Article Contents
| Korean J Pediatr > Volume 60(7); 2017 |
|
Abstract
Purpose
Methods
Results
Conclusion
Acknowledgments
Notes
Conflict of interest:
No potential conflict of interest relevant to this article was reported.
References
Fig.┬Ā1
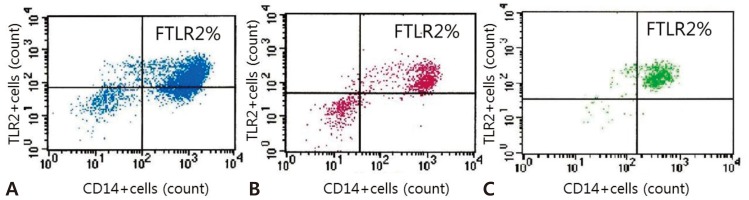
Flow cytometry analysis of whole blood samples from patients with Kawasaki disease and febrile controls (FCs). Toll-like receptor 2 (TLR2) expression in representative cases of CAL (+), CAL (ŌłÆ), and FC groups are shown. Total 50,000 cells were counted from each whole blood sample, and after appropriate gating, monocyte population was identified as CD14-positive (CD14+) cells in the scatter diagrams. FTLR2% was identified as the fraction of cells expressing both CD14 (horizontal axis) and TLR2 (vertical axis), upper right segment in each box. (A) Fraction of cells expressing both CD14 and TLR2 (FTLR2%) from a patient with coronary artery lesions (CAL (+)) is shown in blue. (B) Fraction of cells expressing both CD14 and TLR2 (FTLR2%) from a patient without coronary artery lesions (CAL (ŌłÆ)) is shown in red. (C) Fraction of cells expressing both CD14 and TLR2 (FTLR2%) from an FC is shown in green.
Table┬Ā1
Demographic and laboratory data of patients with Kawasaki disease, with and without coronary artery lesions
Values are presented as mean┬▒standard deviation or number (%).
CALs (+), patients with coronary artery lesions one month after diagnosis; CALs (ŌłÆ), patients without coronary artery lesions one month after diagnosis; IVIG, intravenous immunoglobulin; WBC, white blood cell; CRP, C-reactive protein; AST, aspartate aminotransferase; ALT, alanine aminotransferase.
*P<0.05 when compared with febrile controls.
Table┬Ā2
Demographic and laboratory data of patients with Kawasaki disease who were responsive or resistant to immunoglobulin treatment
Values are presented as mean┬▒standard deviation or number (%).
CALs (+), patients with coronary artery lesions one month after diagnosis; CALs (ŌłÆ), patients without coronary artery lesions one month after diagnosis; IVIG, intravenous immunoglobulin; WBC, white blood cell; CRP, C-reactive protein; AST, aspartate aminotransferase; ALT, alanine aminotransferase.
*P<0.05 when compared with febrile controls. ŌĆĀP<0.05 for comparison of nonresponders vs. responders.
Table┬Ā3
Echocardiographic parameters of left ventricular systolic function in patients with Kawasaki disease
Values are presented as mean┬▒standard deviation.
Nonresponders: patients with Kawasaki disease who did not respond to the initial treatment with intravenous immunoglobulin. Responders: patients with Kawasaki disease who responded to the initial treatment with intravenous immunoglobulin.
CALs (+), patients with coronary artery lesions; CALs (ŌłÆ), patients without coronary artery lesions; LVEDD, left ventricular end-diastolic dimension 1 month after diagnosis; LVESD, left ventricular end-systolic dimension 1 month after diagnosis; FS, left ventricular fractional shortening.
*P value of comparison between CAL (+) vs. CAL (ŌłÆ). ŌĆĀP value of comparison between nonresponders vs. responders.
Table┬Ā4
Frequencies of Toll-like receptor 2 expression/CD14+ monocytes in patients with Kawasaki disease and febrile controls
Values are presented as mean┬▒standard deviation.
KD, Kawasaki disease; CALs (+), patients with coronary artery lesions; CAL (ŌłÆ), patients without coronary artery lesions, KD: Kawasaki disease; TLR2, frequency of circulating TLR2+/CD14+ monocytes before the initial treatment; TLR2P, frequency of circulating TLR2+/CD14+ monocytes after the initial treatment.
*P<0.05 when compared with febrile controls. ŌĆĀP<0.05 for comparison of CALs (+) with CALs (ŌłÆ).



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


