Introduction
Candida arthritis in patients with a malignancy is rare, especially in children. Nevertheless, a few cases of Candida arthritis have been reported in immunocompromised patients and neonates1-4). Here, we report a patient with Ewing sarcoma who developed Candida tropicalis arthritis of the elbow during chemotherapy. An initial non-response to treatment with antibiotics in arthritis, particularly in neutropenic patients, should raise suspicion of a fungal infection like candidiasis. Although, amphotericin B is the first choice treatment for Candida infections4-8), it has many side effects, such as electrolyte imbalance, and is only available in an intravenous form. However, fungal arthritis needs at least 3 months of therapy9). On the other hand, itraconazole is available in intravenous and oral forms. We describe the successful treatment of a 13-year-old Ewing sarcoma patient with Candida arthritis using oral and intraveneous itraconazole.
Case report
A 13 year-old boy presented with fever and not swollen discomfort of the right elbow without swelling and tenderness. Four months previously, he had been diagnosed to have Ewing sarcoma of the right ilium and had undergone five cycles of preoperative chemotherapy. He had contracted a C. tropicalis urinary tract infection during a febrile neutropenia episode after chemotherapy, which was treated with itraconazole and broad spectrum antibiotics for 7 days. No growth was reported in the last urine culture after itraconazole treatment. Recently, he had been on preoperative chemotherapy (vincristine, ifosfamide, doxorubicin, and etoposide), which he had completed 9 days prior to presentation.
At presentation, his body temperature was 37.5℃, pulse rate 120 per minute, respiration rate 20 per minute, and blood pressure 130/70 mmHg. physical examination revealed a slight throat infection, but no sinus tenderness, nor tonsilar nor lymph node enlargement. Heart and lung sounds were normal, liver and spleen were non-palpable, and no edema or pain was found in the lower extremities. He complained of discomfort of the right elbow. Laboratory tests revealed pancytopenia, and complete blood count findings were leukocytes 160/mm3, absolute neutrophil count 40/mm3, hemoglobin 9.4 g/dL, and platelet count 8,000/mm3. Broad-spectrum antibiotics (piperacilin/tazobactam and isepacin) were immediately administered after obtaining blood and urine for culture studies.
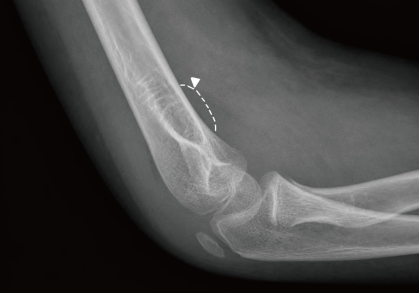
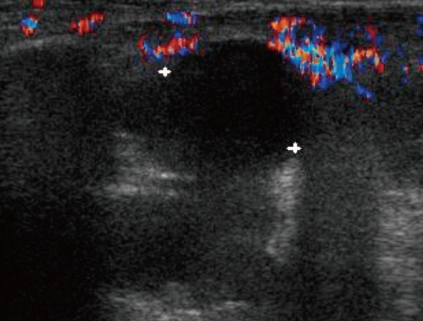
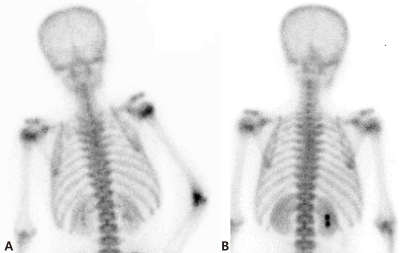
After two days of antibiotic treatment, mild tenderness and swelling (without heating sensation) were detected on the right elbow. A simple radiograph obtained on the 3rd hospital day revealed soft tissue swelling with a positive fat pad sign (Fig. 1). Ultrasonography on the 4th hospital day showed soft tissue edema and articular effusion, and subsequently, 15 mL of yellowish fluid was aspirated from the joint and cultured (Fig. 2). A three phase bone scan (Fig. 3) performed on the 6th hospital day for the differential diagnosis of tumor metastasis and infection showed a limited region of uptake on the right elbow, suggesting the possibility of arthritis.
The symptoms and signs of arthritis aggravated despite a 10-day course of broad-spectrum antibiotics. On the 11th hospital day, cultures of synovial fluid and tissue demonstrated the presence of C. tropicalis. However, blood and urine culture specimens were negative. Follow-up right upper elbow ultrasonography on the 11th hospital day showed elbow articular space effusion and hypertrophic synovium. Emergent decompression was performed on the same day, and a drain tube was placed in the right elbow articular space. During the operation, 2 mL of a yellowish-red turbid synovial fluid sample was obtained and submitted for culture, which later confirmed the presence of C. tropicalis. In addition, abdominal computed tomography on the same day showed no evidence of hepato-splenic candidiasis.
Intraveneous itraconazole 400 mg/day (5 mg/kg/day) was started on the 11th and 12th days. Intravenous itraconazole was decreased to 200 mg/day on the 13th day and then kept at the same dose. Fever, joint swelling, and arthralgia gradually resolved after itraconazole treatment, and after 2 weeks of intravenous itraconazole treatment on the 25th hospital day. Intravenous itraconazole 200 mg/day was substituted for oral itraconazole solution at 200 mg/day.
The antifungal treatment was maintained for three months, and from 14 days after treatment commencement the patient had no elbow pain and was without disability. Furthermore, no relapse of the fungal arthritis occurred during oral itraconazole therapy. Simple radiography and a bone scan carried out after 3 months of antifungal treatment showed no abnormal lesion related to septic arthritis, and oral itraconazole was discontinued.
Discussion
This is the first case report issued on Candida arthritis arising in a child with a solid tumor during chemotherapy in Korea. Previously, only three cases of arthritis have been reported in hematologic malignances in children10-12). Although the exact pathogenesis of Candida arthritis in our case is unclear, it appears to have originated due to disturbed microbial flora levels in the host, and to have spread hematogenously from a previous Candida cystitis infection2,4,13), since dissemination to a joint is possible by direct inoculation (surgery or intra-articular inoculation) or by hematogenous dissemination8). In our case, the arthritis developed 2 weeks after a chemotherapy-induced granulocytopenic period in the absence of any invasive manipulation. Accordingly, hematogenous spread to the synovium of the elbow is believed to be the most likely cause.
Because joint infections (septic arthritis) are rare in the absence of trauma or surgery in children, it is difficult to suspect fungal arthritis in clinical settings. However, joint swelling or arthralgia after a recovery period in neutropenic fever patients should raise suspicion of fungal arthritis4,11). This condition is associated with only mild pain and swelling with persisting low-grade fever. However, growth of the fungus in a synovial fluid culture provides a definite diagnosis. Although, some cases reported facts that synovial fluid in fungal arthritis appears as "yellow-green pus" or "as necrotic tissue", color hardly distinguishes this condition from other types of septic arthritis11).
A review of the literature revealed that Candida albicans and C. tropicalis are major pathogens in adult patients with a hematological malignancy (12 cases of acute leukemia, chronic myeloid leukemia, and small lymphocytic lymphoma), and the knee was the only affected joint in these patients2,4).
Because of its rarity and a lack of experience, no consensus has been reached or guideline issued on the treatment of Candida arthritis9). The objectives of treatment are to relieve symptoms, eradicate infection, and restore joint function. Usually, intravenous amphotericin B (0.5 to 1 mg/kg per day for 6 to 10 weeks) and oral or intravenous fluconazole (6 mg/kg per day for 6 to 12 months) have been used in conjunction with adequate drainage4-8). Some cases have been treated with caspofungin, a newly developed antifungal agent2,12). The described case was treated with itraconazole (at 5 mg/kg per day). Intraveneous itraconazole was administered at 200 mg 12 hourly for 2 days, and then at 200 mg/day, based on evidence that this dosing regimen achieves adequate blood levels more rapidly with less patient-to-patient variability than oral preparations of the drug14). Itraconazole is as effective as amphotericin B in some cases of disseminated Candida infection9,15), and has advantages over amphotericin B for the treatment of granulocytopenia. In fact, the treatment of such infections with amphotericin B is difficult, because of its many side-effects, which include hypotension, fever, shivering, thrombophlebitis, nephrotoxicity, renal tubular acidosis, hypokalemia, anemia and thrombocytopenia15). Thus, prolonged therapy with amphotericin B is difficult in a chemotherapy setting.



 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


