Early prediction of bronchopulmonary dysplasia using new classification in high-risk preterm infants
Article information
Key message
The definition of bronchopulmonary dysplasia (BPD) has continued to evolve. Recently, newer definitions based on respiratory support at 36 weeks’ postmenstrual age better predict long-term outcomes but diagnose BPD relatively late. To address this limitation, the New Japanese Classification uses early postnatal factors, including small for gestational age and bubbly or cystic chest radiographic findings, to predict severe BPD and enable early targeted interventions.
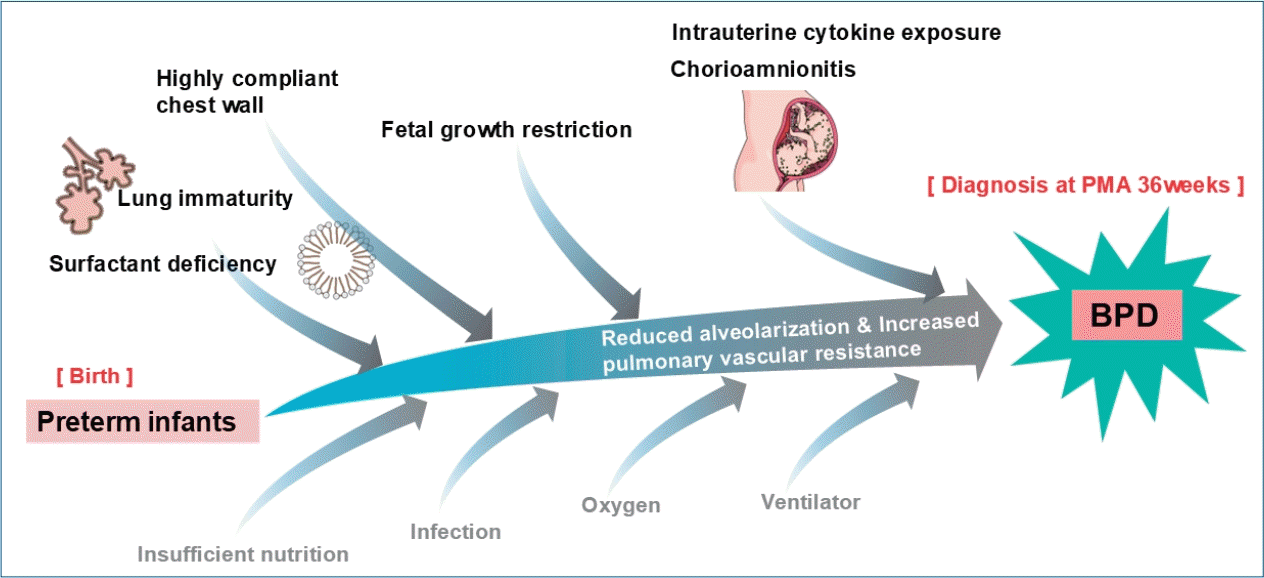
Graphical abstract. PMA, postmenstrual age; BPD, bronchopulmonary dysplasia.
With improved survival of extremely preterm infants (EPIs; gestational age <28 weeks), the number of EPIs surviving with bronchopulmonary dysplasia (BPD) has been increasing [1]. Since its first description, the definition of BPD has been revised several times over the past 50 years [2]. This definition has continued to evolve because EPIs are at the highest risk for BPD, which adversely affects early mortality and has long-lasting impacts on respiratory, neurodevelopmental, and growth outcomes. Therefore, the early and accurate diagnosis and severity grading of BPD are essential for identifying high-risk infants, enabling timely intervention, preventing BPD, and ultimately improving long-term outcomes. To unify the various definitions of BPD, the National Institute of Child Health and Human Development (NICHD) workshop proposed a definition in 2001 that has been widely used for many years [3]. However, this definition requires a precondition of oxygen or respiratory support during the first 28 days of life. Respiratory support modalities developed after 2001 are inadequately classified by this definition; for example, infants receiving high-flow nasal cannula with a fraction of inspired oxygen (FiO2) of 0.21 do not fall into any severity category of the NICHD 2001 definition. In addition, the category of severe BPD defined by the NICHD 2001 criteria is highly heterogeneous, encompassing infants who require oxygen therapy with an FiO2≥0.3 as well as those who require invasive mechanical ventilation. Moreover, mild BPD has long-term outcomes similar to those in infants without BPD, raising questions about the clinical relevance of labeling infants with BPD.
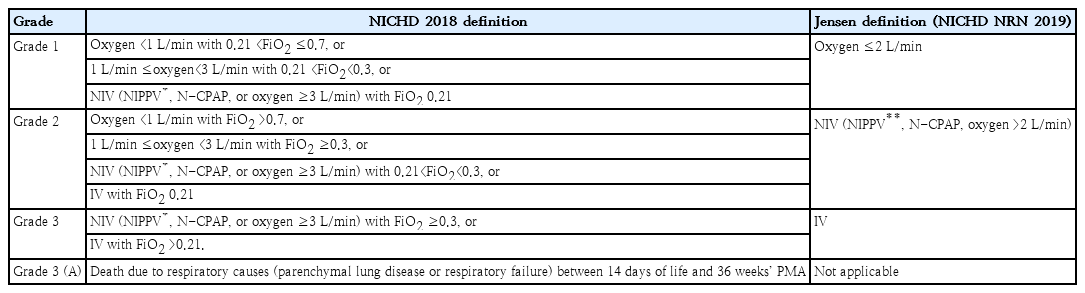
The need for a new definition of BPD is recognized, and the NICHD proposed a revised definition in 2018 [4] along with the definition suggested by Jensen et al. [5] in 2019. These newer definitions eliminated the precondition of oxygen or respiratory support during the first 28 days of life and instead defined and graded BPD severity solely based on the respiratory support required at 36 weeks’ postmenstrual age (PMA). The NICHD 2018 definition is based on oxygen concentration and respiratory support at 36 weeks’ PMA. In contrast, the definition suggested by Jensen et al. in 2019 is based on the level of respiratory support at 36 weeks’ PMA regardless of oxygen concentration (Table 1) [6]. Subsequent studies have shown that defining BPD based on respiratory support at 36 weeks’ PMA better predicts long-term outcomes [7,8].
However, these new definitions of BPD at 36 weeks’ PMA mean that the diagnosis is made relatively late after birth—somewhat delayed, especially for EPIs with a lower gestational age. This limits opportunities for the early identification of infants at high risk of BPD and the implementation of early preventive interventions. To address this limitation, a Japanese group proposed a classification system that uses early postnatal factors to predict BPD [9]. According to the New Japanese Classification, BPD is diagnosed at 28 days of life and categorized into 5 groups based on 3 factors: histologically confirmed chorioamnionitis, the presence of bubbly or cystic findings on chest radiography at day 28, and small for gestational age status. In this system, being small for gestational age and the presence of bubbly or cystic findings on chest radiography at day 28 were predictive of severe BPD, as defined by the NICHD 2001 criteria, at 36 weeks' PMA. Furthermore, severe BPD was strongly associated with bubbly/cystic findings alone on chest radiography or bubbly/cystic findings combined with chorioamnionitis.
This New Japanese Classification may help identify infants at high risk of severe BPD at an early stage and facilitate early targeted interventions. However, this study has several limitations, including its small sample size and the inclusion of infants born more than a decade ago (2006–2009). As a result, it may not reflect improvements in neonatal intensive care quality or associated advances in respiratory management and long-term neurodevelopmental outcomes. Moreover, this study did not evaluate the long-term outcomes related to these classifications. Despite these limitations, the major strength of this approach lies in its ability to predict severe BPD at an early stage using maternal characteristics, perinatal factors, or at the latest, bubbly or cystic findings on chest radiography by 28 days of life. Therefore, to facilitate the global application of the New Japanese Classification, further multicenter studies that include diverse countries and racial backgrounds are needed. These studies should evaluate the prediction of BPD based on newer BPD definitions rather than the NICHD 2001 definition and reflect contemporary neonatal care standards.
In conclusion, the key issue in the evolution of BPD definitions is not which definition should be used but which best enables the earlier identification of high-risk infants, prevention of BPD, and improvement of long-term outcomes. To achieve these goals, the definition of BPD has continued to evolve and will likely continue to do so.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.