Prevalence of human parechovirus and enterovirus in cerebrospinal fluid samples in children in Jinju, Korea
Article information
Abstract
Purpose
Human parechovirus (HPeV) and enterovirus (EV) are causative agents of a sepsis-like illness in neonates and of infections of the central nervous system in young children. The objectives of this study were to assess the prevalence of HPeV3 and EV infection in young children with a sepsis-like illness or with meningitis in Jinju, Korea.
Methods
Cerebrospinal fluid (CSF) samples were collected from 267 patients (age range, 1 day to 5 years) and assessed for HPeV and EV by performing reverse transcription polymerase chain reaction assay. Amplification products of the VP3/VP1 region of HPeV and of the VP1 region of EV were sequenced to identify the virus type.
Results
HPeV and EV were detected in 3.4% and 7.5% of the total CSF samples assessed, respectively. The age distribution of EV-positive patients (median age, 1.4 months) had a significantly broader range than that of HPeV-positive patients (median age, 7.8 months). The peak seasons for HPeV and EV infection were spring and summer, respectively. The clinical symptoms for HPeV and EV infection were similar, and fever was the most common symptom. Pleocytosis was detected in 22.2% of HPeV-positive patients and 35.5% of EV-positive patients. The VP3/VP1 gene sequence of the nine Korean strains clustered most closely with the Japanese strain (AB759202).
Conclusion
The data indicate that HPeV infection is predominant in young infants (<6 months) and that meningitis without pleocytosis was caused by both HPeV and EV infection in children.
Introduction
Human parechovirus (HPeV) is a single-stranded RNA virus that belongs to the Parechovirus genus in the Picornaviridae family1). HPeVs were classified into 16 diverse genotypes2). Among the 16 genotypes of HPeVs, HPeV3 has been identified as a causative agent of sepsis-like illness in neonates and has been shown to cause central nervous system (CNS) infection in young children3,4). The clinical symptoms of HPeV infection are similar to those of enterovirus (EV) infection5), and the most common symptoms include fever, seizures, irritability, rash, and feeding problems6). HPeV has been reported as an important viral cause of sepsis-like illness defined as fever or hypothermia with signs of circulatory and/or respiratory dysfunction defined by tachycardia or bradycardia, low blood pressure, and decreased saturation with negative culture in young children4). EV is the most common cause of aseptic meningitis and EV infection is routinely diagnosed by performing reverse transcription polymerase chain reaction (RT-PCR) assay for detecting the virus in cerebrospinal fluid (CSF) or stool samples of patients with CNS infections and neonatal fever7,8). However, HPeV is not routinely screened in young infants with sepsis-like illness and meningitis.
Epidemiologic studies that characterize HPeV prevalence in pediatric patients have been conducted in several countries4,9,10,11,12,13,14,15,16,17). However, the prevalence of HPeV3 in CSF of young children in Jinju, Korea has not been characterized. Therefore, the objectives of this study were to assess the prevalence of HPeV3 and EV infection in young children with sepsis-like illness or with meningitis. The typing of the virus obtained from CSF samples was also performed and the clinical presentations of HPeV3 infection and EV infection were characterized.
Materials and methods
1. Sample collection
A total of 267 CSF samples were obtained from children (aged 5 years or younger) admitted to Gyeongsang National University Hospital (GNUH), Jinju, between February 2012 and September 2013. Lumbar punctures were performed as part of the initial work up of a feverish child under the impression of sepsis, meningitis, encephalitis or seizure with fever in GNUH. These CSF specimens were previously submitted for routine microbiological testing for common bacterial pathogens. Medical records of the study subjects were used to collect data on the clinical manifestations and laboratory findings. Patients diagnosed with bacterial meningitis or malignancies were excluded. The data were collected from standard case record forms and included age, gender, clinical symptoms (vomiting, diarrhea, seizure, and skin rash), onset, laboratory data (blood counts and blood biochemistry), and CSF profiles (cell count, glucose, and protein) of the patients. Blood biochemistry included aspartate transaminase (AST), alanine transaminase (ALT), and C-reactive protein (CRP) measurements. All previously tested CSF samples were used to assess HPeV and EV prevalence regardless of pleocytosis. CSF pleocytosis was defined using the criteria reported in a previous study18); CSF WBC counts>22/mm3 in subjects aged under 4 weeks, >15/mm3 for subjects aged 4-7 weeks, >5/mm3 for subjects aged over 7 weeks.
2. Laboratory testing for HPeV and EV
All CSF samples obtained from the National Biobank of Korea were tested for HPeV and EV prevalence. Total RNA was extracted from CSF samples (200 µL of sample was used for each extraction) by using QIAamp viral RNA kit (Qiagen, Hilden, Germany). Each RNA extract (5µL) was mixed with RT-mix and the RT reactions were performed at 42℃ for 60 minutes, and 95℃ for 5 minutes, followed by incubation at 4℃. To detect the VP1 region of EV, nested RT-PCR assays were performed using the following primers: 224 (5'-GCIA TGY TIG GIA CIC AYR T-3'), 222 (5'-CIC CIG GIG GIA YRW ACA T-3'), AN89 (5'-CCA GCA CTG ACA GCA GYN GAR AYN GG-3'), and AN88 (5'-TAC TGG ACC ACC TGG NGG NAY RWA CAT-3'), as described previously19).
To detect the 5'-NCR region of HPeVs, nested RT-PCR assays were performed with the following primers: 5'-GGG TGG CAG ATG GCG TGC CAT AA-3' (outer sense, nt253-275), 5'-CCT RCG GGT ACC TTC TGG GCA TCC-3' (outer antisense, nt583-560), 5'-AYA CAG CCA TCC TCT RGT AAG TTT G-3' (inner sense, nt313-339), and 5'-GTG GGC CTT ACA ACT AGG TTT G-3' (inner antisense, nt556-534). The nested PCR was performed using 30 amplification cycles that consisted of three steps (30 seconds at 94℃, 30 seconds at 50℃, and 90 seconds at 72℃), followed by a final extension of 7 minutes, as described previously10). To detect the VP3/VP1 gene of HPeV, RT-PCR was performed using previously described modified primers20) as follows: The first reaction was performed using the primers 2090 (5'-GAY AAT GCY ATM TAY ACW ATY TGT GA-3') and 2523 (5'-ACW GTR AAR ATR TCH ACA TTS ATD G-3'). The second reaction was performed using the primers 2159 (5'-TTY TCM ACH TGG ATG MGG AAR AC-3') and 2523 (5'-ACW GTR AAR ATR TCH ACA TTS ATD G-3'). The amplicon was purified using QIAquick (Qiagen) and sequenced in both directions by using the BigDye Terminator v3.1 Cycle Sequencing kit (Applied Biosystems, Foster City, CA, USA). The sequencing reaction products were resolved with an ABI 3730 XL autoanalyzer (Applied Biosystems). Nucleotides sequences were aligned using BioEdit v7.0 and are presented as a topology tree prepared in MEGA 4.1. The nucleotide sequences identified in this study have been deposited in the GenBank database under the accession numbers KF574277-574285 for VP3/VP1 region.
3. Statistical analysis
Statistical analysis was performed for continuous variables (WBCs, neutrophils, platelet, CRP, AST, and ALT) and the variables were recorded as means±standard deviation and analyzed using the t-test. To analyze the association of gender, fever, and duration of admission with HPeV or EV infection, the chi-square test or Fisher exact test were used. Data were analyzed using IBM SPSS Statistics ver. 21 (IBM Co., Armonk, NY, USA). P values of<0.05 were considered to be statistically significant.
4. Ethics statement
The study protocol was approved by the Institutional Review Board of the Inje University Sanggye Paik Hospital (SP IRB 13-022). The agreement exemption was applied for this study because the serum samples were not used to obtain genetic information. The samples were obtained from the National Biobank of Korea, which acquired informed parental consent by following the Institutional Review Board approved protocols.
Results
1. Study group
Of the 267 children who were hospitalized with sepsis-like illness or meningitis, the age distribution was as follows: 82 (30.7%) aged <1 month, 49 (18.4%) aged 1 month, 51 (19.1%) aged 2 months, 26 (9.7%) aged 3 months, 23 (8.6%) aged 4-6 months, 5 (1.9%) aged 7-11 months, 8 (3.0%) aged 1 years, 3 (1.1%) aged 2 years, 5 (1.9%) aged 3 years, 5 (2.2%) aged 4 years, and 10 (3.4%) aged 5 years. Most of the study subjects (77.9%) were under 3 months of age. The sex ratio of the study population was 2:1 (male, 171 patients; female, 96 patients). The clinical diagnosis data indicate that 182 study subjects (68.2%) had sepsis-like illness, 35 subjects (13.1%) had meningitis, and 50 subjects (18.7%) had other illnesses.
2. Frequency of HPeV and EV infections
In 267 CSF samples, 9 (3.4%) were positive for HPeV and 20 (7.5%) were positive for EV. No mixed infection with both HPeV and EV was detected (Table 1). Typing of nine HPeV positive samples was performed by sequencing the VP3/VP1 gene and all the isolated virus strains were identified as HPeV3. The disease onset data indicate that the majority of the HPeV3 infections occurred in spring of 2013 (3 in March, 1 in April, and 1 in May) and the majority of the EV infections occurred in summer (7 in June and 7 in July; Fig. 1). The majority (88.9%, 8/9) of HPeV3-positive patients were under 2 months of age (5 were neonates, 2 were 1 month old, and 2 were aged between 3 and 4 months). Half of the EV positive patients were under 3 months of age and the remaining patients were older than 1 year (ranged from 1 year to 5 years). The age distribution range of EV positive patients was broader than that of the HPeV patients (P=0.027). The median ages of HPeV and EV positive patients were 7.8 and 1.4 months, respectively. The sex ratios (female:male) of the HPeV3 and EV positive patients were 1:2 and 1:1.5, respectively. The clinical presentations of HPeV3 and EV infections were similar. Fever, the most common symptom, was reported in 70% of HPeV3 patients and in 75.5% of EV patients (Table 2). Vomiting and diarrhea were more common in EV-positive patients than in HPeV-positive patients but the differences were not statistically significant. Apnea was diagnosed in 3 neonates. The laboratory test findings of HPeV3-positive patients were also similar to those of EVpositive patients. Pleocytosis was determined in 22.3% of HPeV3-positive patients (2/7) and in 35.5% of EV-positive patients (6/20) (P=0.517). The CRP levels of HPeV3-positive patients were higher than those of EV-positive patients (P=0.046).

Clinical characteristics and laboratory profiles of human parechovirus-3- and enterovirus-positive patients

Monthly incidence of human parechovirus (HPeV) infection and enterovirus (EV) infection. The monthly incidence of HPeV infection (blue bar) and EV infection (gray bar) was determined over a period of 7 months. The peak seasons for HPeV and EV infection were spring and summer, respectively (P<0.001).
3. HPeV and EV type identification
Fig. 2 shows the phylogenic tree of the VP3/VP1 gene of the nine Korean strains, which showed clustering of all Korean strains with HPeV-3. The VP3/VP1 gene sequence of the nine Korean strains clustered most closely with the Japan strain (AB759202). Korean strains in this study also clustered closely with the Korean strain HPeV3 (CSF066/2011/KR, KC196290), in a previous Korean study17) and the VP3/VP1 gene sequence data revealed similarity among the strains ranged from 98.7% to 99.4% (the amino acid similarity: 100%).

Phylogenetic analysis of the VP3/VP1 region of parechovirus. The tree was constructed using the neighbor-joining method with Kimura two-parameter estimation. The bootstrap values from 1,000 replicates are shown for each branch. The strains in this study (KF574276-85) are shown in boldface.
Molecular typing of EV was performed by sequencing the VP1 gene, which revealed that nine patients had Echo 30 serotype, three patients had CVA9 serotype, three patients had CVB4 serotype, two patients had Echo 5 serotype, two patients had CVB1 serotype, and one patient had CVB3 serotype. Serotype Echo 30 was detected most frequently.
Discussion
Previous epidemiologic studies characterizing HPeV prevalence by using CSF samples obtained from pediatric patients have indicated that HPeV prevalence in children ranged from 0.8% to 17.0%4,9,10,11,12,13,14,15,16,17) and majority of the positive cases were aged 6 months or younger4,9,10,11,12,13,14,16,17). In this study, the overall prevalence of HPeV was 3.5%, which was similar to the HPeV prevalence reported in Israeli children from 2007 to 2009 (3.5%)16). However, the HPeV prevalence in this study was lower than that of children aged 1 day to 15 years in Seoul, Korea (6.5%)17).
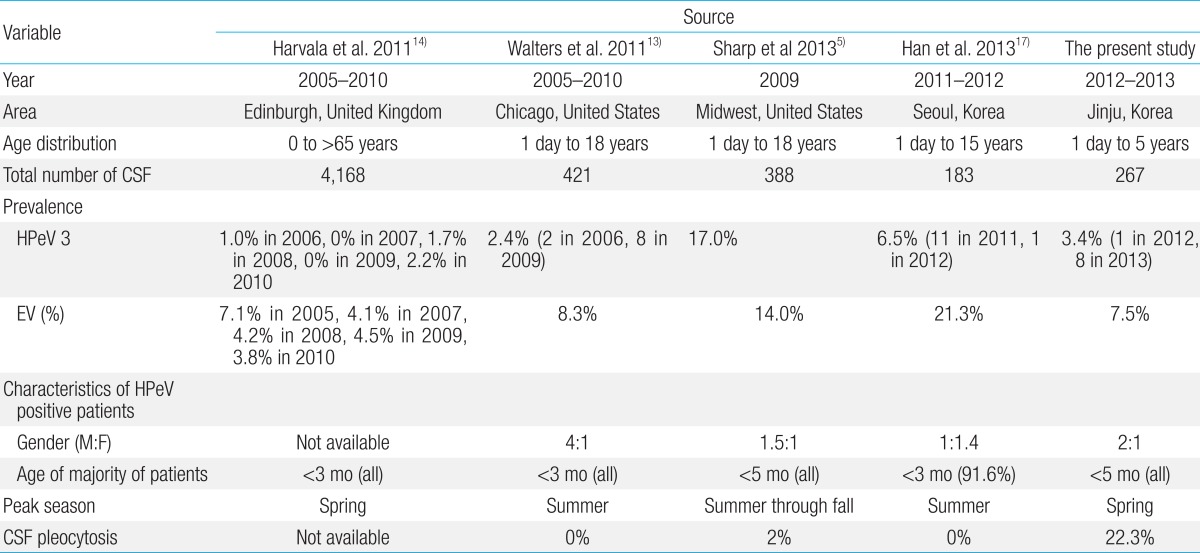
A summary of the findings of four previously reported studies that characterized the prevalence of HPeV infection and EV infection by using CSF samples are shown in Table 2. Previous studies indicated that EV prevalence was higher than HPeV3 prevalence in Edinburgh, and Seoul. This finding was consistent with the findings of this study. However, another previous study showed that EV prevalence was lower than that of HPeV3 in Midwest United States (US). These discrepancies may be due to the differences in susceptibility of various populations.
HPeV3 infections were predominant in infants under the age of 5 months and an increased susceptibility has been previously reported for this age range in studies conducted in various countries5,13,14). A higher prevalence of HPeV infections in younger children may explain the low rates reported in two studies from the United Kingdom (UK)4,10). However, in China, HPeVs were detected in all age groups of children with ages ranging from 2 days to 13 years in 2011. Furthermore, HPeV3 was detected only in three children aged 8 months, 13 months, and 8 years15). However, the epidemiologic profiles of HPeV in China are unknown15). The EV detection rate was more varied than that of HPeV. Harvala et al.14). and Han et al.17). reported that EV infection rate was higher than HPeV infection rate, which is agreement with the findings of our study. However, Sharp et al.5). reported that HPeV infection rate was higher than EV infection rate in a study conducted during the same period that Han et al.17) conducted their study. These results suggest that the circulation pattern of EV varies considerably as compared to the circulation pattern of HPeV. In contrast to the diversity of EVs detected in CSF, all of the 9 parechovirus-positive samples identified were type 3 and similar finding was reported in other studies on prevalence of HPeV infections in CSF samples14,17). Furthermore, EV was detected in infants under the age of 3 months and in other age groups in previous studies14,17) and in this study.
In contrast to the findings of this study, which indicate that most of the HPeV3 infections occurred in spring, previous studies have reported that HPeV3 infections predominantly occur in summer in Europe, America, and Korea13,16,17). However, the spring predominance of HPeV3 infections has been reported in studies conducted in the UK14). The prevalence of HPeV infections can vary annually5,13,17). In 2009, the HPeV prevalence was 0% in the UK14) and 17% in the US5). In this study, 8 of 9 CSF samples that tested positive for HPeV3 collected in 2013. However, this study used a small sample population and was conducted over a short period and a longitudinal study with a larger sample population is required to ascertain whether rate differences in Jinju are influenced by the year and season.
The previously reported findings indicating higher male susceptibility to HPeV35,10,12) were corroborated by this study. However, one study indicated higher female susceptibility in Seoul, Korea17). The data on higher female susceptibility may be influenced by the age groups of the study population and the failure to collect CSF specimens from all patients with fever. In this study, pleocytosis of CSF was determined in 22.3% of the samples and the pleocytosis occurrence was higher than that reported in previous studies (2% in US, and 0% in UK and Seoul, Korea). The high pleocytosis prevalence determined in this study indicates that CSF HPeV PCR testing should be performed for young infants with meningitis.
The principal symptoms of sepsis-like syndrome due to HPeV3 in neonates and young infants are fever, tachycardia, tachypnea, and rashes6). The clinical presentation of the HPeV cases is nonspecific and similar to that of the EV cases5,17). In this study, the clinical manifestations such as fever, vomiting, diarrhea, rash, and apnea were similar in HPeV-positive and EV-positive patients. Infants with HPeV3 had significantly decreased peripheral WBC counts that ranged from 2,630/mm3 to 8,750/mm3 and the mean WBC count of HPeV-positive patients was higher than that of EV-positive patients13), which is similar to the findings of this study. The CRP levels were higher in HPeV3-positive patients than in EV-positive patients. However, the mechanisms underlying the increased CRP levels are unclear because there was no difference in the clinical manifestation of the infection and in other laboratory findings. The platelet count was slightly higher in HPeV-positive patients than in EV-positive patients; however, the platelet count was within the normal range.
In previous Korean studies21,22,23,24), outbreaks of aseptic meningitis caused by various serotypes of EV such as Echo 6, Echo 9, Echo11, Echo13, Echo 18, Echo 25, Echo30, and EV 71 were reported. In this study, Echo 30 (9 patients) was the most frequently detected serotype, which has been previously associated with an epidemic in Gyeongbuk province in 200323). Some of the limitations of this study are that the study was retrospective, included one center study and used sample and subject information that was previously obtained by the Bank.
Overall, the data indicate that HPeV3 infection of the CNS occurred predominantly in young infants during spring and that the clinical presentation of HPeV3 infection was similar to that of EV infection. The screening of the HPeV3 in CSF samples from young infants with sepsis-like illness and meningitis might be necessary in clinical practices evaluating sepsis.
Acknowledgment
The biospecimens used in this study were provided by the Gyeongsang National University Hospital, which is a member of the National Biobank of Korea, which is supported by the Ministry of Health, Welfare and Family Affairs. All samples derived from the National Biobank of Korea were obtained with informed consent under Institutional Review Board of the Inje University Sanggye Paik Hospital (SP IRB 13-022).
Notes
No potential conflict of interest relevant to this article was reported.