrolled either because the monitor device was still in use or only one twin could participate. Two additional infants were excluded due to parental refusal. Consequently, a total of 30 extremely preterm infants (GA, 27.1±2.0 wk; BW, 830±225 g) were included in the neurocritical care bundle group. The enrollment flowchart is demonstrated in Supplementary Fig. 2. Patients’ demographic characteristics are presented in Table 1.
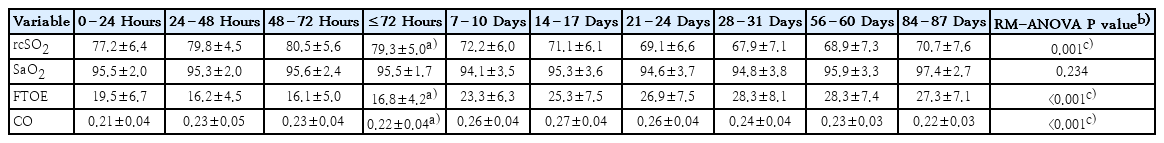
Within the initial 72 hours, the bundle group had mean rcSO2, SaO2 and FTOE of 77.8%±5.7%, 95.1%±1.9% and 17.8%±4.9%, respectively. For this time period, the median duration of rcSO2 <55% was 0.24% (0.12–1.13) of the time. Their overall time-averaged rcSO2 and FTOE were 66.8%±10.3% and 27.7%±10.0%. Neither 72-hour nor overall time-averaged rcSO2 or FTOE correlated with an individual's GA or BW. Also, there was no rcSO2 or FTOE difference seen between different sex, method of delivery or if patient was small-for-gestational-age (SGA, BW<10th percentile) or not. However, we noticed that infants with hsPDA or IVH had specific cerebral saturation patterns. Infants whose hsPDA fulfilled both criteria of diameter >2.0 mm and LA/Ao >1.4 had a tendency of lower rcSO2, higher FTOE, and a significantly higher CO, comparing to infants with only one criterion for hsPDA or infants without hsPDA. In addition, infants who required surgical ligation of PDA had significantly lower rcSO2 and higher FTOE than those did not receive ligation (RM-ANOVA between-group P=0.020 and P=0.024, respectively), especially from postnatal age 7 through 28 days. The trends of lower rcSO2 and higher FTOE persisted even after ligation (Fig. 3A and B). In addition, 2 infants developed high-grade IVH, and subsequent investigation revealed that they had significantly low rcSO2 and high FTOE after high-grade IVH occurred. After excluding 4 mortality cases (2 of them also with high-grade IVH), data from 26 infants (20 without IVH, 6 with grade 1–2 IVH) showed no significant differences in either rcSO2 or FTOE at any specific time points. Furthermore, the trends in rcSO2 and FTOE did not vary much among infants without IVH and with grade 1–2 IVH (RM-ANOVA between-group P=0.403 and P=0.451, respectively) (Fig. 3C and D).
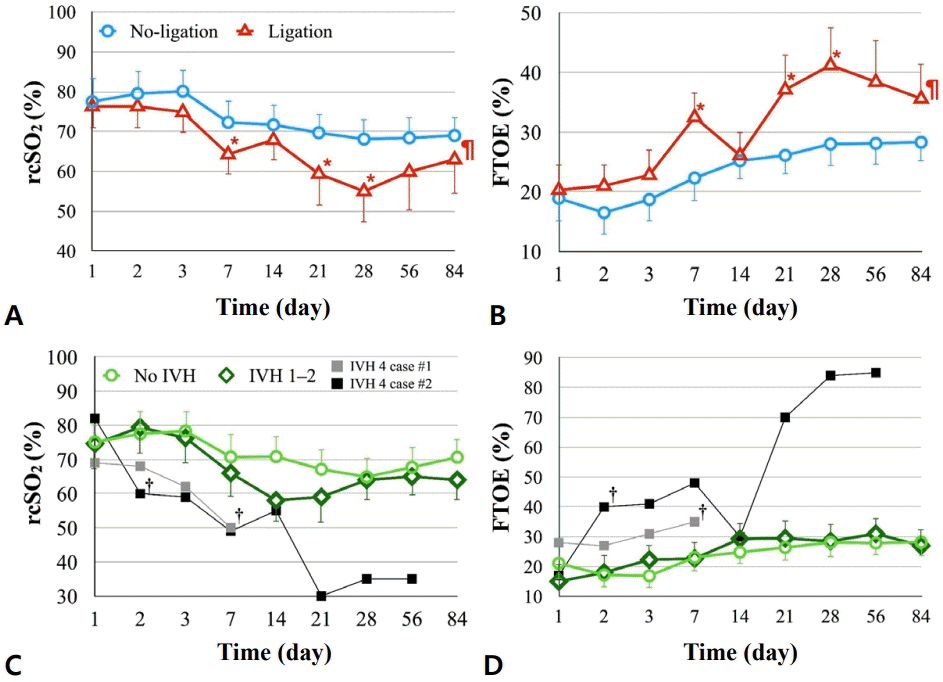
rcSO2 and FTOE changes in extremely preterm infants by PDA status and IVH severity. (A) rcSO2 was significantly lower in infants who underwent PDA ligation (red triangles) than in those who did not require ligation (blue circles) (RM-ANOVA, P=0.020)(¶), with the most notable difference on postnatal days 7, 21, and 28 (*). The pilcrow (¶) symbol indicates significant between-group trending difference, whereas an asterisk (*) symbol indicates significant difference at specific timing points. (B) FTOE expression was correspondingly higher in the ligation group (RMANOVA, P=0.024)(¶), with the greatest differences observed on postnatal days 7, 21, and 28 (*) as well. (C) Markedly low rcSO2 was observed in 2 individual cases of grade 4 IVH (gray and black squares). The dagger (†) symbol indicates the timing of IVH diagnosis on brain echography. No significant difference in rcSO2 was observed between infants without IVH (light green circles) and those with grades 1–2 IVH (dark green diamonds). (D) A similar pattern was observed for changes in the FTOE across these groups. rcSO2, regional cerebral oxygen saturation; FTOE, fraction of tissue oxygen extraction; PDA, patent ductus arteriosus; IVH, intraventricular hemorrhage; RM-ANOVA, repeated-measures analysis of variance.
In terms of primary outcome, there were 4 mortality cases (2 died of respiratory failure at postnatal age 10 and 18 days, 1 renal failure at 3 weeks, 1 hospice after severe NEC at 2 months of age), and 2 of them also developed grade 4 IVH. Three additional infants had abnormal brain fEcho and/or MRI results (2 ventriculomegaly, 1 PVL and 1 CBH). Two infants had EEG-confirmed seizure, and both of them also had abnormal brain images (grade 4 IVH and PVL, respectively). After all, there were 7 patients (23.3%) in the bundle group who had poor neurologic outcomes. We found cases with composite poor outcomes were smaller (BW 585 [533–640] g vs. 890 [755–1,120] g, P<0.001), more likely to be SGA, and had higher percentage in using high frequency oscillator (HFO) and inhaled nitric oxide. They also had lower rcSO2 during initial 72-hour (73.9% [66.6%–77.1%] vs. 79.2% [75.9%–83.1%], P=0.019) and lower time-averaged rcSO2 (51.1% [50.0%–65.2%] vs. 71.8% [67.1%–73.1%], P=0.002) (Fig. 4A and B). In the multivariate regression model including SGA, use of HFO, and time-averaged rcSO2, lower time-averaged rcSO2 remained an independent risk factor for composite poor outcomes.
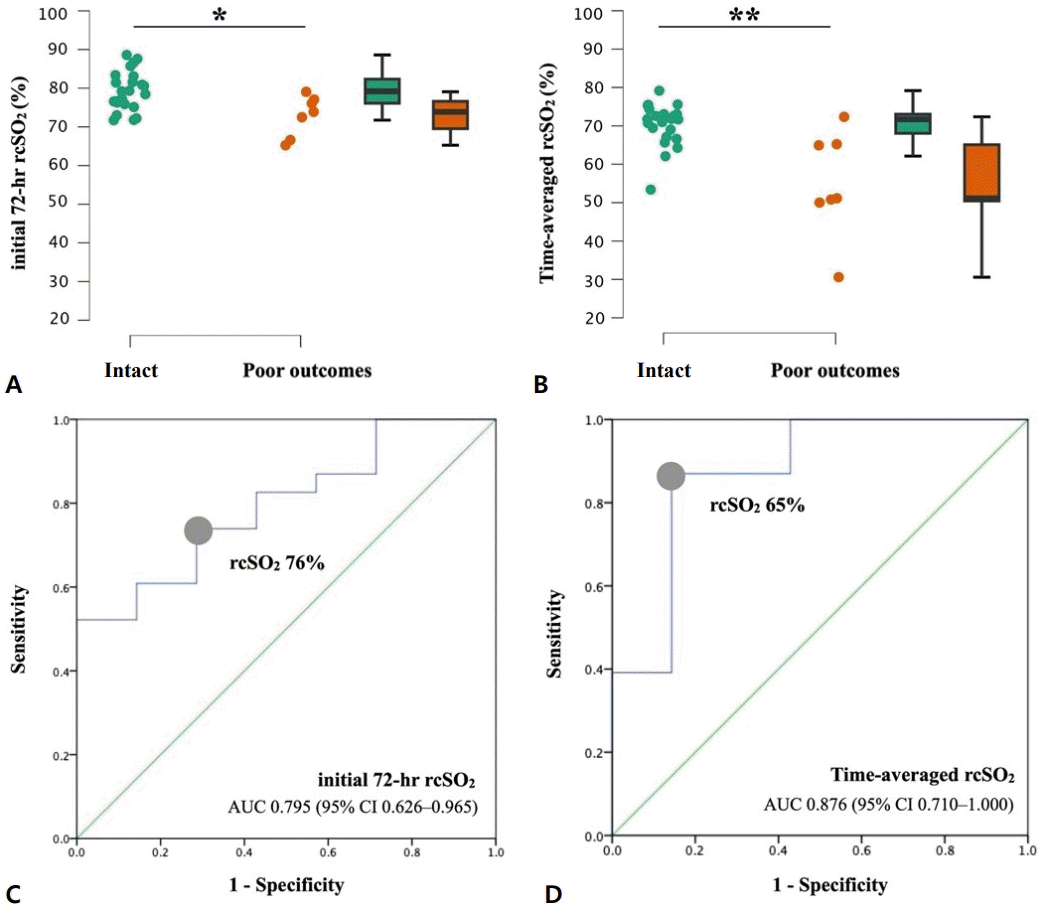
Initial 72-hour and time-averaged rcSO2 of intact survival versus poor outcome groups. Infants with poor outcomes had significantly lower initial 72-hour rcSO2 (*P=0.019) (A) and timeaveraged rcSO2 (**P=0.002) (B) values. ROC curve analysis of initial 72-hour rcSO2 (C) and timeaveraged rcSO2 (D) as predictors of composite poor outcomes in extremely preterm infants. rcSO2, regional cerebral oxygen saturation; ROC, receiver operating characteristic; AUC, area under the ROC curve; CI, confidence interval.
A post hoc analysis was performed to determine the predictive value of initial 72-hour and time-averaged rcSO2 for composite poor outcomes. The ROC for initial 72-hour rcSO2 showed an area under the ROC curve (AUC) of 0.795 (95% confidence interval [CI], 0.626–0.965; P=0.020) and cutoff rcSO2 76% (sensitivity, 0.739; specificity, 0.714), while the ROC for time-averaged rcSO2 showed a more significant AUC 0.876 (95% CI 0.710–1.000), P=0.003) and cut-off rcSO2 65% (sensitivity, 0.857; specificity, 0.870) (Fig. 4C and D).
To demonstrate the physiologic evolution of hemodynamic parameters from early life to later postnatal stages, Table 2 summarized the trajectories of rcSO2, SaO2, FTOE, and CO in neurologically intact infants. We found that mean rcSO2 within the initial 72 hours was higher, while FTOE and CO were lower, compared to the subsequent time points (RM-ANOVA, P<0.001).
Discussion
In this pilot study, we demonstrated that implementing a neurocritical care bundle comprised of multidisciplinary monitoring and stepwise management algorithm for prolonged postnatal critical periods is applicable in the NICU. This is also the first study to monitor rcSO2, FTOE, and CO in combination at specific intervals from birth to discharge or TEA, which enlightened our understanding of the interplays of vital hemodynamic elements in these preterm infants. Notably, we recognized lower rcSO2 a risk factor for in-hospital mortality and/or poor neurologic events in extremely preterm infants.
For a care bundle that relies on multiple monitoring devices, feasibility in routine clinical settings and the capacity of personnel to interpret and manage abnormal values are crucial. In our study, introductory training on NIRS and EC was provided to primary caregivers, including residents and nurses, to familiarize them with the devices. Bedside instruction cards were also available to guide device setup and the stepwise management algorithm. As a result, the care team was able to consistently adhere to the protocol and deliver a prompt first-line response to cerebral saturation disturbances.
We found that neither the first 72-hour nor time-averaged rcSO2 correlated with GA or BW. This finding suggested that a consistent standard could be uniformly applied for monitoring rcSO2 in preterm infants of varying degrees of maturation or diverse demographics. However, postnatal age should be considered as a factor influencing physiologic changes in rcSO2. We observed that rcSO2 during the initial 72 hours was higher than during the subsequent time intervals, despite consistent SaO2 throughout the study period. These findings suggested the presence of distinct hemodynamic setpoints during the fetal-neonatal transitional period, and raise the possibility that cerebral metabolism is relatively suppressed during the short-term perinatal period, which could account for the elevated rcSO2 and reduced FTOE. Similar finding has been reported that cerebral oxygen metabolic rates were relatively low within the first week in preterm infants [21].
This study offered valuable insights in terms of cerebral saturation for infants with hsPDA and IVH. In agreement with previous studies, infants with hsPDA, especially those required PDA ligation, were at risk for cerebral hypoxia and increased FTOE, and the negative impacts persisted in their neonatal period even after treatment [22,23]. This reminded clinicians that surveillance for neurodevelopmental abnormality is warranted for infants at risk even after PDA closure. This study confirmed that infants with severe IVH would encounter cerebral hypoxia (low rcSO2) and hypoperfusion (high FTOE). However, due to scant severe IVH cases, we won’t be able to conclude how bad was the cerebral hemodynamic disturbance or when the severe IVH developed.
In SafeBoosC-III trial, Hansen et al. [18] applied NIRS to monitor and guide treatment within the first 72 hours for preterm infants <28 weeks’ gestation. However, there was no beneficial effects in decrease of mortality or severe brain injury at 36 weeks’ postmenstrual age to the control group. Our study had several key distinctions from the SafeBoosC-III trial. The most notable difference was the duration of NIRS monitoring. We designed a monitoring program extending from birth to TEA or discharge, since the immediate postnatal days, while critical, but are not the only period that cerebral hypoxic-ischemic damage can occur. Extending the monitoring period also demonstrated that a 72-hour window may be insufficient, as the overall time-averaged rcSO2 showed a higher ROC and greater predictive value for neurologic outcomes. Another advantage of our study was simultaneous application of multidisciplinary monitoring. This approach allowed for a comprehensive evaluation of rcSO2 in relation to cardiac function and CBF, which provided more meticulous monitoring than routine care, helped clinicians gather information and recognize subtle hemodynamics changes. The stepwise management algorithm was an additional key strength that aided practitioners in managing abnormal rcSO2 in a reasonable and practical manner. Although there was a management protocol in SafeBoosC-III trial, concerns remained regarding its implementation and adherence across multiple centers. Recently, recommendations have emerged regarding the future directions of NIRS research, aligning closely with our perspective [24].
There were limitations of the current study. Although with sufficient statistical power, this pilot study only had a small sample size to generalize the results. We plan to conduct a randomized controlled trial to seek for stronger evidence to support the benefit of integrated neurocritical care bundle. The current study focused on neurologic outcome at TEA or discharge, but we recognized that longer follow-up duration is necessary to evaluate long-term outcome. Our post-hoc analysis suggested that a time-averaged rcSO2 threshold of 65%, may be beneficial. Future trials incorporating a higher target rcSO2 may further elucidate these potential benefits.
In conclusion, the use of multidiscipline hemodynamic monitors and a structured management algorithm to safeguard rcSO2 is clinically feasible and could aid in achieving better outcome of the patients.
Supplementary materials
Supplementary Figs. 1-2 are available at https://doi.org/10.3345/cep.2025.02117.
Illustration of time-averaged rcSO2 calculation. Time-averaged rcSO2 values were calculated using the mean rcSO2 at specific time points (Ⓐ: <24 hours; Ⓑ: 24–48 hours; Ⓒ: 48–72 hours; Ⓓ: 7 days; Ⓔ: 14 days; Ⓕ: 21 days; Ⓖ: 28 days; Ⓗ: 56 days; Ⓘ: 84 days; Ⓙ: 112 days [if still admitted]) multiplied by the corresponding duration (days). The calculation followed the trapezoidal rule and was divided by the last rcSO2 measurement (at 84 or 112 days) to represent the overall mean rcSO2 during the study period. The time-averaged FTOE was calculated using the same method. FTOE, fraction of tissue oxygen extraction; rcSO2, regional cerebral oxygen saturation.
Flow diagram of enrollment protocol. A total of 84 extremely preterm infants were admitted to our hospital during the study period. Of them, eight were excluded because of perinatal asphyxia or undergoing chest compressions before enrollment. Among the remaining eligible infants, 46 could not participate due to device issues or parental refusal. Consequently, 30 extremely preterm infants were included in the neurocritical care bundle group. GA, gestational age; BW, birth weight; DR, delivery room.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This project was supported by the National Science and Technology Council (Grant ID number 111-2314-B-182A-100-) and Chang Gung Memorial Hospital (Grant ID number BMRPK60). The funder had no role in the design, data collection, data analysis, and reporting of this study.
Acknowledgments
The authors would like to acknowledge for statistical and data analysis assistance by the Center for Big Data Analytics and Statistics, Chang Gung Memorial Hospital, Linkou.
Author Contribution
Conceptualization: KHH, WHW, SYL, CCC, MCC, RL; Data curation: KHH, WHW, SYL, MYL, IHW, RL; Formal analysis: KHH, WHW, CCC, MYL, IHW, MCC, RL; Funding acquisition: KHH; Methodology: KHH, WHW, CCC, SMC, RL; Project administration: WHW, SYL, MYL, IHW, SMC; Visualization: SMC; Writing - original draft: KHH; Writing - review & editing: KHH, MCC, RL