Thrombocytopenia in preterm infants born to mothers with systemic lupus erythematosus: a retrospective cohort study
Article information
Abstract
Background
Thrombocytopenia in preterm infants born to mothers with systemic lupus erythematosus (SLE) is poorly characterized, despite its potential link to adverse outcomes. Our understanding of platelet dynamics, risk factors, and clinical outcomes in this population is limited, necessitating further investigation.
Purpose
This study aimed to characterize the incidence, timing, and severity of thrombocytopenia in this population; to identify associated maternal and neonatal risk factors; and to evaluate its association with adverse outcomes.
Methods
We included 154 preterm infants born to mothers with SLE who were admitted to Shanghai Children’s Medical Center within 24 hours of birth between 2014 and 2024. Logistic regression was used to identify risk factors and outcomes associated with neonatal thrombocytopenia.
Results
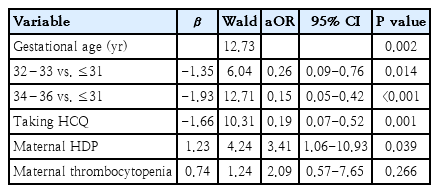
Thrombocytopenia (platelet count < 150 ×109/L) occurred in 32.5% of infants, and severe form (< 50 ×109/L) occurred in 4.6%. The condition peaked on postnatal days 4–5, and 16% of affected infants required intervention. Late preterm infants (adjusted odds ratio [aOR], 0.15; 95% confidence interval [CI], 0.05–0.42), moderate preterm infants (aOR, 0.26; 95% CI, 0.09–0.76), and maternal hydroxychloroquine use (aOR, 0.19; 95% CI, 0.07–0.52) were protective factors. In contrast, maternal hypertensive disorders of pregnancy (HDP; aOR, 3.41; 95% CI, 1.06–10.93) increased the risk. Infants with thrombocytopenia had significantly higher risks of intracranial hemorrhage (aOR, 4.27; 95% CI, 1.65–11.00) and late-onset sepsis (aOR, 11.00; 95% CI, 1.23–98.14).
Conclusion
Preterm infants exposed to maternal SLE frequently developed thrombocytopenia, but most cases were self-limited. Key risk modulators included gestational age, maternal HDP, and hydroxychloroquine use. Furthermore, thrombocytopenia was significantly associated with neonatal morbidity.
Key message
Question: What are the characteristics and clinical implications of thrombocytopenia in preterm neonates born to mothers with systemic lupus erythematosus?
Finding: Nearly one-third of preterm infants developed thrombocytopenia. Key modulators of this risk included gestational age, maternal hypertensive disorders of pregnancy, and hydroxychloroquine use. Thrombocytopenia may be associated with neonatal morbidity.
Meaning: Platelet count should be monitored during the first week of life, and infants should be assessed for potential complications.
Graphical abstract. aOR, adjusted odds ratio; CI, confidence interval; SLE, systemic lupus erythematosus.
Introduction
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease that complicates pregnancy, posing risks to both maternal and neonatal health [1,2]. Advances in maternal care have improved pregnancy outcomes, however, the offspring of mothers with SLE remain vulnerable to a range of complications, including preterm birth, intrauterine growth restriction, and multiple organ dysfunction [3-5]. Among these, thrombocytopenia is one of the most frequent hematologic manifestations observed in preterm SLE-exposed neonates [5] and warrants further investigation due to its potential impact on neonatal outcomes.
The pathogenesis of thrombocytopenia in these infants is complex and multifactorial. While the transplacental transfer of maternal autoantibodies is a characteristic feature of SLE pregnancies and may contribute to immune-mediated platelet destruction [6,7]. Other mechanisms including neonatal alloimmune thrombocytopenia, maternal immune thrombocytopenia, perinatal infections, and placental insufficiency may also be involved. This complexity poses challenges for preterm neonatal care, particularly in identifying the underlying causes of thrombocytopenia and establishing optimal monitoring and treatment strategies for affected infants [8].
This study aims to address these gaps by (1) characterizing the incidence, timing, and severity of thrombocytopenia in preterm cohort born to SLE mothers; (2) identifying maternal and neonatal risk factors; and (3) evaluating associations between thrombocytopenia and adverse outcomes. By leveraging a decade of clinical data from a high-volume tertiary center, we provide robust evidence to inform monitoring strategies and therapeutic interventions for this vulnerable population, ultimately improving neonatal care and outcomes.
Methods
1. Study design, setting, and participants
This retrospective cohort study was conducted at the Neonatology Department of Shanghai Children's Medical Center, Shanghai, China, from January 2014 to December 2024. We identified infants born to SLE mothers through our departmental medical records database during the study period. Exclusion criteria consisted of: (1) infants admitted more than 24 hours after birth; (2) full-term infants (gestational age ≥37 weeks); (3) infants with incomplete clinical data; and (4) early-onset sepsis. All enrolled participants were followed up until discharge. This study protocol was reviewed and approved by the Ethics Board of Shanghai Children’s Medical Center, School of Medicine, Shanghai Jiao Tong University (SCMCIRB-K2021016-1). Written informed consent was obtained from all participants’ legal guardian. Approval to obtain clinical data from the database was received from the office of the medical director of the hospital. All patient information was kept confidential.
2. Data collection
Trained data abstractors at Shanghai Children's Medical Center collected clinical data for all preterm participants. The data were extracted from the neonatology department's electronic medical records system for analysis, including maternal and neonatal characteristics, hematologic indices and immunologic markers.
3. Definitions
SLE was diagnosed according to the 1997 revised classification criteria established by the American College of Rheumatology [9]. We defined thrombocytopenia as a platelet count <150×109/L [10] and severe thrombocytopenia as a platelet count <50×109/L [10] with this condition first occurring within the first 3 weeks of life [2,5]. This definition was also designed to facilitate better observation of platelet count trends over time.
4. Outcomes
The primary outcome was to assess the incidence of thrombocytopenia and identify its influencing factors. The second outcomes were neonatal mortality and morbidities, including late-onset sepsis (LOS), necrotizing enterocolitis (NEC) (stage ≥2), retinopathy of prematurity (ROP) (stage ≥3), bronchopulmonary dysplasia (BPD), intracranial hemorrhage (ICH), and cardiac abnormal manifestations in thrombocytopenic infants. LOS was defined as positive blood culture or clinical sepsis [11]. NEC was determined according to Bell criteria [12]. ROP was diagnosed according to the International Classification of Retinopathy of Prematurity [13]. BPD was defined as the need for supplemental oxygen for at least the first 28 days of life, with subsequent grading determined by the level of respiratory support required at 36 weeks PMA [14]. ICH was defined as the presence of any degree of hemorrhage identified on cranial ultrasonography or magnetic resonance imaging before discharge, including subdural hemorrhage, intraventricular hemorrhage (IVH), intraparenchymal hemorrhage, cerebellar hemorrhage, and mixed hemorrhage. The grading system for IVH was defined as follows: Grade I hemorrhage was confined to the bilateral germinal matrix (subependymal region) without rupture into the ventricles. In grade II, the hemorrhage ruptured through the ependymal lining, resulting in the presence of blood within the ventricular system without ventricular dilation. Grade III hemorrhage was characterized by intraventricular blood accompanied by significant ventricular dilation. Grade IV hemorrhage involved extension of the bleeding beyond the germinal matrix into the surrounding cerebral parenchyma [15]. Cardiac conduction abnormalities included atrioventricular block of any degree, prolonged QTc, and right bundle branch block.
5. Covariates
Hypertensive disorders of pregnancy (HDP) included: chronic hypertension, gestational hypertension, preeclampsia-eclampsia, and chronic hypertension with superimposed preeclampsia [16]. Small for gestational age was defined as birth weight below the 10th percentile according to the Fenton growth curve reference standards [5]. An Apgar score >7 was defined as normal, while a score ≤7 was considered indicative of moderate or severe hypoxia [17].
6. Statistical analysis
We described continuous variables as mean±standard deviation or median (interquartile range) and categorical variables as number (%). To compare continuous variables, we used Student t test or Mann-Whitney U test. Pearson chi-square test or Fisher exact test (both 2-sided) was used for comparison of categorical variables, as appropriate. Each variable that has a P value <0.1 was entered into the multivariable logistic regression model, which was conducted to identify independent risk factors for thrombocytopenia. Binary logistic regression analysis was also performed to find the relationship between thrombocytopenia and outcomes after adjusted gestational age, reporting adjusted odds ratio (aOR) with corresponding 95% confidence interval (CI). A 2-sided P value of <0.05 was considered statistically significant. Data analysis was performed using IBM SPSS Statistics ver. 22.0 (IBM Co., USA) and R ver. 2.1.3 (R Foundation for Statistical Computing, Austria).
Results
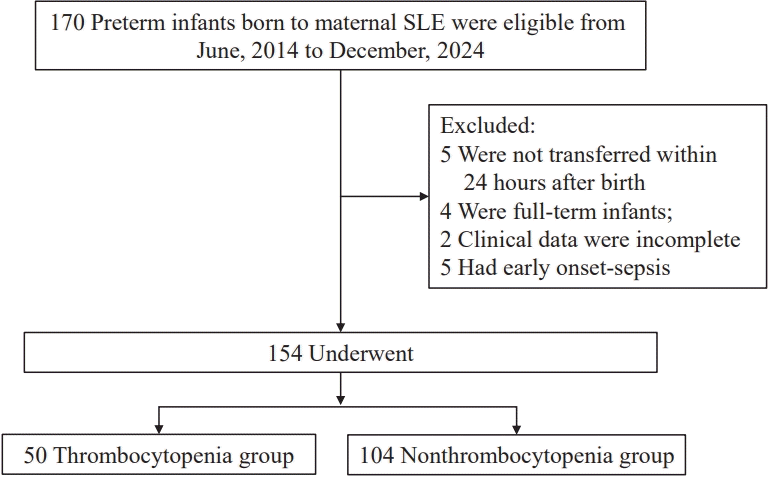
Our initial screening identified 170 preterm neonates born to 156 mothers with SLE who met the eligibility criteria. We excluded 16 infants, resulting in a final study population of 154 preterm infants, 50 of whom had thrombocytopenia (Fig. 1).
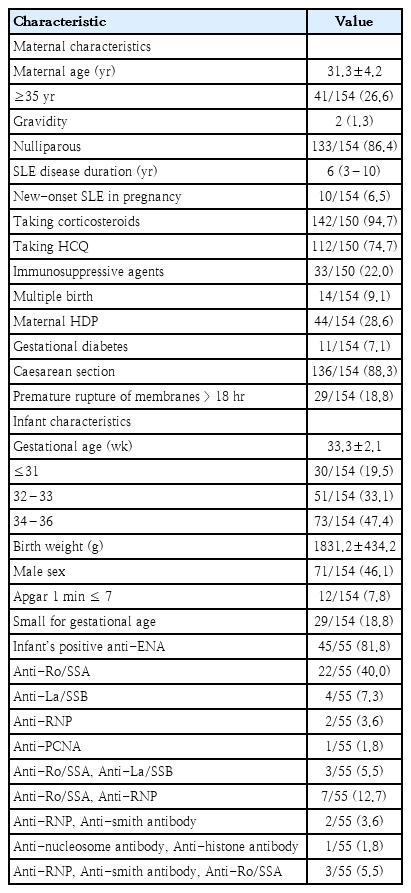
The baseline characteristics of mothers and infants are presented in Table 1. Mothers’ median SLE duration was 6 years, including 10 cases (6.5%) of new-onset SLE during pregnancy. 28.6% of women suffered from HDP, and the cesarean delivery rate was as high as 88.3%. Neonates had mean gestational age of 33.3±2.1weeks and birth weight of 1831.2±434.2 g. Additionally, antinuclear antibody testing was performed in 55 neonates in the study, 45 infants (81.8%) tested positive at birth. Anti-Sjögren syndrome A antibody (anti-Ro/SSA) and antiribonucleoprotein antibody antibodies were detected to be the most prevalent in the study. Seventeen cases were tested positive for 2 or more autoantibodies in concomitance.
1. Characteristics of platelet in preterm infants exposed to mothers with SLE
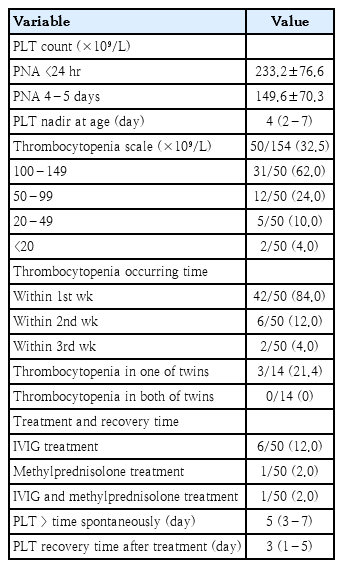
The mean neonatal platelet count within the first 24 hours of life was 233 (range, 14–438) ×109/L, with 50 infants (32.5%) developing thrombocytopenia. Severe thrombocytopenia occurred in 7 neonates, including 2 cases with profound thrombocytopenia (<20 ×109/L). While only one neonate presented with severe thrombocytopenia at birth, most cases developed within the first postpartum week. The temporal trends illustrated that the median time to platelet nadir was 4–5 days of life (Supplementary Fig. 1). Clinical interventions, including intravenous immunoglobulin (IVIG) and methylprednisolone, were required in 8 cases (6 cases of use of IVIG, 1 case of use of IVIG and methylprednisolone, and 1 case of use of methylprednisolone therapy only) due to severe thrombocytopenia or persistent platelet decline (Table 2, Supplementary Tables 1 and 2). In addition, both mean platelet volume and platelet distribution width remained within normal physiological ranges throughout the observation period, we will not discuss this any further.
2. Influencing factors for neonatal thrombocytopenia
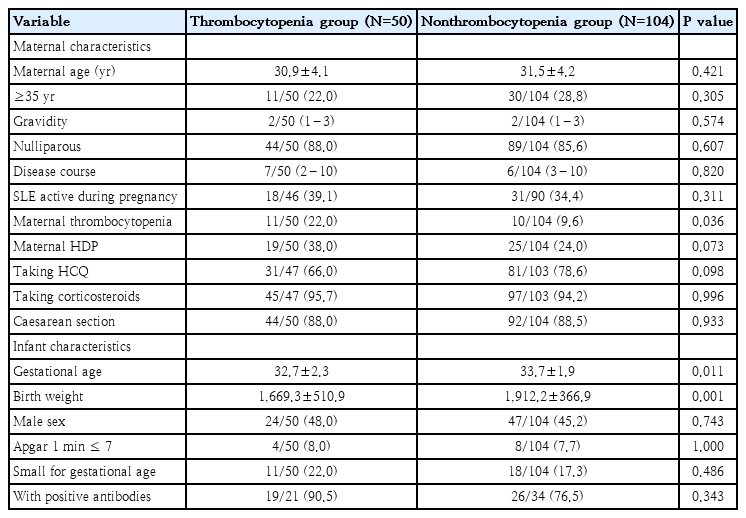
Comparative analysis revealed some significant differences between thrombocytopenic and nonthrombocytopenic infants (Table 3). The thrombocytopenic group exhibited: (1) higher rates of maternal thrombocytopenia (22.0% vs. 9.6%, P=0.036) and maternal HDP (38.0% vs. 24.0 %, P=0.073); (2) lower hydroxychloroquine usage (66.0% vs. 78.6%, P=0.098); and (3) significantly lower mean birth weight (1,669.3 vs. 1,912.2, P=0.001) and gestational age (32.7 vs. 33.7, P=0.011).
Multivariable logistic regression analysis identified gestational age and maternal hydroxychloroquine (HCQ) use (aOR, 0.19; 95% CI, 0.07–0.52) as protective factors against thrombocytopenia. Compared with the very preterm group (<32 weeks), the aOR for late (34–36 weeks) and moderate preterm birth (32–33 weeks) were 0.15 (95% CI, 0.05–0.42) and 0.26 (95% CI, 0.09–0.76), respectively. Conversely, maternal HDP (aOR, 3.41; 95% CI, 1.06–10.93) emerged as a significant risk factor (Table 4).
3. Neonatal outcomes associated with thrombocytopenia
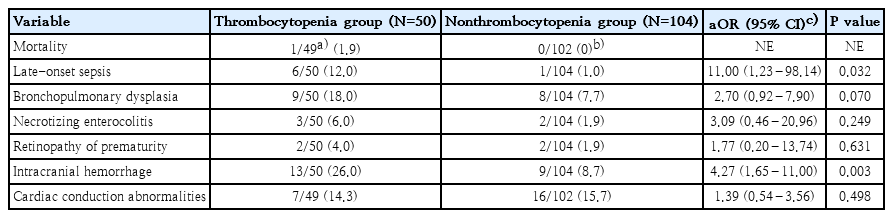
Logistic regression analysis of neonatal outcomes identified thrombocytopenia was associated with ICH (aOR, 4.27; 95% CI, 1.65–11.00) and LOS (aOR, 11.00; 95% CI, 1.23–98.14) in thrombocytopenic infants compared to nonthrombocytopenic controls after adjusted for gestational age. One case died due to severe fetal hydrops in the thrombocytopenic group (Table 5).
The occurrence time of thrombocytopenia and LOS is presented in Supplementary Table 3. In all cases, thrombocytopenia occurred more than 6 days prior to that of LOS. The location of ICH and severity of IVH are summarized in the Supplementary Table 4. Only one case of grade III IVH occurred, exclusively in the thrombocytopenic group. The incidence of IVH ≥ grade II was 30.8% (4 of 13) in neonates with thrombocytopenia versus 22.2% (2 of 9) in those without. Mixed hemorrhage was observed in 23.1% (3 of 13) of thrombocytopenic neonates and 11.1% (1 of 9) of controls.
Discussion
This study demonstrates 3 key findings in preterm infants born to mothers with SLE: (1) Thrombocytopenia occurred in 32.5% of cases, typically peaking on postnatal days 4–5, with 16% required treatments; (2) Higher gestational age and maternal HCQ use were protective, while maternal HDP increased risk; and (3) Thrombocytopenic infants were associated with ICH and LOS. These results provide the most comprehensive characterization to date of platelet dynamics in this high-risk population.
Reported rates of thrombocytopenia in neonates born to mothers with anti-Ro/SSA antibodies have varied widely, ranging from 2% to 26% [18,19]. Such discrepancies are likely attributable to differences in platelet monitoring protocols. For example, single measurements at birth (as in Cimaz et al. [19]) may underestimate the true incidence, given the typical postnatal decline in platelet counts. Additionally, thrombocytopenia is observed in 10% to 25% of infants admitted to a (high dependency) neonatal intensive care unit and 50% of those with a birth weight <1,000 g [8,20,21]. In our longitudinal premature cohort, thrombocytopenia was observed in 32.5% of infants. The nadir platelet counts occurred at 4 or 5 days of life, aligning with previous reports [2,5]. These findings highlight the importance of monitoring platelet counts during at least the first week of life in premature infants born to mothers with SLE, regardless of the underlying etiology.
In neonates with autoimmune thrombocytopenia, platelet levels should be closely monitored; but intervention is typically unnecessary unless the newborn presents with symptoms or severe thrombocytopenia [22]. In this study, only 16% of infants required treatment due to severe or concurrent thrombocytopenia. The recovery time of thrombocytopenia in our cohort was notably short, with a median of 3 days in treated infants and 5 days in those who recovered spontaneously. This is substantially shorter than reported for thrombocytopenia attributable solely to maternal SLE antibodies, where resolution typically coincides with antibody clearance over 4–6 months, or even up to 9 months in some studies [18].
Our results might be interpreted with following reasons. First, as the analysis focused on a preterm cohort, it is possible that the inherent characteristics of the preterm population had a greater impact on platelet counts than the influence of maternal SLE. Previous research has indicated that the incidence of thrombocytopenia can be as high as 50% in infants with a birth weight below 1,000 g [21]. Consistently, we identified very preterm birth as one of a significant risk factor for thrombocytopenia. Multivariate analysis confirmed that both late and moderate preterm delivery were protective, while the protective effect strengthened as gestational age increased. By contrast, autoantibodies did not emerge as a major contributor to thrombocytopenia, contrary to the hypothesized mechanism. Second, the low rate of antibody screening in our cohort may have influenced these findings. Given the limited data, the small sample size precluded definitive statistical evaluation of these as risk factors.
Moreover, we found maternal HDP significantly increased the risk of neonatal thrombocytopenia (aOR=3.41). This association aligns with prior studies linking maternal hypertension or preeclampsia to early-onset thrombocytopenia, particularly in preterm infants [23,24]. The pathophysiology likely involved placental dysfunction, which impaired fetal platelet production through chronic hypoxia and compensatory erythropoiesis—a process that may suppress megakaryocyte development [25,26]. Notably, the incidence of HDP in our SLE cohort (28.6%) was significantly higher compared with that in the general population (4%–5%) [25-27], underscoring the compounding effects of SLE-associated vascular pathology. As some studies had shown, thrombocytopenia of preterm neonates arising from placental insufficiency, perinatal asphyxia, or maternal preeclampsia or diabetes typically recovered spontaneously by day 7 or 14 [28].
Therefore, the shorter recovery period suggested that thrombocytopenia in preterm infants born to mothers with SLE may be more closely related to perinatal factors, such as preterm birth, placental insufficiency or maternal complications, rather than maternal antibody-mediated mechanisms. This observation also indicated that thrombocytopenia in the context of SLE mothers did not invariably signify neonatal lupus. Furthermore, neonatal lupus encompassed other manifestations beyond thrombocytopenia, including congenital complete atrioventricular block, characteristic cutaneous lesions, or hepatic involvement [18].
Our analysis identified maternal HCQ use as a strong protective factor against neonatal thrombocytopenia (aOR=0.19). HCQ has demonstrated safety during pregnancy. The European League Against Rheumatism and other expert guidelines recommend continuing HCQ therapy for SLE throughout gestation [29,30]. Several studies have reported that HCQ reduced pregnancy complications in women with SLE, including maternal lupus flares, neonatal lupus syndrome, preterm birth, fetal growth restriction, and potentially preeclampsia [31,32]. Consistent with these findings, our study demonstrated that maternal HCQ use was significantly associated with a reduced risk of neonatal thrombocytopenia. These results provide robust evidence supporting current guidelines that advocate for HCQ continuation during pregnancy.
This study further demonstrated that neonates with thrombocytopenia had an increased risk of LOS. Platelets play a central role not only in hemostasis, but also in antimicrobial defense and inflammation [33], and are increasingly recognized as important "first responders" to immune threats [34]. A recent study reported that platelet-to-lymphocyte ratio (cutoff 11.73) could predict neonatal sepsis, with sensitivities of 47.9% and specificities of 47.2 % [35]. However, further research is needed to improve the diagnostic accuracy of these biomarkers in clinical practice.
With respect to ICH, consistent with previous reports, thrombocytopenic infants exhibited a significantly higher risk compared to nonthrombocytopenic controls [22]. The most common hemorrhage locations were intraventricular and subdural [36], similar to our findings. Several studies have indicated that platelet count was associated with any grade of IVH and severe IVH [37]. Moreover, IVH ≥grade II was found to occur more frequently in neonates with thrombocytopenia, a relationship that appeared independent of the severity of thrombocytopenia [38]. Our study corroborated these findings.
To our knowledge, this is the first retrospective cohort study utilizing over a decade of clinical data to characterize platelet dynamics and identify thrombocytopenia risk factors in preterm infants born to mothers with SLE. Our findings carry significant clinical implications for managing these vulnerable neonates. First, close monitoring of platelet counts during the critical first postnatal week is essential, particularly for infants exposed to maternal HDP, who face the highest risk of thrombocytopenia. Additionally, our results strongly support the continued use of HCQ in SLE pregnancies, given its demonstrated protective effect against neonatal thrombocytopenia.
Several limitations of this study should be acknowledged. Firstly, the single-center design may limit the generalizability of our findings, and the lack of long-term neurodevelopmental follow-up for infants with ICH represents a critical gap in understanding the full clinical impact. Secondly, ICH was defined based on the final predischarge imaging scan. The lack of a consistent temporal relationship between its occurrence and the timing of thrombocytopenia could potentially dilute the association, indicating that thrombocytopenia might not be the exclusive risk factor for ICH. Moreover, incomplete autoantibody profiling across all infants and unmeasured confounders, such as detailed placental pathology, may have influenced our interpretations.
In conclusion, thrombocytopenia occurred more frequently in preterm infants born to mothers with SLE, with most cases developing within the first postnatal week. The majority of these cases resolved spontaneously without requiring treatment. Our analysis identified 3 key factors potentially associated with neonatal platelet counts: gestational age, mothers with HDP status, and maternal hydroxychloroquine use. Notably, neonatal thrombocytopenia may be associated with an increased risk of ICH and LOS.
Supplementary materials
Supplementary Tables 1-4 and Supplementary Fig. 1 are available at https://doi.org/10.3345/cep.2025.01942.
Severe thrombocytopenic (PLT<50×109/L) cases
Thrombocytopenic cases required intervention
Time of platelet nadir and late-onset sepsis
Location and severity of intracranial hemorrhage
Platelets count changed over days during first 3 weeks of life.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This study was supported by Shanghai Pudong Health and Family Planning Commission Joint Key Project (PW2022D-09).
Author contribution
Conceptualization: RX, XC; Data curation: RX, GZ, JB, XC, FB; Formal analysis: LM, FB; Funding acquisition: RX; Methodology: RX, XC; Visualization: FB; Writing - original draft: RX; Writing - review & editing: RX, GZ, JB, XC, FB