Optimal postnatal corticosteroid regimens to prevent bronchopulmonary dysplasia with minimal adverse effects
Article information
Abstract
Postnatal corticosteroids can facilitate ventilator weaning and reduce the risk of bronchopulmonary dysplasia (BPD); therefore, they are commonly used to prevent or treat BPD in preterm infants, particularly those born extremely preterm. Despite their frequent use in high-risk infants with severe BPD, no clear guidelines have been established for the optimal timing of administration, dosage, corticosteroid type, route of delivery, and indication based on the infant’s baseline risk of BPD. Early systemic corticosteroid administration, particularly dexamethasone within the first week of life, appears to be associated with adverse neurodevelopmental outcomes and is generally not recommended. Dexamethasone and hydrocortisone exhibit distinct biological and clinical effects, yet evidence from direct comparative studies is limited. Dexamethasone may improve cerebral palsy-free survival of infants at high risk of BPD but poses potential harm in those at low risk, highlighting the need for individualized risk-based decision-making. Optimal dosing remains unclear: lower doses may reduce systemic side effects despite the uncertainty of their neurodevelopmental safety, whereas higher doses may be more effective in selected high-risk infants. Inhaled corticosteroids have inconclusive benefits compared with systemic therapy. The intratracheal administration of corticosteroids with surfactant improves distal airway delivery and reduces death or BPD rates; however, short- and long-term safety data remain limited. Overall, postnatal corticosteroids should be used cautiously and selectively in high-risk, ventilator-dependent infants with severe BPD. Future high-quality trials are needed to evaluate long-term survival free of neurodevelopmental impairments.
Key message
Postnatal corticosteroids facilitate ventilator weaning and reduce the risk of bronchopulmonary dysplasia (BPD). Despite being commonly used in high-risk infants, administration guidelines have not been established. Early dexamethasone may cause neurodevelopmental harm, and the optimal dosing remains uncertain. Inhaled corticosteroids show inconclusive benefits, whereas intratracheal administration with surfactant appears promising. Thus, corticosteroids should be used cautiously in high-risk, ventilator-dependent infants with severe BPD, and further high-quality trials are needed.
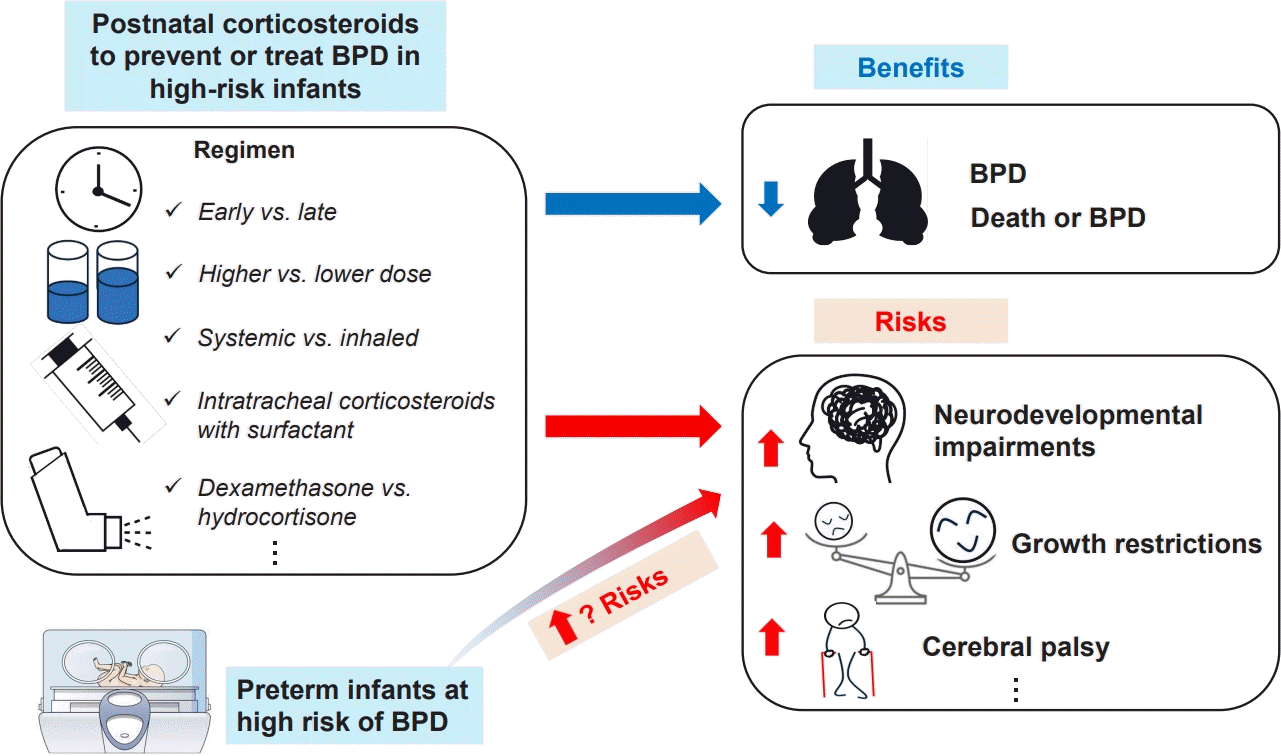
Graphical abstract. BPD, bronchopulmonary dysplasia.
Introduction
The use of postnatal corticosteroids for the treatment or prevention of bronchopulmonary dysplasia (BPD) remains one of the most controversial topics in neonatal intensive care. With the improved survival of preterm infants, the incidence of BPD, a chronic lung disease of prematurity, has increased [1]. Preterm infants requiring prolonged mechanical ventilation or supplemental oxygen represent a substantial burden on themselves, their families, and society. Mechanical ventilation can alter systemic blood flow and induce cytokine release, thereby causing injury to the lungs and brain [2]. In preterm infants with underdeveloped cerebral autoregulation, mechanical ventilation may alter cerebral blood flow, thereby increasing the risk of intraventricular hemorrhage [3]. Moreover, mechanical ventilation was reported to induce proinflammatory cytokine production and microglial activation in animal models, leading to neuronal apoptosis [4]. Therefore, postnatal corticosteroid therapy is indispensable for preventing and treating BPD as well as facilitating ventilator weaning and earlier extubation, particularly in extremely preterm infants (EPIs; gestational age <28 weeks) who are at high risk of developing BPD [5,6)].
Along with these findings, as survival rates in EPI have improved, the number of BPD survivors has also increased. Consequently, postnatal corticosteroid therapy to prevent or treat BPD in EPIs has increased substantially, including more frequent use, longer treatment durations, higher doses, and earlier postnatal administration. Consequently, concerns have also increased regarding potential corticosteroid-related complications such as neurodevelopmental impairment (NDI) and cerebral palsy, particularly among EPIs who are already at high risk for these adverse outcomes [7-9]. Therefore, this review aimed to evaluate the safety and short- and long-term complications associated with postnatal corticosteroid therapy according to administration timing, corticosteroid type, dosage, delivery route, and the infant’s baseline risk of BPD. It also sought to identify the optimal regimen for preventing BPD while minimizing adverse effects.
Timing of administration: early versus late
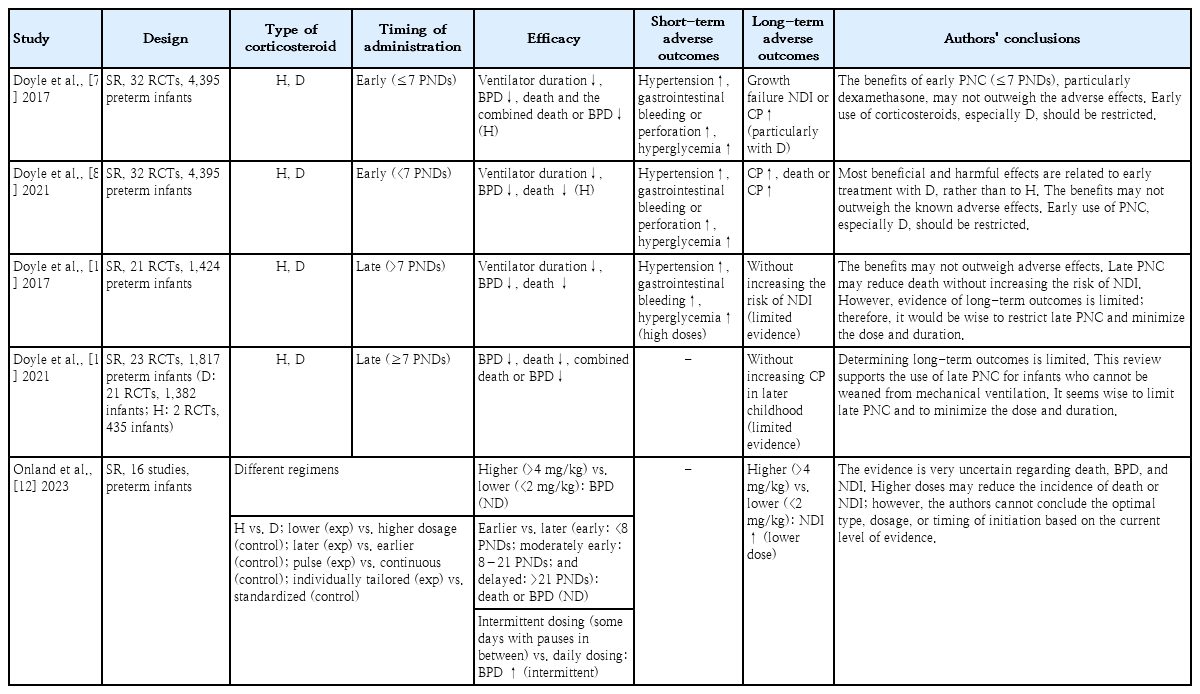
Systematic reviews have reported that the early administration of dexamethasone (within the first week of life) facilitates extubation and reduces the risk of BPD; however, it is associated with an increased risk of short-term adverse effects such as gastrointestinal bleeding, intestinal perforation, hyperglycemia, and hypertension, as well as long-term adverse effects including growth failure, NDI, and cerebral palsy (Table 1) [7,8]. In contrast, other systematic reviews have reported that the late administration of dexamethasone (after the first week of life) reduces the risk of death, BPD, and the combined outcome of death or BPD without evidence of an increased risk of long-term NDI [10,11]. These systematic reviews support the late administration of systemic corticosteroids for infants who cannot be weaned from mechanical ventilation. However, each review had limited ability to determine long-term outcomes, and none were adequately powered to detect an increased risk of NDI.
A recent systematic review reported no significant differences in efficacy or adverse outcomes between earlier and later postnatal corticosteroid administration (early, <8 postnatal days; moderately early, 8–21 postnatal days; and delayed, >21 postnatal days), resulting in somewhat inconclusive findings [12]. Moreover, no differences were noted in BPD; composite outcome of death or BPD; death at 28 postnatal days, 36 weeks of postmenstrual age, or hospital discharge; gastrointestinal perforation; necrotizing enterocolitis, and cerebral palsy among survivors; or the composite outcome of death or cerebral palsy.
Taken together, multiple studies have shown that early systemic corticosteroid administration (≤7 postnatal days) is not recommended, as the risk of long-term adverse outcomes such as NDI and cerebral palsy outweighs the potential benefits.
Hydrocortisone versus dexamethasone
Mineralocorticoid receptors are distributed in the hippocampus and limbic areas of the brain, which are critical for learning and memory, whereas glucocorticoid receptors are widely distributed throughout the brain. Glucocorticoid activity demonstrated association with apoptosis, whereas mineralocorticoid stimulation was protective against neuronal injury [13]. Dexamethasone demonstrates glucocorticoid activity only, whereas hydrocortisone possesses both mild glucocorticoid and mineralocorticoid activities. Therefore, hydrocortisone is believed to be associated with less neurotoxicity and fewer deleterious effects on neurodevelopment than dexamethasone [14]. Moreover, sustained glucocorticoid receptor stimulation, such as with dexamethasone use, can suppress the hypothalamic-pituitary-adrenal axis and result in mineralocorticoid deficiency in mineralocorticoid receptor-sensitive regions of the brain. This may result in the inhibition of neuronal growth and proliferation, increased apoptosis, and delayed myelination [13].
In a randomized clinical trial, EPIs born at 24–27 weeks of gestation were enrolled within 24 hours of birth and randomly assigned to receive either placebo or hydrocortisone (0.5 mg/kg twice daily for 7 days, followed by 0.5 mg/kg once daily for 3 days) [15]. In this trial, early administration of low-dose hydrocortisone was not associated with a statistically significant difference in neurodevelopmental outcomes at 2 years of age compared with placebo [15].
Another study showed that hydrocortisone administration 7–14 postnatal days did not improve the composite outcome of death or BPD compared with placebo [16]. A subsequent follow-up study at 2 years of corrected age found no significant difference in the composite outcome of death or NDI between the hydrocortisone (administered 7–14 postnatal days) and placebo groups [17].
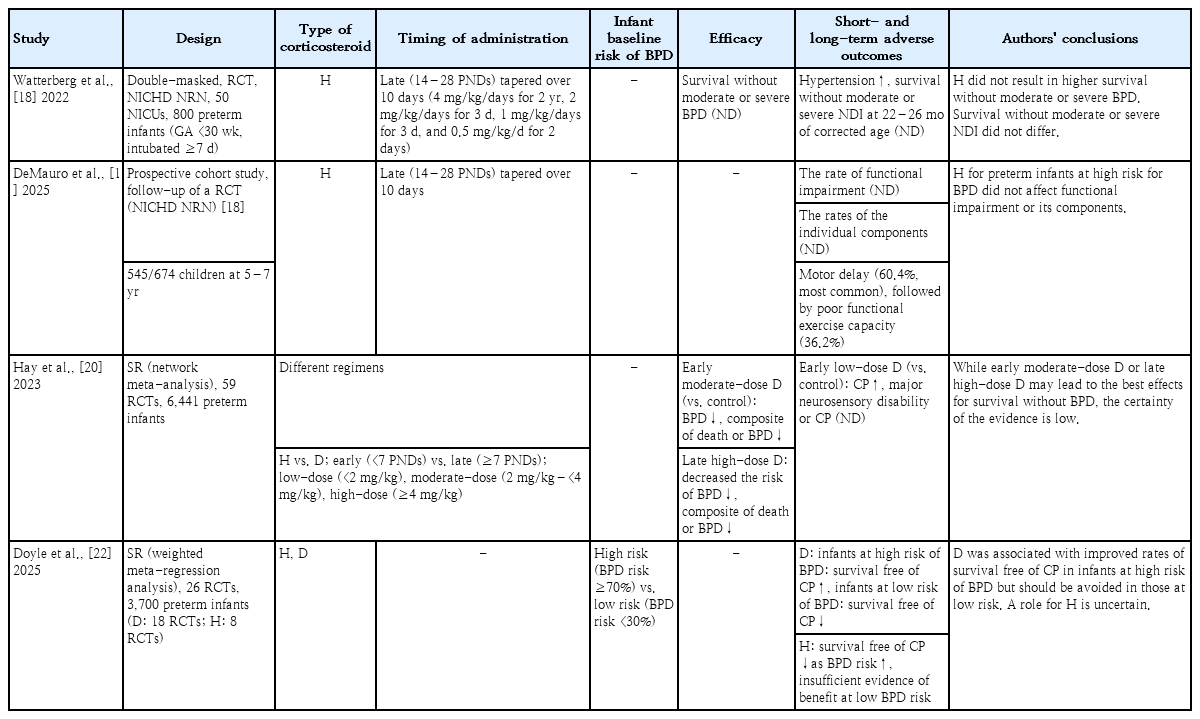
In a randomized controlled trial conducted at 19 Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network (NICHD NRN) centers, hydrocortisone was initiated 14–28 postnatal days and tapered over 10 days in infants at high risk for BPD who had been intubated for ≥7 days. No differences in survival without moderate or severe BPD or survival without moderate or severe NDI were found between the hydrocortisone and placebo groups [18]. In contrast, hypertension requiring medication was more frequent in the hydrocortisone group than in the placebo group. The follow-up of this cohort at early school age (corrected age, 5–7 years) showed no differences in functional impairment or its individual components between the hydrocortisone and placebo groups [19]. The authors concluded that hydrocortisone did not affect functional impairment, even with long-term follow-up into school age (Table 2).
In a systematic review, early hydrocortisone administration (≤7 postnatal days) had little to no effect on the composite outcome of death or cerebral palsy, whereas early dexamethasone administration appeared to increase this risk [8]. However, another systematic review revealed that early treatment (≤7 postnatal days) with moderate-dose dexamethasone (2 mg/kg ≤cumulative dose <4 mg/kg) decreased the risk of BPD as well as the composite outcome of death or BPD compared with the control intervention [20].
Dexamethasone and hydrocortisone have important biological and clinical differences; theoretically, hydrocortisone may have a less negative effect on neurodevelopmental outcomes than dexamethasone. However, few studies have directly compared the long-term outcomes of these 2 agents.
Taken together, hydrocortisone did not improve outcomes and had little to no effect on BPD or cerebral palsy in clinical studies. Dexamethasone decreased the risk of BPD; however, early (≤7 postnatal days) dexamethasone administration was associated with an increased risk of NDI.
Infant baseline risk of BPD: high versus low risk
In a previous systematic review analyzing 20 studies (N=1,721 infants), postnatal corticosteroid administration reduced the risk of death or cerebral palsy in infants with a BPD risk ≥65% but significantly increased this risk in infants with a BPD risk ≤35% [21]. The authors recently updated their systematic review; in that analysis of 26 studies (N=3,700 infants), dexamethasone, compared with placebo, improved cerebral palsy-free survival among infants at high risk of BPD, showing benefit in those with a BPD risk ≥70% but harm in those with a risk < 30% [22]. In contrast, hydrocortisone was associated with worsening cerebral palsy-free survival as BPD risk increased, with insufficient evidence of benefit at a low BPD risk. The authors concluded that dexamethasone improves cerebral palsy-free survival only in infants at high risk of BPD. In addition, in the dexamethasone-treated group, no association was noted between the treatment effect and BPD risk according to dexamethasone administration timing (early vs. late) (Table 2).
In a retrospective propensity score–matched cohort study, the risk of death or NDI associated with postnatal corticosteroid administration (75.3% received dexamethasone) decreased by 2.7% for every 10% increase in the risk of death or grade ≥2 BPD [23]. Postnatal corticosteroid administration was associated with a reduced risk of death or disability in infants at high risk of death or grade ≥2 BPD but with potential harm in those at lower risk.
However, regarding hydrocortisone, in the NICHD NRN Hydrocortisone Trial, among preterm infants at risk of death or grade ≥2 BPD, hydrocortisone administration 14–28 postnatal days did not improve the composite outcome of death or grade ≥2 BPD compared with controls. In addition, this treatment did not improve the composite outcome of death or moderate-to-severe NDI at a corrected age of 22–26 months [24].
Taken together, dexamethasone has been shown to decrease the risk of death or cerebral palsy and improve cerebral palsy-free survival among infants at high risk of BPD. However, in infants at low risk, its use may be associated with potential harm and, therefore, should be approached with caution.
Higher versus lower dose
In a systematic review of 16 studies, higher- versus lower-dose dexamethasone (high dose, >4 mg/kg; moderate dose, 2–4 mg/kg; low dose, <2 mg/kg) showed no difference in BPD development; however, infants receiving the lower dose had an increased risk of NDI. Hig0her-dose dexamethasone may reduce the risk of death or NDI [12]. Nevertheless, based on the current level of evidence, the authors could not determine the optimal dosage for BPD prevention that does not adversely affect neurodevelopment.
In a network meta-analysis, early treatment (administered before 7 days of life) with low-dose dexamethasone (cumulative dose <2 mg/kg) was associated with an increased risk of cerebral palsy. Early treatment with moderate-dose dexamethasone (2 mg/kg ≤cumulative dose <4 mg/kg) was associated with a decreased risk of BPD and the composite outcome of death or BPD compared with the control group. Late treatment (administered on or after 7 days of life) with high-dose dexamethasone (cumulative dose ≥4 mg/kg) reduced the risk of BPD as well as the composite outcome of death or BPD [20]. Therefore, in this study, early treatment with moderate-dose dexamethasone or late treatment with high-dose dexamethasone was considered the most effective strategy for achieving BPD-free survival.
Marr et al. [25] conducted a single-center study of 59 EPIs at very high risk of BPD. They reported that infants who received dexamethasone tapered slowly over 42 days (cumulative dose, 7.56 mg/kg) had shorter ventilation durations, earlier achievement of full enteral feeding, and higher handicap-free survival rates at school age (defined at 7 years as normal neurological examination, intelligence quotient >70, and without the need for special education) than those who received dexamethasone tapered rapidly over 9 days (cumulative dose, 4.04 mg/kg).
Taken together, these findings suggest that the optimal corticosteroid dosage remains uncertain. Although lower doses (<2 mg/kg) may reduce systemic adverse effects, safety from adverse effects on neurodevelopment remains unclear. In contrast, higher doses (>4 mg/kg) may be more effective at reducing the incidence of death or BPD in selected high-risk populations.
Inhaled versus systemic corticosteroids
As concerns regarding the short- and long-term adverse effects of systemic corticosteroids have increased, inhaled corticosteroids have emerged as an alternative, prompting further investigations. Because inhaled corticosteroids are administered directly into the respiratory system, they exert local therapeutic effects while causing fewer systemic side effects. A systematic review of 10 qualifying trials (N=1,644 neonates) revealed that early inhaled corticosteroids did not significantly reduce the incidence of BPD among all enrolled neonates [26]. However, early inhaled corticosteroids significantly reduced the incidence of BPD among survivors. In addition, early inhaled corticosteroid treatment was associated with a significant reduction in the composite outcome of death or BPD among all enrolled neonates and survivors.
In another systematic review, late inhaled corticosteroid therapy (initiated at ≥7 days of life) did not reduce the incidence of death or BPD [27]. In addition, the late administration of inhaled corticosteroids did not significantly affect the total duration of mechanical ventilation or oxygen therapy. Therefore, inhaled corticosteroid therapy initiated at ≥7 days of life showed no clinically meaningful ability to reduce death or BPD and are not recommended for infants at high risk of BPD. In a subsequent study, based on the currently available evidence from the systematic review, it remains uncertain whether late inhaled corticosteroids (initiated at ≥7 days of life) reduce the incidence of death or BPD among infants at high risk of BPD [28]. In a systematic review including 3 trials (N=431 infants), inhaled versus systemic corticosteroids showed no difference in efficacy or adverse effects [29]. There were also no differences in death or BPD at 36 weeks of postmenstrual age, duration of mechanical ventilation, or length of hospital stay. Moreover, the rates of adverse effects—including hyperglycemia, hypertension, necrotizing enterocolitis, and sepsis—as well as neurodevelopmental outcomes at 7 years of age were comparable between groups. Thus, to enhance the effectiveness of inhaled corticosteroids without increasing adverse effects, improved delivery systems capable of selectively targeting the alveoli are needed.
Taken together, the available evidence regarding the benefits of inhaled versus systemic corticosteroids remains inconclusive.
Intratracheal administration of corticosteroids with surfactant
The administration of inhaled corticosteroids as aerosol particles to preterm infants is technically challenging, and inhalation is ineffective because it cannot deliver corticosteroids to the distal lungs. Additionally, the direct intratracheal administration of corticosteroids alone ineffectively delivers the drug to the alveoli [30]. Therefore, intratracheal administration with a vehicle is proposed method to deliver corticosteroids to the distal lungs. Compared with drug administration alone, the use of surfactant as a vehicle was reported to improve drug distribution and spread. Accordingly, the intratracheal administration of budesonide, which has strong local anti-inflammatory effects, combined with surfactant was found to reduce BPD in high-risk preterm infants [31].
Yeh et al. [32] reported that in preterm infants with severe respiratory distress syndrome, the intratracheal administration of a mixture of budesonide (0.25 mg/kg) and surfactant (100 mg/kg) significantly reduced the incidence of death or BPD, decreased the need for surfactant readministration, and lowered levels of interleukin (IL)-1, IL-6, and IL-8 in tracheal aspirates. Similarly, in preterm infants with severe respiratory distress syndrome, the intratracheal administration of budesonide with surfactant (a mixture of 0.25 mg/kg budesonide and 105 mg/kg surfactant) was reported to reduce BPD and mortality without impairing surfactant function [33]. In addition, the intratracheal administration of a mixture of budesonide (0.25 mg/kg) and surfactant (100 mg/kg) did not affect the surface tension-lowering activity or pulmonary distribution of surfactant; moreover, the surfactant/budesonide suspension was biophysically and chemically stable [34].
Since the introduction of the intratracheal administration of budesonide combined with surfactant, ongoing studies have continued to evaluate this approach. A recent systematic review also reported that the intratracheal administration of budesonide with surfactant was associated with reduced rates of death and BPD [35].
Taken together, these findings suggest that the intratracheal administration of corticosteroids with surfactant (a mixture of 0.25 mg/kg of budesonide and 100 mg/kg of surfactant) enhances drug delivery to the distal airways and lowers the incidence of death or BPD; therefore, it represents a promising strategy that remains under investigation. However, long-term neurodevelopmental safety data remain limited, highlighting the need for further large-scale trials.
Conclusion
The optimal administration regimen of postnatal corticosteroids for treating or preventing BPD remains controversial. Evidence suggests that postnatal corticosteroids can facilitate ventilator weaning and reduce BPD risk; however, these benefits must be weighed against potential adverse outcomes, particularly NDI or cerebral palsy. The benefits and risks of postnatal corticosteroids vary depending on the timing of administration, corticosteroid type, dosage, route of delivery, and the infant’s baseline risk of BPD. This emphasizes the need for individualized, risk-based decision-making. Furthermore, determining whether adverse neurodevelopmental outcomes are attributable to extreme prematurity itself, BPD-associated NDI, or dexamethasone or hydrocortisone exposure remains challenging.
Despite the lack of an established optimal regimen, based on the studies conducted to date, a reasonable recommendation is that late dexamethasone administration (>7 postnatal days) to high-risk, ventilator-dependent infants may reduce the rates of death and BPD in the short term and reduce the incidence of cerebral palsy and NDI in the long term.
In conclusion, this review suggested that postnatal corticosteroids should be used cautiously and selectively in ventilator-dependent high-risk infants. Future high-quality multicenter trials should directly compare dexamethasone and hydrocortisone, with long-term NDI-free survival as the primary outcome. Moreover, further research is needed to evaluate the long-term neurodevelopmental outcomes of the intratracheal administration of corticosteroids with surfactant.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contribution
All contributions to this paper were made by the single author (Ga Won Jeon).