Development of multifocal nodular lesions of a liver mimicking hepatic metastasis, following resection of an insulinoma in a child
Article information
Abstract
Insulinoma, which arises from insulin-producing pancreatic beta cells, is a rare tumor in children. Only 5%-10% of insulinomas are malignant and undergo metastasis. We report a case of an 11-year-old girl who experienced hypoglycemia-related seizures induced by an insulinoma; after resection of the primary tumor, she developed hepatic focal nodular hyperplasia (FNH). Laboratory test results indicated marked hypoglycemia with hyperinsulinemia. Abdominal ultrasonography (US) and computed tomography results were normal; however, magnetic resonance imaging (MRI) showed a solid mass in the pancreatic tail. Therefore, laparoscopic distal pancreatectomy was performed. Two months after the surgery, an abdominal MRI revealed multiple nodular lesions in the liver. An US-guided liver biopsy was then performed, and histological examination revealed FNH without necrosis or mitotic activity. The patient has been free of hypoglycemia for 2 years, and recent MRI studies showed a decrease in the size of FNH lesions, without any evidence of metastasis. Even though no metastatic lesions are noted on imaging, close observation and follow-up imaging studies are required in a child with insulinoma that has malignant potential on histopathologic findings.
Introduction
Insulinoma is a rare neuroendocrine tumor of the pancreas, accounting for 2% of all pancreatic tumors. Its occurrence is 1:100,000 and it is more common in women over 50 years of age. However, it may occur in all age groups1). Insulinomas are usually solitary benign tumors, and less than 10% of them are malignant2,3). Malignant insulinomas are diagnosed when metastases occur, but distinguishing malignant insulinomas from benign insulinomas is difficult histologically.
Focal nodular hyperplasia (FNH) represents a localized liver cell hyperplasia containing central and/or stellate fibrous scar4). FNH is also uncommon in childhood. Children account for only 7% of cases of this tumor, and FNH comprises only 2% of all pediatric liver tumors5,6). We report a case of an insulinoma in an 11-year-old child who subsequently developed multifocal nodular lesions of the liver after resections of the insulinoma.
Case report
An 11-year-old girl was admitted for evaluation of recurrent seizures that had begun 5 months before admittance, especially in the morning, despite administration of oral antiepileptic medication. Palpitation and sweating were accompanied with a seizure. Although brain magnetic resonance imaging (MRI) and electroencephalogram studies were all normal 2 months before admittance, she again had a seizure and was prescribed with oral levetiracetam to control her seizure attacks. Ten days prior to admittance, the patient had been brought to the Emergency Department due to an ongoing seizure. Her serum glucose level was 35 mg/dL and she was controlled seizure by intravenous infusion of 10% dextrose fluids. After discharge, she experienced 3 additional attacks of seizures that had been relieved by grape juice. Her family history was unremarkable.
Upon admittance, her height was 161.3 cm (>97th percentile), and her weight was 47.6 kg (75th<percentile<90th) and her body mass index was 18.8 kg/m2 (50th<percentile<75th). All vital signs and the physical characteristics of the organs were all normal. Neurological exams were also normal.
Initial laboratory results, including a serum glucose level of 104 mg/dL, were unremarkable. A 72-hour fast test was initiated, and the patient demonstrated drowsiness, trembling, and tachycardia after less than 6 hours. Laboratory test results during the fast period showed hyperinsulinemic hypoglycemia without ketonuria: the serum insulin level was 50.1 µU/mL (normal range, 2.0-25.0 µU/mL), serum C-peptide level was 1.81 ng/mL (0.59-1.56 ng/mL), and plasma glucose level was 21 mg/dL (70-100 mg/dL). Subsequent glucagon stimulation test results revealed an increase in insulin level to 85.8 µU/mL. Other metabolic and endocrine causes were excluded.
The MRI studies revealed a solid mass, 3 cm×2.4 cm in size, with lobulated margins, in the pancreatic tail (Fig. 1), with no metastasis, while abdominal ultrasonography (US) and computed tomography (CT) results were normal. The patient then underwent laparoscopic distal pancreatectomy. The tumor was easily detected due to its superficial position at the end of the pancreatic tail, and no enlarged lymph nodes were detected around the pancreas or metastatic lesions in the abdominal cavity. The pathologic examination indicated that the resected tumor (3 cm×3 cm×2.5 cm in size) was multilobulated and not encapsulated, and no necrosis was evident. The tumor was of neuroendocrine origin, well differentiated, with an intermediate grade (mitoses 4/10 high-power fields) with angioinvasion. Immunohistochemical staining was positive for insulin, chromogranin, and synaptophysin. The Ki-67 index, a cellular marker for proliferation which <3% means a low grade tumor, was 1% (Fig. 2). After surgery, fasting serum glucose levels were maintained between 110 to 140 mg/dL without any treatment.

Magnetic resonance images of the abdomen. (A) T1-weighted images taken at admission reveal a 3×2.4-cm solid mass (arrow) with lobulated margins in the pancreatic tail. (B, C) Liver acquisition with volume acquisition images taken 2 months later reveal multiple 1.3×1-cm hypointense nodular lesions (arrow) in segment S8 (B) and 0.5×1-cm lesions (arrow) in segment S5 and S6 (C) of the liver.
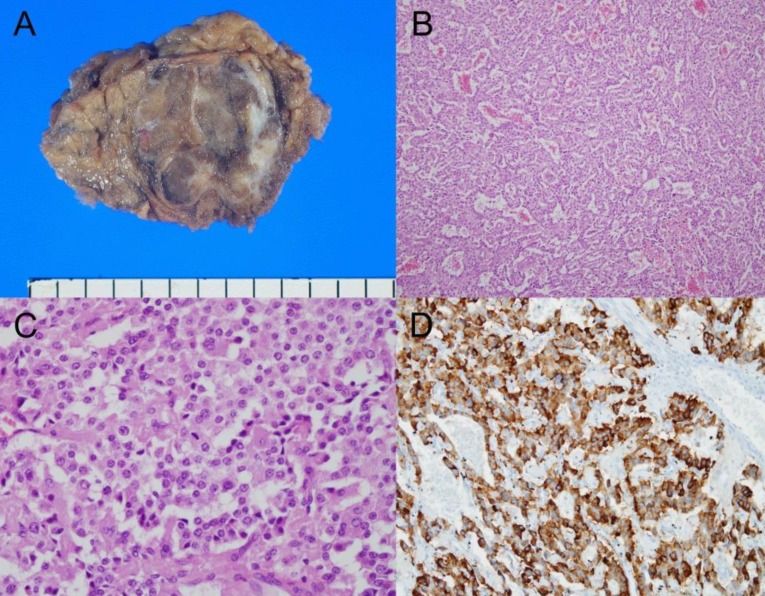
Histological features of the excised insulinoma. (A) A grossly resected specimen of the mass at the pancreatic tail shows a multilobulated solid appearance without hemorrhage or necrosis. (B) Low-power view reveals nesting, trabecular, and gyriform arrangement of the tumor with angioinvasion (H&E, ×100). (C) High-power view reveals uniform tumor cells, showing amphiphilic to eosinophilic cytoplasm and a coarsely granular chromatin pattern with mitoses (H&E, ×400). (D) Immunohistochemical staining for insulin reveals diffuse and a strong-positive reaction in the cytoplasm (×200).
Follow-up MRI studies were conducted 2 months after tumor removal, and revealed multiple hypointense nodular lesions in the hepatic segments S5, S6, and S8 (Fig. 1). US-guided needle biopsy of the liver yielded a diagnosis of the lesions as FNH, with no necrosis or mitotic activity was noted. Since then, the patient has undergone regular follow-up to monitor any possible recurrences. The patient has been free of hypoglycemia for 2 years, and there was not the learning problem in her school. Recent MRI studies have shown that the FNH lesions have decreased in size, without any evidence of metastasis.
Discussion
The classical diagnosis of insulinomas is based on Whipple's triad: hypoglycemia of serum glucose lower than 50 mg/dL, neuroglycopenic symptoms (such as behavioral or personality changes and seizure), and prompt alleviation of symptoms after administering glucose. The neuroglycopenic symptoms often lead to a delay in the correct diagnosis. Approximately 20% of patients with insulinoma are initially misdiagnosed as having epilepsy7). Thus, the suspicion of symptomatic hypoglycemia is essential for early diagnosis of insulinomas.
Malignant insulinoma is extremely rare in the pediatric population, with only a few cases reported in the English literature. Distinctions between benign and malignant insulinomas are based on metastasis or the invasion of the primary tumor into the lymph nodes, tissues, or other organs8). Insulinomas larger than 3 cm are also more likely be malignant, with local invasion or metastases to the liver9). Recently, Janem et al.3) reported a pediatric case of malignant insulinoma in the context of a literature review of 9 preceding cases. Among the nine cases, five cases showed liver metastasis, two cases showed capsular invasion, and data for the remaining two cases were not available. Considering the criteria of malignant insulinoma, which require metastasis or local invasion of adjacent lymph nodes or tissues, these five cases with liver metastasis-together with the case reported by Janem et al.3) involving metastasis to the liver, bone, and bone marrow-may be the only true reported malignant insulinomas to date. However, Sata et al.10) and Janez11) reported cases of malignant insulinomas initially diagnosed as benign, that recurred as liver metastases 8 and 15 years, respectively, after initial removal. Neither of these studies confirmed metastasis or invasion during the initial surgery or in subsequent histologic exams of the initial specimens. Thus, insulinomas may be considered as tentatively malignant because these rare tumors have unpredictable features and progression through the body.
Neuroendocrine tumors have recently been identified as risk factors that are strongly associated with aggressive tumor behavior leading to malignancy12). Histopathologic grading of tumor cells based on mitosis may aid in prediction of future insulinoma outcomes, as the biologic behavior of low-grade tumors is often rather nonaggressive, whereas high-grade tumors are very aggressive12). Our histopathologic findings showed a well-differentiated, intermediate neuroendocrine tumor of the resected insulinoma and our operation field findings revealed no evidence of metastasis. Nevertheless, the possibility of malignancy was included due to the size of the tumor and its histological angioinvasion. The unusual multifocal hepatic lesions following tumor removal were suspected as liver metastases in our case.
FNH of the liver is an uncommon benign tumor in children. It has been infrequently documented in adults and children following chemotherapy, radiotherapy, and stem cell transplantation for different types of solid tumors13). The cause is unknown, although one hypothesis is that obstruction of hepatic vessels or abnormal vascularization could account for FNH, as suggested by the reported association with clinical and anatomic findings like hypoplasia or agenesis of the portal vein, vascular malformations, hemangioma and vascular dysplasia, Budd-Chiari syndrome, and hereditary hemorrhagic telangiectasia14).
Although CT and MRI findings of FNH may commonly include the presence of a central scar and typical enhancement patterns, biopsy is necessary in difficult cases with differential diagnosis of liver adenomas and carcinomas. Treatment recommendations regarding FNH are based on longitudinal follow-up of a small series of patients. Because FNH is a benign tumor with rare complications, most lesions are followed nonoperatively with serial US or MRI, provided that a reliable diagnosis can be achieved using radiologic imaging3,15).
Hepatic lesions can induce diagnostic problems when metastasis must be ruled out. In our case, hepatic nodular lesions were identified as FNH by needle biopsy, but the association of an insulinoma with the FNH after surgery remains unclear.
To our knowledge, this is the first pediatric case report of an insulinoma in which diagnosis was confused due to newly discovered hepatic lesions after tumor resection. These lesions mimicked liver metastasis but were eventually identified as FNH. Close observation and follow-up imaging studies are required in patients who show malignant potential on histopathologic findings.
Notes
No potential conflict of interest relevant to this article was reported.