Hemodynamics and cerebral oxygenation in the neonatal transition: a prospective pilot study
Article information
Abstract
Background
The impact of arterial blood pressure on cerebral oxygenation during immediate postnatal transition is poorly understood.
Purpose
Here we investigated the association between arterial blood pressure (BP), cerebral tissue oxygenation index (cTOI), and cerebral fractional tissue oxygen extraction (cFTOE) during the immediate postnatal transition in preterm and full-term neonates.
Methods
This prospective observational study included preterm and term neonates who did versus did not require respiratory support. Oscillometric BP was measured at 5, 10, and 15 minutes after birth. cTOI was continuously monitored using near-infrared spectroscopy, while arterial oxygen saturation (SpO2) was recorded using pulse oximetry. cFTOE was calculated from cTOI and SpO. cTOI and cFTOE values were correlated with systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean arterial pressure (MAP) at all time points. Correlation and mixed-model analyses were then performed.
Results
A total of 102 neonates were enrolled: 51 preterm (33 with, 18 without respiratory support) and 51 term (14 with, 37 without respiratory support). In preterm neonates requiring respiratory support, cTOI was positively correlated with DABP at all time points and with MABP at 15 minutes. cFTOE was negatively correlated with MABP and DABP throughout the experiment and with SABP at 5 minutes. No significant correlations were observedamong the other groups. A mixed-model analysis showed that MABP and DABP were significantly associated with cFTOE but not cTOI independent of gestational age or respiratory support.
Conclusion
Among preterm neonates requiring respiratory support, cTOI and cFTOE were associated with arterial BP during the immediate neonatal transition. Only cFTOE showed a slight association with BP among stable preterm and full-term neonates. These findings suggest impaired cerebral autoregulation in compromised preterm infants during the immediate postnatal transition.
Key message
Question: How is arterial blood pressure related to cerebral oxygenation during the immediate postnatal transition in neonates?
Finding: Among preterm infants requiring respiratory support, cerebral oxygenation was associated with diastolic and mean arterial pressures, indicating passive pressure perfusion.
Meaning: Compromised preterm infants are vulnerable to impaired autoregulation with cerebral oxygenation fluctuations, highlighting the need for hemodynamic and cerebral monitoring plus routine monitoring of arterial oxygen saturation and heart rate.
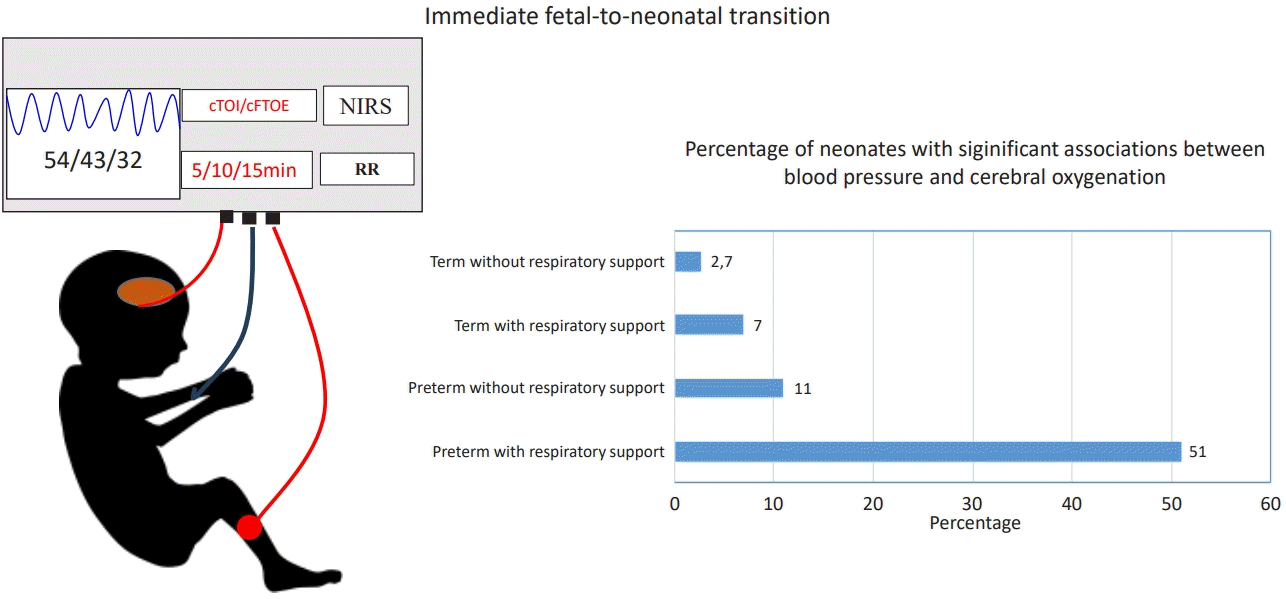
Graphical abstract. NIRS, near-infrared spectroscopy; RR, Riva-Rocci.
Introduction
Cerebral tissue oxygenation is primarily determined by 2 factors: oxygen delivery to the brain and cerebral oxygen consumption [1]. Oxygen delivery depends on the oxygen content of the blood, which is affected by hemoglobin levels and arterial oxygen saturation (SpO2), as well as cerebral blood flow (CBF). CBF is mainly influenced by cerebral perfusion pressure (CPP) and cerebral vascular resistance (CVR). In neonates, the open cranial sutures and fontanelles provide the skull with flexibility to accommodate intracranial volume fluctuations, allowing intracranial pressure to be relatively negligible [1]. Therefore, CPP is mainly influenced by systemic arterial blood pressure (BP). CVR is regulated by different factors due to cerebral autoregulation (CA).
CA is the brain’s intrinsic ability to maintain a relatively constant CBF despite changes in systemic BP, achieved through adjustments in cerebrovascular tone. This protective mechanism helps stabilize cerebral oxygen delivery and shields the vulnerable neonatal brain from hypo- or hyperperfusion.
In healthy neonates, CBF is maintained stable due to effective CA. However, in compromised neonates, CA may be impaired, making systemic arterial BP the major determinant of CBF. During the neonatal transition, systemic BP is highly dynamic, potentially resulting in fluctuations in CBF. Yet, these fluctuations can lead to hypo- and hyperperfusion, which are associated with intraventricular hemorrhage resulting in neurodevelopmental disorders [2,3]. Therefore, it is crucial to identify episodes of impaired CA.
Data suggest that near-infrared spectroscopy (NIRS) monitoring in combination with arterial BP measurements allows non-invasive monitoring of CA [4]. This approach can be particularly valuable during the immediate transition period from fetal to neonatal life, which involves major physiological changes [5]. Thus, the present study aimed to investigate the influence of arterial BP (systolic [SABP], diastolic [DABP], and mean [MABP]) on cerebral tissue oxygenation index (cTOI) and cerebral fractional tissue oxygen extraction (cFTOE) in preterm and term neonates during the immediate neonatal transition after birth. We hypothesized that compromised neonates receiving respiratory support would have impaired CA, leading to higher BP values being associated with higher cTOI and lower cFTOE values. In stable neonates, however, effective CA should keep cTOI and cFTOE independent of BP.
Methods
1. Design
The present study is a prospective observational study, which was conducted at the Division of Neonatology, Department of Pediatrics and Adolescent Medicine, Medical University of Graz, Austria, between July 2022 and November 2023. The institutional ethics committee approved the study (34-221 ex 21/22]).
2. Inclusion and exclusion criteria
Preterm and term neonates delivered by Caesarean section within the specified study period were eligible, with written informed parental consent obtained prior to birth. Preterm neonates were defined as infants born ≥28+0 and <37+0 weeks of gestational age, with a birth weight ≥1,000 g. Term neonates were defined as ≥37+0 weeks of gestation. Multiple births were eligible, however, in twin pregnancies only the first-born infant was included in the analysis to avoid correlated data within the cohort.
Exclusion criteria were decisions against full life support, major congenital malformations, and umbilical artery pH below 7.00. A decision against full life support was defined as a predelivery agreement with the parents to provide comfort care only due to an anticipated poor prognosis, with no initiation of life-sustaining interventions.
3. Data collection and monitoring
Demographics and antepartum medical histories were extracted from patient records. All neonates were promptly transferred to the resuscitation table after birth.
During the first 15 minutes after birth, SpO2 and heart rate (HR) were continuously obtained by pulse oximetry, with the sensor being placed on the right hand or wrist (IntelliVue MP 30 Monitor, Philips, The Netherlands). cTOI was continuously measured by NIRS using a cerebral oximeter monitor (T-NIRS, Hamamatsu Photonics K.K., Japan). The sensor was placed on the left frontoparietal region of the neonate’s head and secured with an elastic bandage (Peha-haft, Harmann, Germany) or a modified continuous positive airway pressure cap. All included infants who required respiratory support received noninvasive ventilation.
cFTOE was calculated using the following equation: cFTOE=(SpO2–cTOI)/SpO2.6) Arterial BP was measured noninvasively at 5,10, and 15 minutes after birth with an oscillometric BP cuff (IntelliVue MP 30 Monitor, Philips) of appropriate size (#1, #2 or #3) placed on the right calf. Cuff size was selected according to the circumference of the infant’s right calf. The right calf was chosen to avoid interference with the pulse oximetry measurements at the infant’s right hand or wrist.
All data were continuously recorded in a polygraphic system (alpha trace digital MM, BEST Medical Systems, Austria) for subsequent analyses. Missing data were minimal (2 cTOI values at 5 minutes and 3 cFTOE values at 15 minutes); analyses were performed on available cases without imputation.
4. Statistical analysis
The study was powered for the primary comparison between preterm and term neonates. Assuming a two-sided α=0.05, 80% power, equal allocation, and a moderate effect size, a total of 100 infants was required (50 per group; minimal detectable effect size d≈0.56). Demographic and clinical data are presented as mean and standard deviation for normally distributed variables or as median and interquartile range for nonnormally distributed variables.
Baseline characteristics were compared using the chi-square test or Fisher exact test for categorical variables, and Student t test or Mann-Whitney U test for continuous variables, as appropriate. For comparisons across more than 2 groups, the Kruskal-Wallis test was applied. Normality was assessed using the Kolmogorov-Smirnov and Shapiro-Wilk tests. Correlation analyses between NIRS parameters (cTOI and cFTOE) and arterial BP
SABP, DABP, and MABP at 5, 10, and 15 minutes after birth were performed separately for preterm and term neonates, with and without respiratory support. Pearson correlation was used for normally distributed data and Spearman rank correlation for skewed distributions.
To evaluate cerebrovascular autoregulation, we calculated Spearman rank correlation coefficients (ρ) between BP and cTOI as well as cFTOE at each time point (5, 10, 15 minutes) within each group (preterm±respiratory support; term±respiratory support). Impaired autoregulation was defined in the groups a priori as a positive correlation between BP and cTOI together with a negative correlation between BP and cFTOE at the same time point. Additionally, at an individual level, in each patient arterial BP, cTOI, and cFTOE were classified at every time point relative to the cohort median values. Autoregulatory failure with pressure-passive cerebral perfusion was defined when elevated cTOI and BP (both above the group median) occurred simultaneously with reduced cFTOE (below the group median) at 2 consecutive time points.
Conversely, autoregulatory failure was also assumed when reduced cTOI and BP (both below the group median) coincided with elevated cFTOE (above the group median) at 2 consecutive time points. Proportions of infants fulfilling this criterion were reported by group.
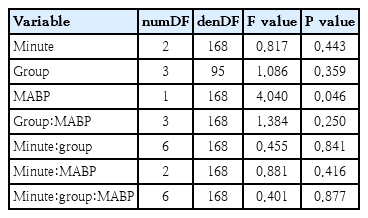
For longitudinal modeling of cTOI and cFTOE, linear mixed-effects models were fitted with patient as a random factor to account for repeated measures. Fixed effects included time after birth (5, 10, 15 minutes), group (preterm vs. term;±respiratory support), and BP (SABP, DABP, MABP). Interaction terms (group×BP; group×minute) were tested. An autoregressive (AR[1]) correlation structure was applied to model within-subject dependencies.
A P value <0.05 was considered statistically significant. Given the exploratory nature of the study, no formal correction for multiple testing was performed; interpretation focused on clinically relevant effect sizes. Analyses were conducted using IBM SPSS Statistics ver. 26.0 (IBM Co., USA).
Results
A total of 102 neonates were enrolled in this study, including 51 preterm infants (33 of 51 [64.7%] with and 18 of 51 [35.3%] without respiratory support) and 51 term infants (14 of 51 [27.5%] with and 37 of 51 [72.5%] without respiratory support) (Supplementary Fig. 1). None required cardiocirculatory support.
Among preterm neonates, Caesarean delivery was most commonly performed due to premature rupture of membranes (13 of 51, 25.5%), followed by preterm labor (9 of 51, 17.6%), preeclampsia/HELLP syndrome or maternal hypertension (8 of 51, 15.7%), and intrauterine growth restriction (7 of 51, 13.7%). Other maternal or fetal indications accounted for 14 of 51 (27.5%) of cases.
Among term neonates, the leading indications were repeated Caesarean section (22 of 51, 43.1%), followed by labor arrest (10 of 51, 19.6%), breech presentation (7 of 51, 13.7%), and macrosomia (4 of 51, 7.8%). Other indications were present in 8 of 51 (15.7%) of deliveries.
1. Demographic and clinical data
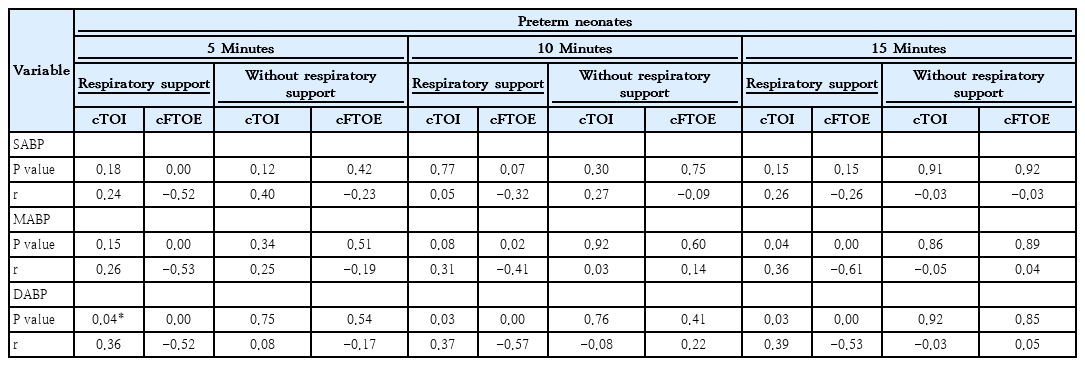
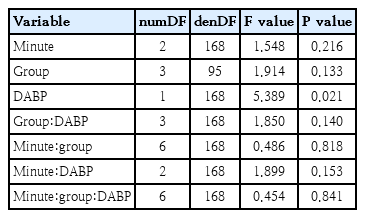
Preterm neonates with respiratory support had significantly lower gestational age, Apgar scores and arterial SpO2 compared to those without respiratory support. Birth weight, sex and umbilical artery pH did not differ (Table 1). Among term neonates, gestational age, birth weight, sex, and umbilical artery pH were similar; however, term infants with respiratory support had lower Apgar scores and SpO2 (Table 2).
Physiological and demographic characteristics of preterm neonates who did versus did not require respiratory support
2. Monitoring parameters
Preterm neonates with respiratory support showed lower cTOI and cFTOE and HR at 5 and 15 minutes, but higher SABP at 10 minutes (Table 1). Term neonates with respiratory support exhibited significantly lower HR and higher SABP at 5 minutes (Table 2).
3. Median-based classification of impaired CA
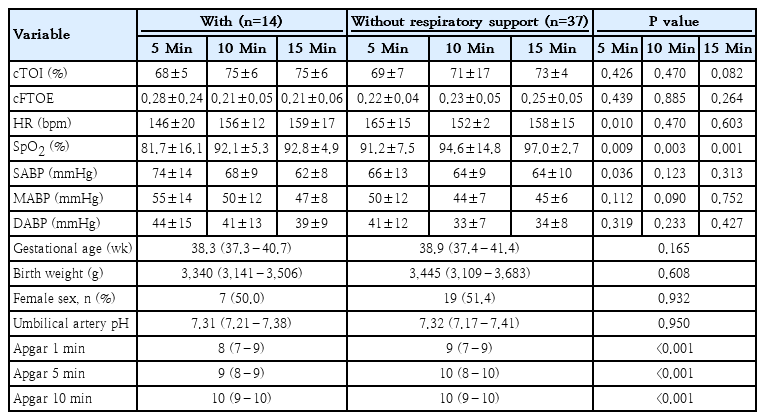
Impaired CA (≥2 consecutive time points) occurred in 17 of 33 (51%) preterm infants with respiratory support, 2 of 18 (11%) without, 1 of 14 (7%) term with, and 1 of 37 (2.7%) term without respiratory support (Supplementary Table 1). Given that cTOI reflects cerebral oxygen delivery and cFTOE reflects metabolic oxygen extraction, this pattern suggests that a substantial proportion of preterm neonates requiring respiratory support show pressure-passive cerebral perfusion, consistent with impaired autoregulation.
4. Correlation analyses and mixed-model analyses
1) Preterm neonates
(1) SABP
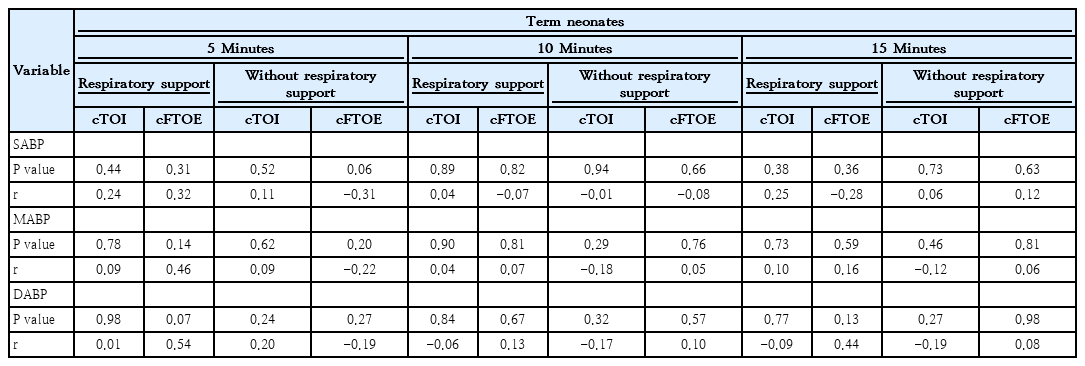
The correlation analyses revealed a negative correlation between SABP and cFTOE at 5 minutes preterm neonates with respiratory support, but not between SABP and cTOI at this time point. Afterwards, there was no significant relationship between SABP and cTOI or cFTOE in preterm neonates, irrespective of respiratory support status (Table 3). These findings were consistent with the results from the mixed-model analyses (Table 4). Thus, in preterm neonates without respiratory support no correlations suggesting intact CA, whereas in preterm neonates with respiratory support there was a significant correlation of cerebral oxygenation (cFTOE) and SABP.
(2) MABP
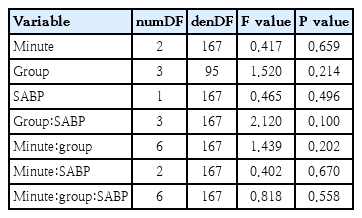
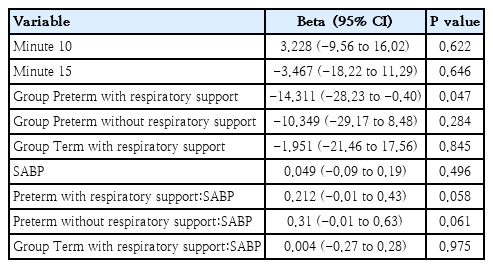
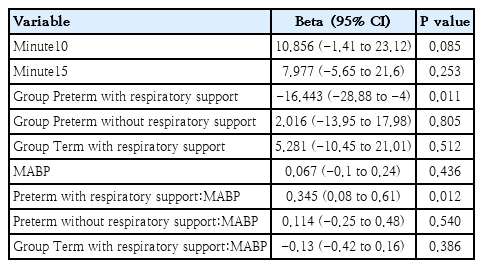
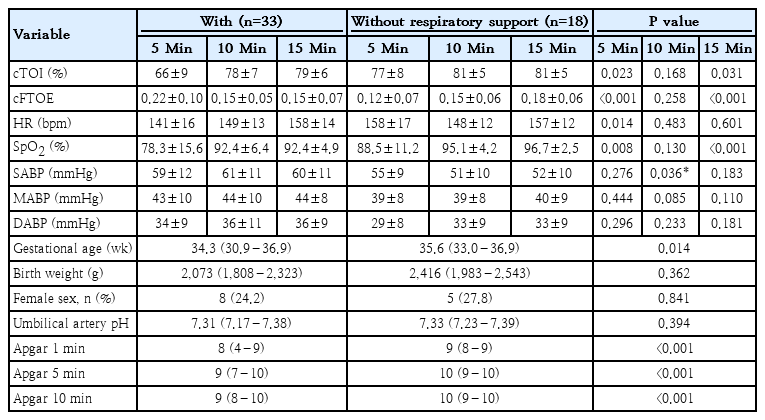
The correlation analyses showed a significant positive correlation between MABP and cTOI and a significant negative correlation between MABP and cFTOE in preterm neonates with respiratory support at 15 minutes after birth (Table 3). Mixed models confirmed this effect (Table 5). The positive correlation between MABP and cTOI indicated that rising blood pressure directly increased cerebral oxygenation—a pattern that normally should be prevented by intact autoregulation.
Mixed model for mean blood pressure
Results of the linear mixed-effects model for cerebral tissue oxygenation index (cTOI)
Conversely, the negative correlation between MABP and cFTOE showed that higher blood pressure reduced cerebral oxygen extraction, again consistent with pressure-passive flow rather than active vascular regulation.
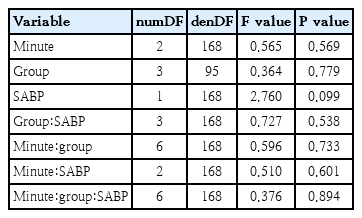
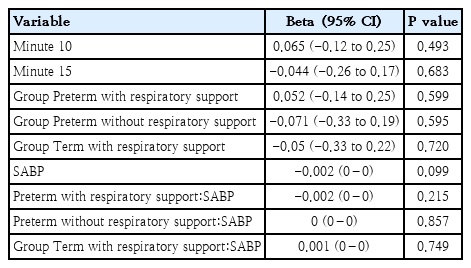
(3) DABP
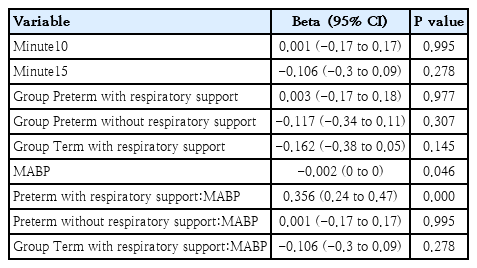
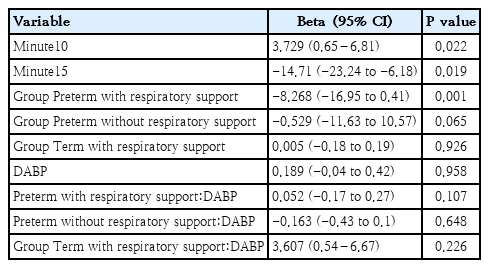
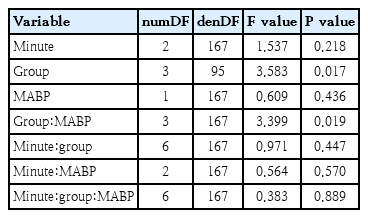
The correlation analyses showed a positive significant relationship between DABP and cTOI and a significant negative correlation between DABP and cFTOE in preterm neonates with respiratory support at 5, 10 and 15 minutes after birth (Table 3). Again, mixed models supported these findings (Table 6).
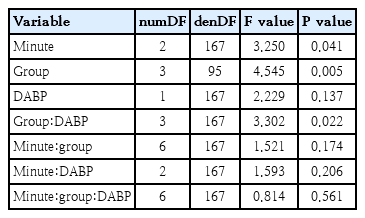
Mixed model for diastolic blood pressure
Results of the linear mixed-effects model for cerebral tissue oxygenation index (cTOI)
The positive correlation with cTOI reflected an increased cerebral oxygenation with rising pressure, while the negative correlation with cFTOE indicated reduced metabolic extraction.
No relevant associations were found in the correlation analysis in preterm infants without respiratory support. Mixed models, however, still detected a significant overall negative effect of MABP and DABP on cFTOE regardless of respiratory status or gestational age (Tables 4–6).
2) Term neonates
SABP, MABP and DABP: Correlation analyses revealed no significant relationships with cTOI or cFTOE in term infants, regardless of respiratory support (Table 3). This lack of correlations suggests that cerebral perfusion is maintained despite blood pressure fluctuations. Mixed models, however, still detected a significant overall negative effect of MABP and DABP on cFTOE regardless of respiratory status or gestational age (Tables 4–6). This mixed-model analysis demonstrated that cFTOE might be more sensitive to small BP changes than cTOI.
Discussion
This study describes for the first time differences in the associations of arterial BP measurements and cerebral oxygenation during the first 15 minutes after birth in term and preterm neonates, with and without respiratory support. In preterm neonates requiring respiratory support, cTOI and cFTOE were associated with arterial BP at different time points during immediate transition. In stable preterm and term neonates only cFTOE showed slight associations with arterial BP. The findings suggest pronounced impaired CA in compromised preterm infants during the immediate postnatal transition.
Preterm neonates needing respiratory support displayed distinct patterns compared to moderate to late preterm neonates without such support. While no statistically significant correlations were observed between SABP and cTOI at any of the 3 time points—both in correlation and mixed-model analyses—a trend approaching significance was evident. This suggests that with a larger sample size, a statistically meaningful relationship might emerge, implying that SABP could influence cerebral oxygenation in this subgroup. This aligns with other studies [7], which also reported a similar borderline significance, potentially influenced by the presence of an open ductus arteriosus—a fetal circulatory structure that can affect systemic blood flow [8].
Interestingly, SABP and cFTOE showed a significant correlation only at 5 minutes after birth in the correlation analysis, with no corresponding significance in the mixed-model analyses. This disparity underscores the need for cautious interpretation and further investigation into the early hemodynamic shifts in preterm neonates with respiratory support.
Regarding MABP, no significant correlation with cTOI was detected at 5 and 10 minutes, but a significant positive correlation emerged at 15 minutes, indicating that the relationship between MABP and cerebral oxygenation strengthens over time during the immediate transition. These findings are supported by other studies [2,7,9], which identified altered tissue oxygenation dynamics in preterm neonates requiring respiratory support. In contrast, Baik et al. [10] did not observe such a relationship, likely due to heterogeneity in their study cohort, which combined preterm neonates with and without respiratory support.
Concerning cFTOE, a significant negative correlation with MABP was observed at all time points, suggesting that higher MABP is associated with reduced cerebral oxygen extraction. This relationship intensified over time, peaking at 15 minutes. Both correlation and mixed-model analyses confirmed this finding, reinforcing existing literature that indicates impaired CA in preterm neonates requiring respiratory support [2,9-13].
Our analysis of DABP revealed significant positive correlations with cTOI and negative correlations with cFTOE at all time points, with increasing strength over time. These findings suggest that DABP progressively influences cerebral oxygenation and extraction during early neonatal transition. The consistency across analytical methods emphasizes the importance of DABP in this context and supports the hypothesis that cerebral perfusion in preterm neonates with respiratory support may be passively dependent on systemic BP, indicative of impaired autoregulation. Importantly, DABP may be the most reliable BP parameter in neonates. One possible reason is the influence of the ductus arteriosus, which affects SABP, pulse pressure, and indirectly MABP [8]. Several studies have indicated that MABP calculations, traditionally designed for adults, may not be accurate for neonates. Adjusting the formula (e.g., using 0.45–0.50 instead of one-third) may yield more reliable results [14,15].
Moreover, elevated partial pressure of arterial carbon dioxide (PaCO2) levels could contribute to the positive correlations observed with MABP and DABP, as CO2 affects cerebral vasodilation, potentially explaining differences between preterm neonates with and without respiratory support [16]. However, this does not account for the absence of similar effects in term neonates receiving respiratory support, suggesting that immaturity of cerebral vasoregulation may underlie passive perfusion patterns in preterm infants.
Additionally, increased intrathoracic pressure from respiratory support may cause venous congestion, impacting cerebral hemodynamics—particularly in preterm neonates [17,18]—but again, this effect was not evident in term neonates, reinforcing the role of developmental immaturity.
Taken together, our findings emphasize the critical importance of closely monitoring diastolic BP during immediate transition, as prolonged passive cerebral perfusion increases the risk for intraventricular hemorrhage and cerebral injury [19,20]. These results suggest that early, individualized hemodynamic assessment—ideally including continuous BP monitoring—could help clinicians detect impaired autoregulation and intervene before cerebral injury occurs. In practice, this might inform decisions about volume support, vasoactive medication, and ventilatory strategies to optimize systemic and cerebral perfusion. Future studies should aim to define safe BP thresholds to maintain autoregulation, guide intervention timing, and integrate these values into evidence-based monitoring protocols or decision-support tools for delivery rooms and neonatal intensive care units.
In term neonates, no significant correlations were found between SABP, MABP, DABP and cTOI or cFTOE at any time point, irrespective of respiratory support. Despite this, mixed-model analyses revealed significant effects of MABP and DABP on cFTOE, suggesting that cFTOE may be more sensitive to subtle BP fluctuations, even when cTOI remains stable.
This apparent discordance between correlation and mixed-model analysis reflects the complexity of CA. The stable cTOI values indicate that term neonates effectively preserve CA, ensuring consistent cerebral perfusion despite systemic changes due to birth. The cFTOE response, however, may reflect fine-tuned adjustments to compensate for the physiological changes such as reduced cardiac output due to blood pooling in the lung after aeration to maintain cerebral tissue SpO2.
Our findings align with animal studies, such as term piglet models, where CA was intact during transition [21]. Similarly, Baik et al. [10] reported that MABP did not significantly affect cerebral oxygenation but consumption (cFTOE) in term neonates at 15 minutes, reinforcing the concept of preserved autoregulation in term infants immediately after birth.
A further reason for the discordant findings between the simple correlation analyses and the mixed-model results, especially in term neonates, may lie in methodological differences. Correlation analyses are univariate and time point-specific, whereas mixed models incorporate repeated measures across all time points and adjust for within-patient dependency and group effects. Consequently, mixed models can detect overall trends with greater statistical power and are therefore considered more accurate.
A strength is the comparatively large cohort size for a neonatal study during immediate transition, which enhances the generalizability of our findings. However, the sample size still remains relatively small, representing a limitation even though NIRS is routinely used in many neonatal intensive care units. The use of mixed-model analysis adds statistical rigor and robustness. However, the observational design precludes firm causal inference. Moreover, restricting the cohort to neonates delivered by Caesarean section improves homogeneity but limits broader applicability. PaCO2 levels, intrathoracic pressures, and ductus arteriosus patency were not directly assessed, although the ductus is expected to remain patent early after birth, and ventilatory settings were comparable in infants receiving support. Subgroup analyses by respiratory support were exploratory and not powered a priori; thus, very small differences might have been missed, though such effects are unlikely to be clinically meaningful. No formal correction for multiple testing was applied given the exploratory nature of the study, but interpretation focused on clinically relevant effect sizes.
Despite these limitations, the findings highlight a critical clinical issue, warranting future interventional studies to establish optimal BP targets for ensuring CA in vulnerable neonates.
In conclusion, our study suggests that term and stable preterm neonates are widely able to maintain effective CA during immediate postnatal transition. In contrast, moderate to late preterm neonates requiring respiratory support demonstrate pronounced impaired autoregulation, making them more vulnerable to BP fluctuations. This may result in unstable cerebral perfusion and oxygenation, with potential long-term neurodevelopmental consequences. These results support routine monitoring of DABP, especially in combination with NIRS, to manage cerebral hemodynamics during this critical period.
Supplementary materials
Supplementary Table 1 and Supplementary Fig. 1 are available at https://doi.org/10.3345/cep.2025.02572.
Median-based classification of impaired cerebral autoregulation (CA)
Inclusion and exclusion of eligible neonates.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data availability
The data that support the findings of this study can be provided by the corresponding author upon reasonable request.
Author contribution
Conceptualization: DP, GP; Data curation: DP, CS, NH, GP; Formal analysis: DP, MH, GP; Funding acquisition: GP; Methodology: DP, GP; Project administration: DP, GP; Visualization: DP, GP; Writing - original draft: DP, GP; Writing - review & editing: DP, CS, NH, BS, LPM, NBS, MH, GP