Introduction
Infantile hemangiomas are the most common benign tumors, occurring in up to 10% of infants. Although the majority of hemangiomas occur in otherwise healthy infants, there are increasing reports of associated systemic anomalies. The association of large facial hemangiomas with underlying cerebrovascular anomalies was first reported by Pascual-Castroviejo1). In 1996, Frieden et al.2) proposed the definition of PHACE association to describe these findings associated with large facial hemangiomas. The acronym refers to the association of Posterior fossa malformations, facial Hemangiomas, Arterial cerebrovascular anomalies, Cardiovascular anomalies, and Eye anomalies. More recently, the acronym has been enlarged to include ventral developmental defects (Sternal defects or Supraumbilical raphe)3), thus PHACES association. Most previous studies have stressed the heterogeneity of the association, and strict criteria of inclusion or exclusion have not been universally developed. To our knowledge, PHACE association has been rarely described in Korea. In addition, intracranial hemangiomas are rare as a finding of PHACE association, and concomitant oropharyngeal hemangioma has not been previously reported in the literature of PHACE association. We report a premature infant case of PHACE association with facial, intracranial, and oropharyngeal hemangiomas with evidence of the Dandy-Walker variant and complicated cardiovascular anomalies, including a right-sided aortic arch and an atypical patent ductus arteriosus (PDA) arising from the tortuous left subclavian artery.
Case report
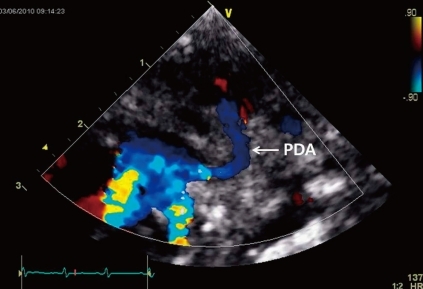
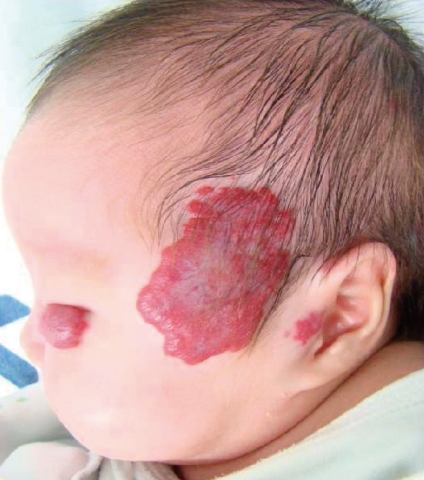
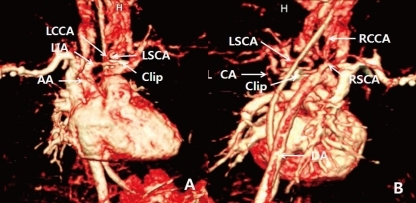
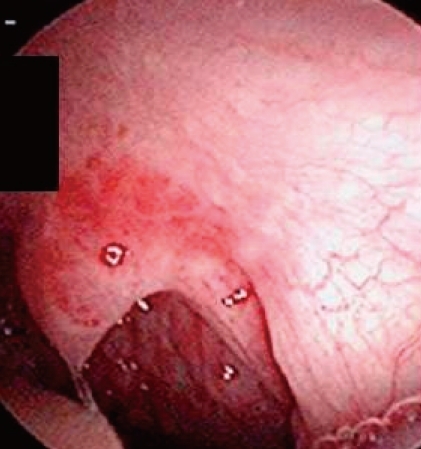
A female infant was born by Caesarean section at a gestational age of 30 weeks and 5 days to a healthy 29-year-old mother. There were no abnormalities demonstrated on routine antenatal ultrasound scans. The birth weight was 1,545 g, and the Apgar scores were 7 and 9 at 1 and 5 minutes, respectively. At 3 days of age, heart murmur and cardiomegaly were detected. Doppler echocardiography done at 4 days of age revealed normal intra-cardiac structure and a suspected PDA measuring 4 mm in diameter between the left subclavian artery and the main pulmonary artery (Fig. 1). Two cycles of oral ibuprofen treatment (10 mg/kg for the first dose, followed at 24-hour intervals by 2 additional doses, 5 mg/kg/dose) were given, but failed to close the PDA. At 1 week of age, small hemangiomas involving the left periauricular, temporal, and infraocular area were detected. The hemangiomas subsequently developed into large facial hemangiomas that threatened the left eye (Fig. 2). Surgery at 2 weeks of age was performed because of the failure of oral ibuprofen treatment and persistent cardiomegaly. Surgery showed an atypical PDA arising from the left subclavian artery and entering the main pulmonary artery, which was closed by surgical clipping. A subsequent three-dimensional cardiac computed tomography (CT) scan and angiography not only confirmed this diagnosis, but also revealed complicated cardiovascular anomalies, including a right-sided aortic arch and the tortuous left subclavian artery. In detail, a three-dimensional cardiac CT scan and angiography showed a mirror image right-sided aortic arch. The first branch of the right-sided aortic arch was a left innominate artery which was divided into a left common carotid and subclavian artery. The second was the right common carotid artery, and the third a right subclavian artery. The PDA arising from the base of the left subclavian artery was closed by surgical clipping, and did not form a vascular ring. A small collateral artery between the thoracic aorta and the left subclavian artery was observed (Fig. 3). Because of the large size of the facial hemangioma, oral prednisolone (1 mg/kg/day) and topical clobetasol-17-propionate were initiated at 1 month of age. At 6 weeks of age, brain magnetic resonance imaging (MRI) and angiography (MRA) were obtained because PHACE association was suspected. These showed the presence of the Dandy-Walker variant with hypoplastic cerebellar vermis and multiple intracranial hemangiomas in left cerebellopontine angle (Fig. 4). No definite abnormalities in the intracranial large vessels were noted on the brain MRA. At 7 weeks of age, laryngoscopic examination revealed a hyperemic lesion on the uvula, and oropharyngeal hemangioma was suspected (Fig. 5). Ophthalmologic and audiometric examinations showed no abnormal findings. No ventral developmental skin defects were noted. The thyroid profile was normal. In addition, no specific findings were detected in the chromosomal analysis, including array comparative genomic hybridization (CGH) to identify gene copy number variations. The prednisolone therapy was gradually tapered and oral propranolol was added at 10 weeks of age. Thereafter, she was treated with intralesional triamcinolone and pulsed dye laser in addition to oral propranolol. This appeared to reduce the proliferation of the hemangioma. No significant changes were noted on a subsequent laryngoscopic examination done at 3 and 8 months of age. At the follow-up examination at 12 months of age, the hemangioma in the infraocular area was significantly decreased. Neurological examinations at 6 and 12 months of age demonstrated no developmental delay for her corrected age. Seizures were not noted. In addition, physical measurement at 12 months showed a significant growth retardation in weight, height, and head circumference (<5th percentile for her age).
Discussion
The PHACE association can be diagnosed in children presenting with a characteristic hemangioma and at least one associated extracutaneous anomaly4). Approximately 30% of affected children exhibit two or more extracutaneous manifestations of the disease, but this is most certainly an underestimate since only a minority of patients has been completely evaluated5). Although the true incidence of PHACE is unknown, the syndrome appears to be uncommon. Metry et al.6) reported that they have collected a database of 201 published cases and 25 new cases, and their PHACE patients represented 2.3% of children with facial hemangiomas in their overall study, and 19.7% of those with segmental hemangiomas of the face. It has been hypothesized that PHACE might be caused by microdeletion(s), a hypothesis which is based partly on the observation that several developmental syndromes with many features in common with PHACE have been found to be caused by microdeletions. A large, multicenter study is under way to investigate the genetic basis of PHACE using array CGH analysis7). In our patient, no abnormal findings were detected in array CGH.
As in previous reports, our patient did not present the full spectrum of abnormalities associated with PHACE association, but nonetheless presented a wide range: facial, intracranial, and oropharyngeal hemangiomas, posterior fossa malformations of Dandy-Walker variants, and complicated cardiovascular anomalies. Large facial hemangiomas are the hallmark of PHACE. Most of the hemangiomas associated with PHACE have a unique appearance, described as "segmental". A segmental hemangioma is defined as a large, plaque-like, and geographic patterning over a territory of skin. Many patients present with an isolated facial segmental hemangioma, although extracutaneous hemangiomas have also been found in approximately one third of patients, the subglottic airway being the most common location, one which can give rise to stridor2,5). On laryngoscopic examnination, our patient had a small hyperemic lesion on the uvula, and we suspected an oroharyngeal hemangioma, instead of a subglottic hemangioma. To our knowledge, an oropharyngeal hemagioma has not been previously described in other reports of PHACE association. Because the patient may be at an increased risk of respiratory or digestive problems with gradual proliferation of the hemangioma, careful monitoring by serial laryngoscopic examinations and proper management will be required. Our patient also had ipsilateral intracranial hemangiomas in the left cerebellopontine angle with large segmental facial hemangiomas. Intracranial hemangiomas are rare as a finding of PHACE although they are being increasingly reported in PHACE patients, likely as a result of more frequent and improved screening with contrast-enhanced neuroimaging3,6,8-12). The intracranial hemangiomas were mainly located in the internal auditory canal and cerebellopontine angle, and involuted in parallel with the facial hemangiomas8,11,13).
Cardiovascular abnormalities are also quite common with PHACE association, and at least one third of infants with this syndrome have cardiovascular anomalies5,6). Aortic arch anomalies, specifically coarctation of the aorta, are the most frequently described cardiovascular anomalies14). Other cardiac anomalies described in PHACE syndrome include right-sided aortic arch, aberrant origin of the subclavian artery, PDA, atrial and ventricular septal defects, persistent left superior vena cava, and rarely, complex congenital heart disease7). Our patient showed complicated malformations, including a right-sided aortic arch and an atypical PDA arising from the tortuous left subclavian artery entering the left pulmonary artery, instead of common malformations such as aortic coarctation or aneurysm. Aberrant origin of PDA and the tortuosity of the left subclavian artery shown in our patient are rare as a finding of PHACE association and quite distinct from other related cardiovascular anomalies reported in PHACE patients. These complicated anomalies also make it difficult to understand the anatomy of the supra-cardiac arteries.
Between 43% and 90% of patients with PHACE have a central nervous system (CNS) structural malformation2,5,6,9,15). Our patient also exhibited a posterior fossa anomaly, Dandy-Walker variant. The posterior fossa malformations constitute the most commonly observed developmental brain abnormalities associated with PHACE, and range from focal cerebellar hypoplasia to Dandy-Walker complex. Additionally, the coexistence of CNS structural malformations and cerebrovascular anomalies is very common. Cerebrovascular anomalies, particularly stenosis and tortuosity may lead to progressive neurologic sequelae, including PHACE-related ischemic stroke. Our patient was not found to have any cerebrovascular anomalies on MRI and MRA. However, because most of the strokes reported occurred at the average age of 8.8 months (range, 3 to 18 months) with progressive obstructive arterial changes and proliferation of the hemangiomas, serial neuroimaging may also be justified in patients without cerebrovascular anomalies on initial examination, as with our patient16,17).
Our patient showed no developmental delays, but significant growth retardation. Despite the multidisciplinary nature of this association, no large, collaborative, cross-specialty studies have been taken. Accordingly, very little is known about the natural history of PHACE, or outcomes of affected infants including premature infants like our patient7).
This case is significant in that intracranial, oropharyngeal hemangiomas and an atypical PDA arising from the tortuous left subclavian artery are specific and rare findings in PHACE patients. In infants presenting with large, plaque-like facial hemangiomas, it is important to conduct active neurological and cardiovascular evaluations. Especially, special attention should be given to the laryngoscopic examination to search for hemangiomas in airway.




 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


