Perinatal risk factors for hemodynamically significant patent ductus arteriosus in very low birth weight infants
Article information
Abstract
Background
Multiple perinatal factors influence hemodynamically significant patent ductus arteriosus (HS PDA) in preterm infants.
Purpose
This study aimed to identify the risk factors associated with HS PDA in very low birth weight infants (VLBWIs) and determine the predictors of surgical ligation.
Methods
This retrospective study included VLBWIs born at 23–32 weeks’ gestation whose HS PDA properties could be identified using echocardiography. The infants were stratified into 2 groups based on gestational age (23–27 and 28–32 weeks).
Results
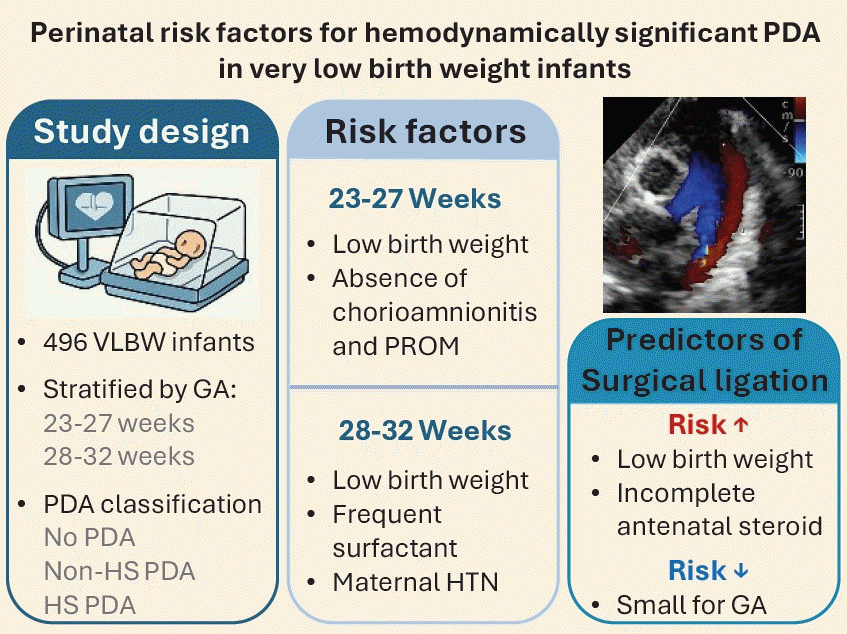
Among the 496 included VLBWIs, 171 had no PDA, 90 had non-HS PDA, and 235 had HS PDA. In infants born at 23–27 weeks’ gestation, risk factors for HS PDA included low birth weight, the absence of histological chorioamnionitis, and premature rupture of membranes. For VLBWIs born at 28–32 weeks’ gestation, HS PDA was associated with lower birth weight, frequent surfactant treatment, and maternal hypertension. Within the HS PDA group, infants with a lower birth weight or who received incomplete antenatal steroid administration had an increased likelihood of requiring surgical ligation, whereas those with a small-for-gestational-age status had a decreased need for surgical ligation.
Conclusion
Recognizing these risk factors can aid the development of targeted treatment strategies for HS PDA in VLBWIs, enabling early ligation and potentially reducing the need for surgical management.
Key message
Question: What perinatal factors influence patent ductus arteriosus (PDA) in and improve outcomes of preterm infants?
Finding: Lower birth weight, frequent surfactant treatment, and maternal hypertension increased hemodynamically significant PDA risk in very low birth weight infants, whereas chorioamnionitis and premature rupture of membranes protected against it. A lower birth weight and incomplete antenatal steroid use increased the risk of surgical ligation, whereas small for gestational age reduced it.
Meaning: Recognizing these factors may enable early, targeted intervention and reduce the need for surgical management.
Graphical abstract. PDA, patent ductus arteriosus; HS PDA, hemodynamically significant PDA; VLBW, very low birth weight; GA, gestational age; PROM, premature rupture of membranes.
Introduction
The incidence of patent ductus arteriosus (PDA) in extremely premature infants ranges from 40%–70% in infants born at 26–27 weeks’ gestation and increases to approximately 80% in those born at 24–25 weeks’ gestation [1,2]. In Korea, the prevalence of PDA is 81 per 10,000 live births, with an incidence of approximately 45.2% in very low birth weight infants (VLBWIs) [3]. PDA is a major risk factor for morbidity and mortality in VLBWIs, with a 25%–30% higher mortality rate when hemodynamically significant PDA (HS PDA) is involved than when it is not [4]. Persistent ductal patency can lead to pulmonary overcirculation, systemic hypoperfusion, and subsequent complications such as bronchopulmonary dysplasia (BPD), necrotizing enterocolitis, intraventricular hemorrhage (IVH), and adverse neurodevelopmental outcomes [4,5]. Thus, early prediction and timely intervention for HS PDA are critical.
The prognosis of HS PDA in VLBWIs is influenced by multiple factors, including gestational age (GA), birth weight, chronological age, genetic factors, and the timing of treatment [5]. However, despite its clinical importance, standardized treatment guidelines for PDA in VLBWIs remain unclear, and management strategies vary widely across institutions and countries [5]. To provide appropriate diagnosis and individualized treatment, it is essential to identify the fetal and perinatal factors that influence the delayed closure of the preterm PDA. Several perinatal risk factors for preterm PDA have been identified, including low GA and birth weight, as well as maternal chorioamnionitis, antenatal steroid exposure, mode of delivery, sex, neonatal respiratory status, and surfactant treatment [6-8]. Nevertheless, findings across studies have been inconsistent, largely because of heterogeneity in study populations, definitions of HS PDA, and treatment approaches. In particular, while GA is the strongest predictor of PDA, the associations between perinatal risk factors and PDA have varied across different GA groups, emphasizing the importance of further research focusing on GA-specific risk factors [8-11].
Therefore, the aim of the present study was to evaluate the perinatal risk factors associated with HS PDA in VLBWIs, with a particular focus on subgroup analyses based on GA. Furthermore, we analyzed the clinical outcomes and risk factors related to surgical ligation compared with those related to conservative or medical treatment.
Methods
1. Patient population
We retrospectively reviewed the medical records of 496 VLBWIs admitted to Keimyung University Dongsan Hospital, Daegu, Korea between 2014 and 2020 (Fig. 1). Exclusion criteria included inability to assess PDA properties using 2-dimensional (2D) echocardiography, unknown PDA treatment status, birth weight <500 g, complex congenital heart disease or other major anomalies, receipt of prophylactic treatment, and death before 3 postnatal days. Additionally, we excluded patients with persistent pulmonary hypertension of the newborn who were treated with inhaled nitric oxide or those with early-onset sepsis because of the potential impact of these conditions on hemodynamic status. Patients were categorized into 2 groups based on GA: 23–27 weeks and 28–32 weeks. Data were analyzed for the overall cohort and separately within each GA subgroup.
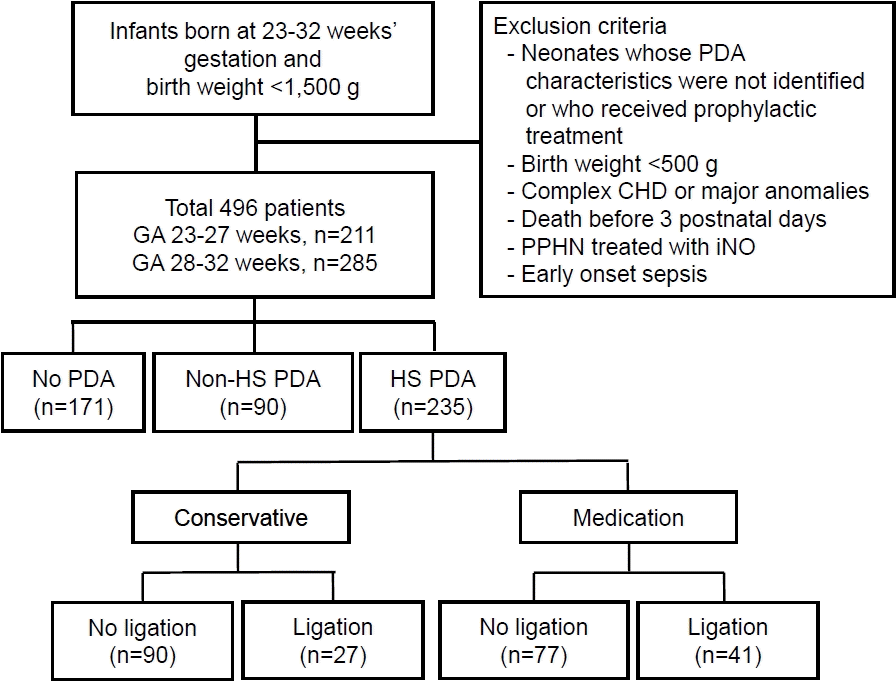
Distribution of study population. CHD, congenital heart disease; GA, gestational age; PDA, patent ductus arteriosus; HS PDA, hemodynamically significant PDA; iNO, inhaled nitric oxide; PPHN, persistent pulmonary hypertension of the newborn.
This study is a retrospective and observational study and does not contain any studies with human participants or animals. This study was approved by the Institutional Review Board (IRB) of the Keimyung University Dongsan Medical Center (IRB number: DSMC 2021-10-018).
2. Risk factor variables
We investigated neonatal variables including GA, sex, birth weight, birth height, and small for GA (SGA), as well as maternal variables including maternal age, diabetes mellitus (DM), hypertension, oligohydramnios, histological chorioamnionitis, premature rupture of membranes (PROM), and antenatal steroid use. SGA was defined as a birth weight below the 10th percentile for GA and sex [12]. Maternal hypertension included gestational hypertension, preeclampsia, eclampsia, and chronic hypertension. Histological chorioamnionitis was defined as chorioamnionitis confirmed using placental biopsy, which refers to an intrauterine inflammation occurring between the maternal tissues and the fetal membranes or within the feta adnexa [13]. PROM was defined as the rupture of amniotic membranes ≥18 hours before delivery. A complete course of antenatal steroids was defined as the administration of 2 doses of betamethasone given 24 hours apart or 4 doses of dexamethasone given 12 hours apart. Courses that did not meet these criteria were classified as incomplete.
Perinatal characteristics that were investigated included initial body temperature, pH, and base deficit after birth, Apgar scores at 1 and 5 minutes, presence of positive pressure ventilation (PPV) in the delivery room, respiratory distress syndrome (RDS), and number of surfactant replacement therapy (SRT) administrations. Postnatal outcomes, including severe IVH (grades 3–4), severe BPD, duration of invasive ventilator care, and mortality rate, were also investigated. Severe IVH was classified according to the Papile classification [14], and severe BPD was defined as requiring an FiO2 ≥ 30% or PPV at 36 weeks’ postmenstrual age [15].
3. Definition of HS PDA and treatment unresponsiveness
A single pediatric cardiologist performed 2D echocardiography (Acuson Sequoia C512; Siemens Medical Solutions and GE Vivid I; GE Healthcare, USA) at 3–14 days after birth for all patients. HS PDA was defined as a ductal diameter ≥1.5 mm with at least one of the following echocardiographic findings: (1) ductal left-to-right flow velocity <2.0 m/sec, (2) left atrium-to-aortic root ratio >1.5, (3) left pulmonary artery diastolic flow velocity >0.3 m/sec, and (4) mitral regurgitation. Patients were categorized into 3 groups based on echocardiographic findings: no PDA, non-HS PDA, and HS PDA.
Conservative management included blood pressure maintenance using inotropic agents or hydrocortisone, and fluid restriction. Mechanical ventilator support was initiated in cases of recurrent apnea or hypercapnia during nasal respiratory support, suggesting possible acute exacerbation of pulmonary edema or heart failure due to HS PDA. In VLBWIs with PDA who showed persistent clinical signs of HS PDA despite conservative management, ibuprofen was administered for 3 consecutive days, either orally or intravenously, depending on the patients' clinical conditions. The dosing regimen comprised 10 mg/kg on the first day, followed by 5 mg/kg per day on the second and third days. In cases with contraindications to medical treatment, such as thrombocytopenia (platelet count <100,000/mm3) or azotemia (serum creatinine >1.0 mg/dL), surgical ligation was considered. Surgical ligation was performed at the bedside in the neonatal intensive care unit for infants who demonstrated poor response to medical and conservative management and required continued mechanical ventilator support.
4. Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics ver. 29.0 (IBM Co., USA). Data are presented as frequencies for categorical variables and as means±standard deviations for continuous variables. The chi-square test was used for categorical variables, whereas analysis of variance with Bonferroni correction was applied for continuous variables across the 3 groups: no PDA, non-HS PDA, and HS PDA.
To identify factors associated with the development of HS PDA, we initially performed univariable logistic regression analyses within each GA subgroup. Variables with a P value <0.05 in the univariable analyses were entered into subsequent multivariable logistic regression models. In the overall cohort, the multivariable analysis was adjusted for GA. Results are presented as odds ratios (ORs) for the univariable analyses and as adjusted ORs for the multivariable analyses with 95% confidence intervals (CIs). Statistical significance was set at P<0.05.
Results
1. Patient characteristics
In total, 496 VLBWIs were enrolled: 171 in the no PDA group, 90 in the non-HS PDA group, and 235 in the HS PDA group. Among them, 265 were males and 231 were females, with a mean GA of 27.7±2.0 weeks and a mean birth weight of 1048.6±248.3 g. HS PDA was more frequent in infants born at 23–27 weeks’ gestation than in those born at 28–32 weeks’ gestation (60.4% vs. 39.3%, P<0.001).
2. Risk factors for HS PDA
The analysis results for infants born at 23–27 and 28–32 weeks’ gestation are presented in Tables 1 and 2, respectively.
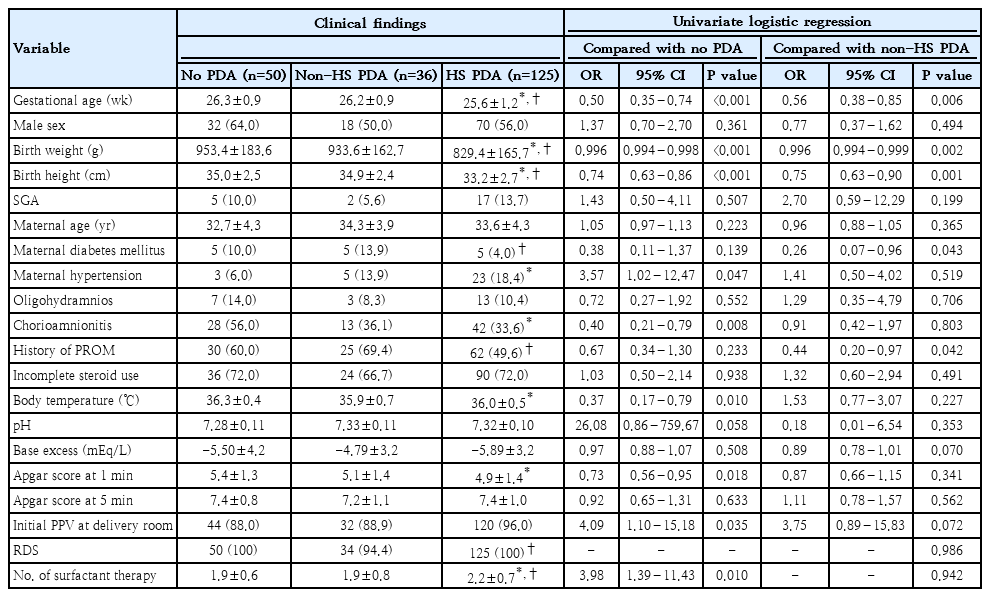
Univariate logistic regression analyses of risk factors for HS PDA in VLBWIs born at 23–27 weeks’ gestation
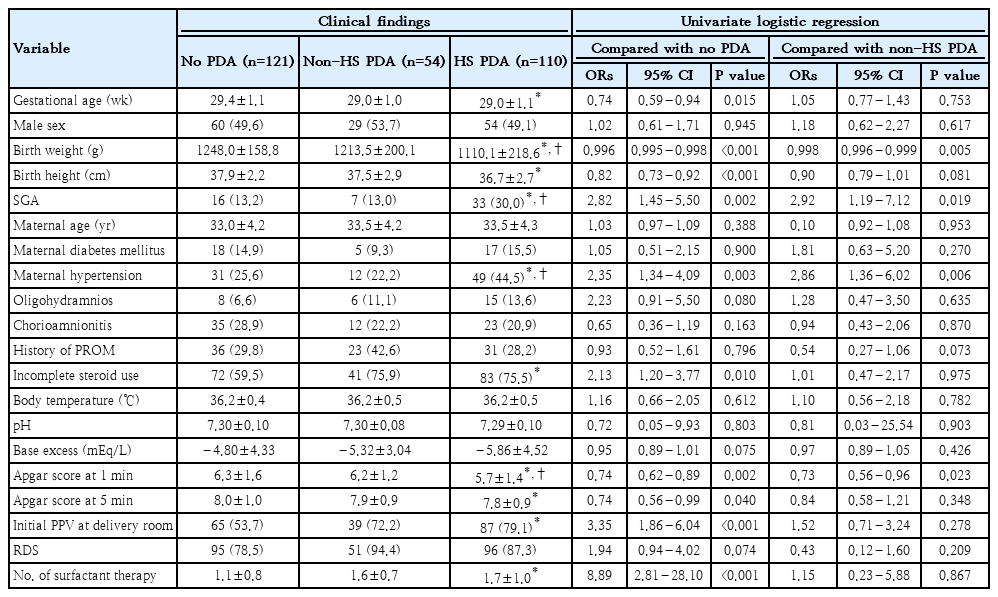
Univariate logistic regression analyses of risk factors for HS PDA in VLBWIs born at 28–32 weeks’ gestation
For infants born at 23–27 weeks’ gestation, when compared with those in the no PDA group, significant risk factors for HS PDA, included lower GA (P<0.001), birth weight (P<0.001), birth height (P<0.001), initial body temperature (P<0.05), and Apgar score at 1 minute (P<0.05); maternal hypertension (P<0.05), absence of histological chorioamnionitis (P<0.01), PPV in the delivery room (P<0.05), and more frequent SRT (P<0.05). Meanwhile, when compared with those in the non-HS PDA group, lower GA (P<0.01), birth weight (P<0.01), and birth height (P<0.05), along with absence of maternal DM (P<0.05), and absence of PROM (P<0.05) were associated with higher HS PDA risk.
For infants born at 28–32 weeks’ gestation, when compared with those in the no PDA group, significant risk factors for HS PDA included lower GA (P<0.05), birth weight (P<0.001), birth height (P<0.001), and Apgar score at 1 minute (P<0.01) and 5 minutes (P<0.05); presence of SGA (P<0.01), presence of maternal hypertension (P<0.01), incomplete antenatal steroids administration (P<0.05), presence of PPV in the delivery room (P<0.001), and more frequent SRT (P<0.001). Moreover, lower birth weight (P<0.01), presence of SGA (P<0.05), the presence of maternal hypertension (P<0.01), and lower Apgar score at 1 minute (P<0.05) were significantly associated with a higher risk of HS PDA when compared with those in the non-HS PDA group.
3. Multivariable logistic regression analysis of risk factors for HS PDA
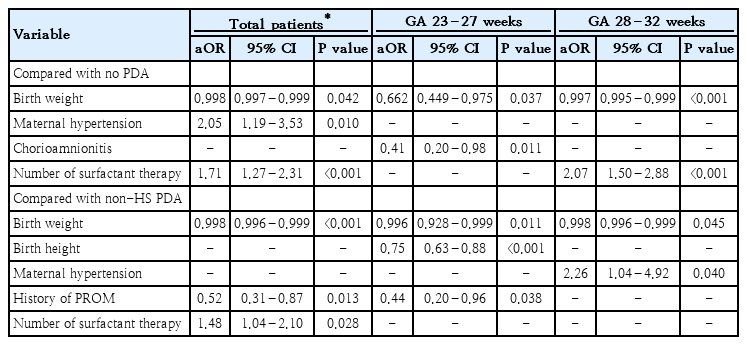
In the overall cohort, HS PDA was significantly associated with lower birth weight (P<0.05), maternal hypertension (P<0.05), and increased SRT (P<0.001) when compared with those in the no PDA group (Table 3). When compared with those in the non-HS PDA group, lower birth weight (P<0.001) and a higher frequency of SRT (P<0.05) increased the risk of HS PDA, whereas PROM (P<0.05) decreased it.
In VLBWIs born at 23–27 weeks’ gestation, HS PDA was associated with lower birth weight (P<0.05) and the absence of histological chorioamnionitis (P<0.05) when compared with those in the no PDA group. However, when compared with that in the non-HS PDA group, lower birth weight (P<0.05) and birth height (P<0.001) increased the risk of HS PDA, whereas PROM (P<0.05) decreased it.
In VLBWIs born at 28–32 weeks’ gestation, HS PDA was associated with lower birth weight (P<0.001) and a higher frequency of SRT (P<0.001) when compared with those in the no PDA group. When compared with those in the non-HS PDA group, lower birth weight (P<0.05) and the presence of maternal hypertension (P<0.05) increased the risk of HS PDA.
4. Clinical outcomes and risk factors of surgical ligation in HS PDA
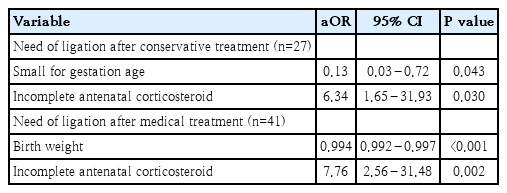
Patients who required surgical ligation after conservative treatment had a significantly higher rate of severe BPD (P<0.05) and longer duration of hospitalization (P<0.01) than those in the conservative treatment group (Supplementary Table 1). In the multivariable logistic regression analysis, incomplete antenatal steroids administration (P<0.05) increased the need for ligation after conservative treatment, whereas SGA (P<0.05) decreased it (Table 4).
Patients who required ligation after medical treatment had a significantly longer duration of invasive ventilator care (P<0.001) and hospitalization (P<0.05) than those in the medical treatment group. Multivariable logistic regression analysis showed that lower birth weight (P<0.001) and incomplete antenatal steroids (P<0.01) increased the need for ligation after medical treatment.
Discussion
In our study, we identified key risk factors for HS PDA in VLBWIs. Among infants born at 23–27 weeks’ gestation, lower birth weight and the absence of histological chorioamnionitis and PROM were associated with HS PDA. Meanwhile, among VLBWIs born at 28–32 weeks’ gestation, HS PDA was associated with lower birth weight, more frequent SRT, and maternal hypertension. Specifically, lower birth weight and incomplete antenatal steroids administration increased the need for surgical ligation, whereas SGA reduced it.
In previous studies, lower GA and birth weight were consistently identified as major risk factors for preterm PDA [6,8]. Preterm infants are less likely to achieve ductus arteriosus closure after birth because their ductal smooth muscle cells are more sensitive to prostaglandins and nitric oxide and less responsive to oxygen, and the muscular wall of the ductus is structurally immature [11]. Because GA is a major determinant of PDA and also affects the development of other perinatal factors, adjusting for GA can diminish the statistical significance of certain risk factors or even yield contradictory results [8-11]. Therefore, careful adjustment or stratification by GA is required when evaluating risk factors. In our analysis, patients were stratified into 2 GA subgroups (23–27 weeks and 28–32 weeks), and logistic regression analyses were used to adjust for GA. Our findings reaffirmed the association of lower birth weight, maternal hypertension, and increased SRT frequency with HS PDA and suggest a protective role of chorioamnionitis and PROM, particularly in the 23–27 weeks’ gestation group.
Maternal medical conditions such as hypertension and DM may influence PDA development. Hypertensive disorders during pregnancy are a major cause of SGA, and maternal hypertension and SGA have been associated with PDA in preterm infants [8,9,11,16-18]. However, these associations remain conflicting. Studies reporting a decreased incidence of PDA in infants with maternal hypertension or those who are SGA have been based solely on the classical assumption that intrauterine stress associated with intrauterine growth restriction accelerates lung maturation, thereby lowering the risk of RDS [11,17,18]. Furthermore, other studies have revealed that infants born to mothers with hypertensive disorders or SGA infants may be exposed to higher levels of corticosteroids from antenatal treatment and exhibit lower levels of nitric oxide and carbon monoxide markers, which play important roles in maintaining ductal patency [9,16]. Conversely, studies reporting an increased PDA incidence in maternal hypertension or SGA infants have proposed that placental dysfunction leads to chronic prenatal hypoxia and impaired maturation, with histological alterations in the ductus arteriosus (media and adventitia), thereby enhancing ductal patency [17-19].
The potential confounding effect of GA should also be considered. In previous studies comparing SGA and non-SGA groups, the relatively higher GA in the SGA group may account for the apparently lower incidence of PDA observed in these infants. These findings suggest that differences in GA may have a stronger influence on PDA development than on maternal hypertension or SGA itself [17,18]. Indeed, the effects of maternal hypertension and SGA vary by GA. In particular, a decreased PDA incidence has been observed in infants born at <26 weeks’ gestation, whereas an increased incidence has been reported in those born at 26–29 weeks [8,9,16,18]. Our results similarly showed that the association between maternal hypertension and PDA was more evident in infants with higher GA. SGA was significantly associated with HS PDA only in the univariable analysis, while maternal hypertension was positively associated with HS PDA in infants born at 28–32 weeks’ gestation.
Chorioamnionitis also affects PDA patency; however, previous studies have shown conflicting results regarding the relationship between histological chorioamnionitis and symptomatic PDA [10,20-22]. Some studies have shown that chorioamnionitis increases the risk of PDA, proposing mechanisms that include the effects of prematurity itself and enhanced ductal patency mediated by systemic inflammatory responses, such as bacterial products, cytokines, and vasoactive mediators [17,20,21,23]. In contrast, studies reporting a decreased PDA risk in the setting of chorioamnionitis have shown that placental prostaglandin E2 produced during chorioamnionitis may be transferred to the fetus, promoting ductus arteriosus closure after birth by stimulating smooth muscle migration and vascular remodeling via the prostaglandin E receptor 4 pathway [22,24]. Furthermore, meta-analyses adjusting for GA have shown that chorioamnionitis may play a protective role, possibly by inducing fetal lung maturation through inflammation and reducing the need for surfactant and mechanical ventilation [10]. This finding indicates that the association between chorioamnionitis and PDA may be largely confounded by prematurity itself rather than resulting from a direct inflammatory effect [17]. Supporting this, a national cohort study in Korea, in which infants were stratified by GA, identified histological chorioamnionitis as a protective factor against symptomatic PDA in infants born at 26–29 weeks’ gestation [8]. In line with these findings, our study showed that chorioamnionitis and PROM were protective against HS PDA in the 23–27 weeks’ gestation group, whereas no significant associations were observed in the 28–32 weeks’ group.
The relationship between RDS and PDA has been well established, with the severity of lung disease, oxygen requirement, and the need for mechanical ventilation consistently reported as risk factors for PDA in preterm infants [6,25-27]. In our study, although RDS and the administration of surfactant itself were not significantly associated with HS PDA, the number of surfactant doses was identified as a significant predictor of HS PDA in infants born at 28–32 weeks’ gestation. This finding is consistent with previous research reporting that surfactant use was a significant risk factor for symptomatic PDA in infants born at 26–27 weeks’ gestation, but not those born at ≤25 weeks’ gestation [8]. These results suggest that the association between surfactant therapy and PDA may vary based on GA, with repeated administration indicating more severe lung disease and a higher risk of HS PDA.
Antenatal steroid administration promotes fetal lung maturation and reduces the risk of RDS, a known contributor to preterm PDA [28]. In several studies, a decreased incidence of PDA in premature infants after antenatal steroid administration has been reported, particularly in those exposed to chorioamnionitis [8,20,23]. In animal models, betamethasone enhanced oxygen-induced ductal constriction and inhibited vasodilator signaling in the premature ductus arteriosus [28]. Notably, incomplete or inadequate steroid treatment did not produce significant effects on ductal constriction [29]. Previous studies have also shown that the effect of antenatal steroids on PDA may vary based on GA, with the ductal constriction effect of betamethasone being more pronounced in infants born at 26–27 weeks’ gestation than in those born at ≤25 weeks’ gestation [28]. Similarly, antenatal steroid use decreased the risk of symptomatic PDA in the 26–29 weeks’ GA group, but not in the 22–25 weeks’ GA group [8]. In our study, nearly all preterm infants received antenatal steroids, and the frequency of incomplete steroid administration did not significantly affect PDA incidence. However, this incomplete administration frequency was associated with an increased risk of surgical ligation following failure of conservative or medical treatment.
Several studies have identified predictors of medical treatment failure in preterm PDA, including lower GA, lower birth weight, resuscitation with intubation at delivery, lower Apgar scores at 1 minute, reduced antenatal glucocorticoid exposure, severe respiratory distress, and Caucasian race [6,8,30,31]. In contrast, infants born to mothers with hypertensive disorders have shown a better response to pharmacological treatment for PDA, and chorioamnionitis has not been shown to increase the surgical ligation risk [17,32]. SGA reportedly increases the likelihood of surgical ligation, possibly because of elevated levels of inflammation-related proteins that may impair responsiveness to pharmacological agents [33-35]. However, in another study, the surgical ligation frequency did not differ between infants who were SGA and those appropriate for GA [19]. In our study, lower birth weight and incomplete antenatal steroid administration increased the likelihood of surgical ligation after medical treatment, consistent with previous findings. However, SGA was associated with a reduced risk of requiring surgical ligation after conservative treatment. This discrepancy in findings may reflect the fact that PDA development and treatment response in infants who are SGA can vary based on their GA and involve multiple underlying mechanisms. Additionally, our analysis did not account for various clinical factors influencing treatment decisions, such as clinical hemodynamic parameters or comorbidities, which may have influenced the risk factor analysis. Overall, the decision to perform surgical ligation for preterm PDA is multifactorial and remains inconclusive, warranting further studies to clarify these associations.
This study has some limitations. It was a retrospective, single-center observational study with a relatively small sample size within each subgroup. In addition, patients were classified as having HS PDA based solely on echocardiographic criteria, without considering clinical hemodynamic parameters such as respiratory support, oxygen requirement, hypotension, oliguria, and comorbidities. This limitation may have led to discrepancies between echocardiographic findings and clinical decision-making regarding PDA treatment, potentially influencing the results of risk factor analysis.
In conclusion, this study's findings revealed lower birth weight, increased frequency of SRT, and maternal hypertension as significant risk factors for HS PDA in VLBWIs, while chorioamnionitis and PROM appeared to have a protective effect. Additionally, lower birth weight and incomplete antenatal steroid administration increased the likelihood of surgical ligation, whereas SGA reduced it. Notably, risk factors varied by GA, with other perinatal factors having GA-specific influences. These findings highlight the importance of GA-specific risk assessment and early, targeted management strategies to improve outcomes in VLBWIs.
Supplementary materials
Supplementary Table 1 is available at https://doi.org/10.3345/cep.2025.01893.
The clinical outcomes and risk factors according to management in HS PDA patients
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contribution
Conceptualization: JHJ, HJC; Data curation: JHJ; Methodology: SYS, JHP, HJC; Visualization: HJC; Writing - original draft: JHJ, JHP, CSK, HJC; Writing - review & editing: SYS, JHP, CSK, HJC