Article Contents
| Clin Exp Pediatr > Volume 65(6); 2022 |
|
Abstract
Background
Congenital muscular torticollis (CMT) is a common musculoskeletal disorder occurring at birth or in infancy.
Purpose
This study aimed to investigate the risk of comorbidities in CMT and explore the differences in neurodevelopmental outcomes between children who received physical therapy and those who did not.
Methods
Children with CMT born in 2008 and 2009 in Korea were included. CMT was defined as a primary diagnosis of congenital deformity of sternocleidomastoid muscle according to the International Classification of Diseases 10th revision. Here we investigated the associated neurological/musculoskeletal comorbidities of children with CMT versus those of the general population. We divided those with torticollis into treatment and nontreatment groups and assessed their developmental outcomes within both groups of children using the Korean-Ages and Stages Questionnaire (K-ASQ).
Results
Of the 917,707 children, 0.2% (n=1,719) were diagnosed with CMT. In children with torticollis, the prevalence of congenital hip deformities significantly increased to 4.5% (n=78). The prevalence of congenital head/spine deformities and other congenital malformations of the skull and facial bones increased to 2.6% (n=44), while the prevalence of congenital foot deformities was 2.4% (n=42). The risk ratio (RR) for delayed development based on the K-ASQ was higher for the total assessment (adjusted RR=0.97; 95% confidence interval, 0.93ŌĆō0.99) in the CMT patients without physical therapy than in those with therapy. There was no significant intergroup difference in the assessment of each developmental area.
Graphical abstract.
Torticollis, literally meaning ŌĆ£twisted neck,ŌĆØ may present as a single disease. However, torticollis may be a clinical manifestation of various underlying conditions such as congenital disorders, trauma, inflammation, and neurological diseases.
Congenital muscular torticollis (CMT), the most common musculoskeletal disorder that occurs at birth or in infancy, is characterized by a contracted and shortened sternocleidomastoid muscle (SCM), which results in tilting of the head toward the affected muscle and rotation of the chin to the opposite side [1-3]. CMT is presumed to occur as local fibrosis with a contracture accompanied by muscle-localized ischemia caused by compression, deformation, or stretching of the SCM [4].
In children with CMT, the incidence of developmental dysplasia of the hip may increase due to intrauterine crowding, and positional comorbidities may occur due to the twisted position [3,4]. Disorders associated with CMT include plagiocephaly, facial asymmetry, and positional musculoskeletal deformities such as cervical scoliosis, vertebral wedge degeneration, metatarsus adductus, and calcaneovalgus feet (talipes calcaneovalgus). Although there are several prior studies of comorbidities associated with CMT, they have not explored other comprehensive neuromuscular skeletal disorders and have been limited to a small scale [5-8].
If CMT is not treated early, it can affect the childŌĆÖs appearance, and cause visual impairment and neurodevelopmental delay [9-13]. Conservative treatment of CMT includes home stretching and Physical and Rehabilitation Medicine (PRM) intervention. Postural torticollis identified in an early phase can be rapidly resolved with positioning or simple home stretching, but most cases diagnosed after 3ŌĆō6 months of age or severe type with SCM mass require long-term exercise therapy by PRM physician [14]. Exercise therapy for CMT includes several components, such as manual stretching of SCM muscle, active rotation exercises, positioning and handling, and environmental adaptation, parent/caregiver education [15].
There have been controversies over early treatment in children with CMT. Many studiesŌĆÖ results indicated that early interventional treatment can reduce secondary damage or complications in CMT patients [4,14,16]. Other studies reported that most patients spontaneously resolved within the first year of birth [7,17]. To our knowledge, there has been no large-scale study that has investigated and shown the developmental outcomes of the treatment and nontreatment cases in CMT.
Accordingly, the aim of this study was to investigate the risk of more comprehensive comorbidities related to CMT using a population-based nationwide design. In addition, we attempted to explore the difference in neurodevelopmental outcomes between the children with CMT who received physical therapy and those who did not.
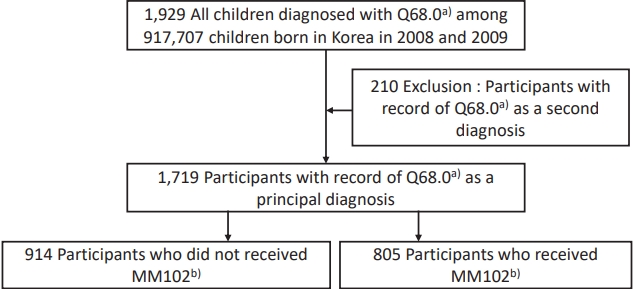
This retrospective study was conducted on children diagnosed with CMT among 917,707 children born in Korea in 2008 (n=469,248) and 2009 (n=448,459) and followed up through 2017. Of the 917,707 children, 10,000 who had never been diagnosed with CMT were randomly selected as the control group. We investigated the frequency of comorbid diseases commonly associated with CMT in each group, respectively. We divided children with CMT into treatment and nontreatment groups and compared neurodevelopmental outcomes. The analysis included children who had been screened by at least one National Health Screening Program for Infants and Children (NHSPIC) for children from ages 42 to 71 months (Fig. 1).
Data were collected from the National Health Insurance Service (NHIS) and the NHSPIC. The NHIS is a single insurer system that covers almost all of KoreaŌĆÖs population and the NHSPIC is a child surveillance program consisting of 7 follow-up screening surveys for infants and children from the age of 4 to 71 months. The NHIS provides insurance benefits for medical services including injuries, childbirth, deaths, health promotions, rehabilitations, or preventions of diseases, and maintains national records for healthcare utilization, prescriptions, and health screening programs. The NHSPIC survey includes a general health questionnaire, developmental screening, an oral health questionnaire, oral examinations and guidance, anthropometric examinations, physical examinations, and anticipatory guidance [18]. Clinical information of children was collected from medical charts and recorded using the International Classification of Disease, 10th version (ICD-10).
The protocol of this study was reviewed and approved by the Institutional Review Board of the Korea National Institute for Bioethics Policy (P01-201603-21-005) and conducted in accordance with the WMA Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects.
CMT was defined as cases diagnosed with the primary diagnosis of ŌĆ£congenital deformity of sternocleidomastoid muscleŌĆØ(Q68.0) according to the ICD-10 code. ŌĆ£Congenital deformity of sternocleidomastoid muscleŌĆØ included ŌĆ£congenital (sternomastoid) torticollis,ŌĆØ ŌĆ£contracture of sternocleidomastoid (muscle)ŌĆØ and ŌĆ£sternomastoid tumor (congenital).ŌĆØ Physical therapy for CMT was defined as receiving complex therapeutic exercise by physical therapists.
The outcomes of this study were the comorbidities and neurodevelopmental outcomes in children with CMT. Comorbidities were defined as musculoskeletal diseases known as related disorders with CMT, and malformations of the nervous system. The prevalence of comorbidities was evaluated by ICD-10 diagnosis. These included congenital malformations of the nervous system (Q00-Q07), congenital malformations and deformations of the musculoskeletal system (Q65-Q79), and chromosomal abnormalities (Q90-Q99). Musculoskeletal deformations were classified into congenital hip deformities (Q65.*), congenital foot deformities (Q66.*), congenital musculoskeletal deformities of head (Q67.0ŌĆōQ67.5), other congenital musculoskeletal deformities (68.* except 68.0), polydactyly, and syndactyly (Q69.*ŌĆōQ70.*), reduction of defect of upper lower, unspecified limb (Q71.*ŌĆōQ74.*), other congenital malformations of skull and face bones (Q75.*), congenital malformations of spine (Q76.0ŌĆōQ76.4), osteochondrodysplasia (Q77.*ŌĆōQ78.*), and other congenital malformation of musculoskeletal system (Q79.*).
In this study, the neurodevelopmental outcomes were compared between the physical therapy and the nonphysical therapy groups using the developmental screening test. The developmental screening test is a major screening tool of the NHSPIC and is performed after 9 months of age (2nd through 7th screening surveys). From 2008 to 2013, the Korean Ages and Stages Questionnaire (K-ASQ) was used as the main developmental screening tool, and from 2014 onward, the Korean Developmental Screening Test for Infants and Children was mainly used. For the identicalness of research methods in this study, we only used the results of the 5th to the 7th developmental screening tests conducted through the K-ASQ. K- ASQ comprises 5 developmental areas (communication, gross motor skills, fine motor skills, problem-solving, and personal-social interrelation) that are assessed under 2 categories: ŌĆ£appropriateŌĆØ and ŌĆ£need for follow-up.ŌĆØ The total evaluation assesses the domains under 3 categories: ŌĆ£appropriate,ŌĆØ ŌĆ£need for follow-up,ŌĆØ and ŌĆ£recommendation for further evaluation.ŌĆØ If a child is considered to have developmental delays based on further evaluation, the result is classified as ŌĆ£need for consistent management.ŌĆØ If at least one of the responses to the ŌĆ£need for follow-up,ŌĆØ ŌĆ£recommendation for further evaluation,ŌĆØ and/or ŌĆ£need for consistent managementŌĆØ categories in the total evaluation is ŌĆ£yes,ŌĆØ the result is classified as ŌĆ£need for evaluation.ŌĆØ [19] There was some variability in the final study sample because there were children who failed to complete some areas of the K-ASQ.
All covariates, such as sex, residence at birth, income quintile, type of milk feeding, and prematurity were collected from the NHIS database. The NHIS provides a public database of healthcare utilization, sociodemographic parameters, and outcomes of the NHSPIC. The economic status was divided into quintiles and determined by the amount of insurance copayment (1Q: poorest quintile, 5Q: richest quintile). Residence at birth was categorized as follows: Seoul, metropolitan areas (Busan, Daegu, Incheon, Gwangju, Daejeon, Ulsan), city (urban areas), and rural (noncity) areas.
The statistical comparison of the incidence of comorbidities in children with CMT was conducted by independent t test. The difference between the treatment group and nontreatment group was significant if the P value was less than 0.05. We calculated risk ratio (RR) and 95% confidence interval (CI) using modified Poisson regression model to compare the difference in neurodevelopmental outcome between the 2 groups. We adjusted for RR considering sex, the residence of birth, household income, type of milk feeding, prematurity, and birth weight, as recorded in the database. All analyses were performed using SAS Enterprise Guide ver. 7.1 (SAS Institute Inc., Cary, NC, USA).
Of the 917,707 children, 1,719 (0.2%) were diagnosed with CMT, and median age at diagnosis was 4 months of age (mean age┬▒standard deviation [SD], 16.5┬▒20.1 months). Of children with CMT, 47% (n=805) received physical therapy and 53% (n=914) did not. The median age at onset of physical therapy in treatment group was 2 months of age (mean┬▒SD, 3.2┬▒2.5 months). All children of the treatment group started therapy within 1 year of birth; 83.7% (674 of 805) started within the first 6 months, and 16.3% (131 of 805) started at 6ŌĆō12 months of age.
The general characteristics of children with CMT and controls are described in Table 1. The male to female ratio was 6:4 in the CMT group, and males were more prevalent. There was no difference in birth residence, income quintile, feeding type, and prematurity.
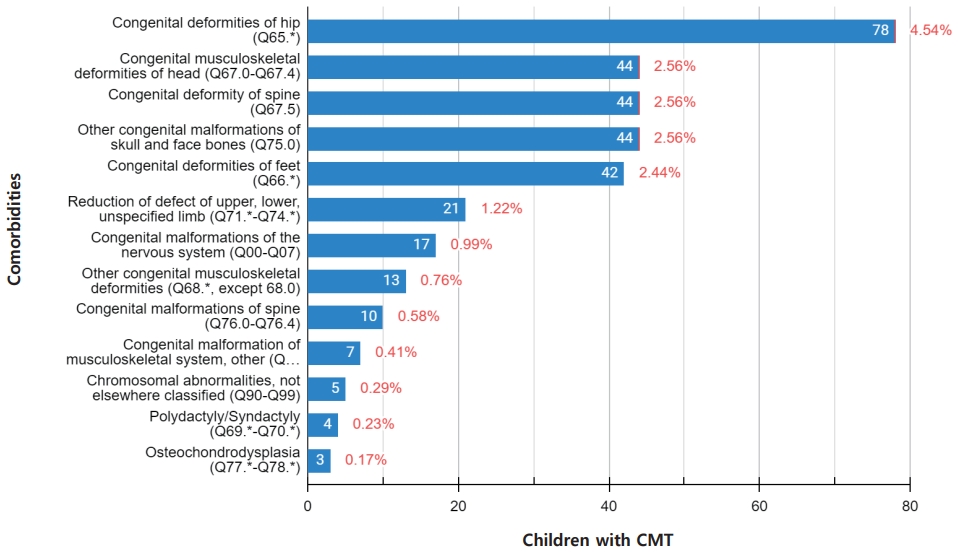
In children with CMT, the incidence of congenital hip deformities was significantly increased to 4.5% (n=78), and congenital deformities of the head including plagiocephaly and facial asymmetry, spinal deformities, other congenital malformations of the skull and face bones were increased to 2.6% (n=44). The incidence of congenital foot deformities including metatarsus adductus, and talipes calcaneovalgus was 2.4% (n=42). However, the incidence of neurologic deformities (n=17) and chromosomal abnormalities (n=5) was less than 1% (Fig. 2, Table 2). Of the 1,719 children with CMT, 13.6% (n=233) had one or more congenital musculoskeletal deformities. One hundred sixty-five children had 1 comorbidity, 22 children had 2, and 46 children had more than 3 comorbidities (Table 3).
The RR for delayed development based on the results from the 5thŌĆō7th K-ASQ was higher for the total developmental area (adjusted RR=0.97; 95% CI, 0.93ŌĆō0.99) in the nontreatment group than in the treatment group. There was no significant difference between the 2 groups in the assessment of communication, gross motor, fine motor, problem-solving, or personal/social developmental areas (Table 4).
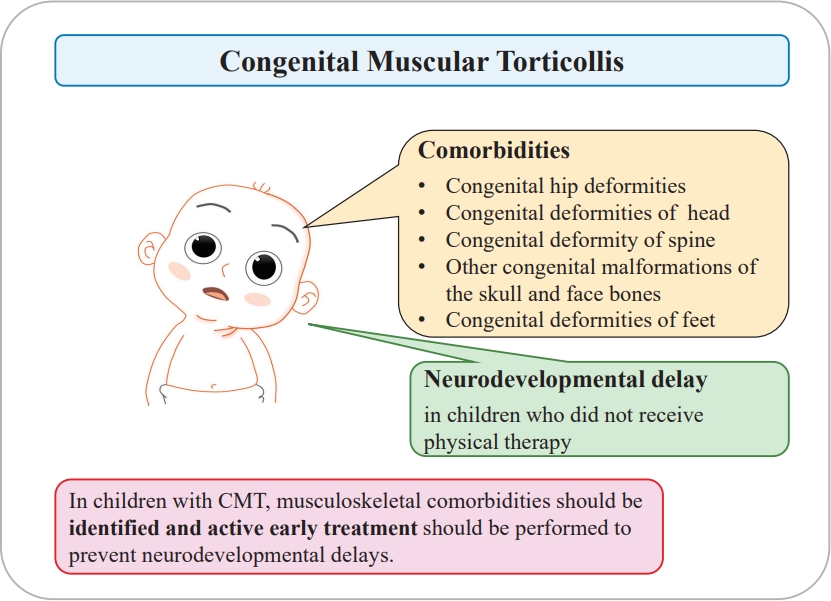
The prevalence of CMT in South Korea was 0.2%. The risk of congenital hip deformities was the highest in CMT, and the risk of congenital deformities of the head, skull and facial bones, spine, and feet was also increased significantly. The risk of developmental delay was higher for children who did not receive physical therapy than those who did not.
The prevalence of CMT in South Korea was 0.2% in this study. Male to female ratio was 6:4. The incidence of CMT is reportedly 0.3% to 2% [4,17]; however, the incidence may be higher. Stellwagen et al. [20] reported that CMT affected 16% (N=102) of newborns. The incidence of CMT in our study was lower than that reported in other studies. However, in our study, only cases with congenital deformity of the SCM were included. Since most of SCM tightness and postural torticollis without tumor were excluded by inclusion criteria, it is possible that our results are lower than reality. In this study, CMT was more common in males and was similar to previous studies [21,22].
Although the etiology of CMT remains unclear, birth trauma [23], perinatal compartment syndrome [24], and impairment of the developing SCM caused by intrauterine restrictions [25-27] are known causes. In addition, breech presentation, multiple pregnancies, primiparity, and dystocic labor are considered risk factors for CMT [14]. However, in our study, we could not investigate risk factors for CMT such as abnormal fetal positioning, continuous neck rotation in utero, and difficult delivery. In CMT, pathological features of the affected SCM include excessive endomysial and perimysial fibrosis, adipocyte hyperplasia, and muscle fiber atrophy. These features cause tightness of the involved SCM and limit cervical movement. An immunohistochemical study mentioned that deposition of type III collagen is a key factor in the SCM fibrosis that causes CMT, and that this hyperplasia is associated with accelerated apoptosis and overexpression of transforming growth factor-beta [28].
We analyzed the incidence of comorbidities in children diagnosed with CMT. In our study, the risk of congenital hip deformities was significantly increased. We also found the risk of congenital deformities of the head, skull and facial bones, spine, and feet to be increased. According to previous studies, 6%ŌĆō20% of children with CMT have congenital musculoskeletal disorders including congenital hip dysplasia, craniofacial asymmetry [5], cervical spine dysmorphism [6,29], and plagiocephaly [7,30]. Hip dysplasia has been identified in 0%ŌĆō20% children with CMT [8]. In our study, hip dysplasia was found in 4.5% of the children with CMT. In addition, congenital deformities of the head including plagiocephaly and facial asymmetry, spinal deformities, and other congenital malformations of the skull and face bone were observed in 2.6%. The incidence of congenital foot deformities including metatarsus adductus and talipes calcaneovalgus was 2.4%. Our study suggests that hip dysplasia is the most common musculoskeletal anomaly in CMT, and that the risk of plagiocephaly, skull and facial asymmetry, and spinal and foot deformities is also increased. Among children with CMT, 13.6% had one or more congenital musculoskeletal deformities. This suggests that CMT can often occur in the complex syndrome of the musculoskeletal system with various congenital deformities.
We compared the neurodevelopmental outcomes of children with CMT who received physical therapy versus those who did not. In the total assessment, the risk of neurodevelopmental delay was higher in the untreated children than in the treated children. However, there was no significant difference between the 2 groups in each developmental area.
Several studies have shown that the risk of neurodevelopmental delays increases in children with CMT. ├¢hman et al. [11] studied 82 infants with CMT between 2 and 18 months of age and found an increased risk of delay in early motor milestones until 10 months of age. Schertz et al. [12] examined neurodevelopmental outcomes based on telephone interviews for children who were aged 7ŌĆō9 years and had a history of CMT, and they found developmental delays/disorders in 30 of 68 (44.1%) of the children. They suggested that the risk of neurodevelopmental disorder such as developmental coordination disorder (32%), and attention deficit hyperactivity disorder (44%) increased in children with CMT. Some studies have also reported contradictory outcomes. In a recent study of children aged 3.5ŌĆō5 years, patients diagnosed with CMT in infancy were not at a higher risk of delayed motor development at preschool age [31]. Although the mechanism underlying the increased risk of delayed motor development and neurodevelopmental disorders in children with CMT remains unclear, various hypotheses have been proposed. Hylton [21] reported that postural torticollis or tone dysfunction may affect antigravity postural control or balance. Michel and Harkins [32] reported that complex postural asymmetry affects the development of motor preferences, as shown by the development of dextral bias in handedness development. In addition, in children with CMT, asymmetry or absence of fidgety movements in early infancy may present other variants of abnormal general movement or provide an important factor that increases the risk of later neurodevelopmental disorders [12].
There is evidence that early physical therapies for neckstretching improve neurodevelopmental outcomes in children with CMT. In one study that followed up the gross motor and cognitive development of 101 children with CMT at a mean age of 2.9 months, the risk of a delay in early gross motor development was increased; however, physical therapy normalized most of the children by 10ŌĆō12 months of age [9].
In our study, physical therapy for CMT was found to be effective in overall development, but the effect of development in individual areas is unclear. We assumed that this is not because physical therapy is insufficient. Rather, this may be due to most infants with CMT appearing to have an appropriate outcome in communication, problem-solving, and personal/ social developmental area. Although this study did not show clear evidence that the motor domains of ASQ were improved in patients with CMT after neck-stretching therapy, it would be important to implement of early rehabilitation treatment, as it showed an overall improvement in neurodevelopment after physical therapy.
Early physical therapy with neck-stretching is more effective than later therapy in children with CMT. Infants who started early intervention within 6 weeks of age showed better outcomes than those in whom early intervention was initiated 6 weeks later [16]. One study reported that if treatment is started by 1 month of age, 98% of infants are normalized within 1.5 months; however, if treatment is not started by 1 month of age, physical therapy is prolonged for up to 6 months. Furthermore, if treatment is not started by 6 months of age, then it can be required for 9ŌĆō10 months [33]. Demirbilek and Atayurt [34] reported that surgery was not required for infants who started physical therapy before 3 months of age; however, 25% of those who started treatment after 3 months of age required surgery.
In our study, 83.7% of the treatment group received physical therapy within the first 6 months of life, and the remaining 16.3% received between 6ŌĆō12 months of age. Because these children did not receive any further physical therapy after 1 year of age, we can assume that most children with CMT who do not have comorbidities are presumed to have good outcomes.
The strength of our study is that it is a large-scale study based on comprehensive and nationwide data of children with CMT. In addition, to our knowledge, this is the first study to analyze the incidence of comorbidities and neurodevelopmental outcomes for children with CMT who were born in South Korea.
However, this study also has some limitations. We have only performed a retrospective analysis of data recorded using ICD codes. Therefore, we may not have derived detailed and accurate results of comorbid disorders and diseases in children with CMT. We did not include positional torticollis and did not suggest risk factors of CMT individually such as fetal position in the uterus and difficult delivery. Furthermore, we have not been able to complete long-term follow-up on the neurodevelopmental outcomes of children with torticollis. Therefore, detailed studies on long-term follow-up of neurodevelopmental outcomes in children with CMT will be needed.
In conclusion, if CMT is suspected, early diagnosis should be established, and comorbidities should be identified. In addition, active early treatment should be performed to prevent neurodevelopmental delays and associated musculoskeletal disorders and to promote normal growth and developmental outcomes.
Fig.┬Ā1.
Flow diagram of the study participant enrollment. a)Q68.0 means congenital deformity of sternocleidomastoid muscle in the International Classification of Diseases 10th revision codes. b)MM102 means complex therapeutic exercise.
Table┬Ā1.
Demographic characteristics of the study population
| Characteristic | Congenital muscular torticollis (n=1,719) | General population (n=10,000) |
|---|---|---|
| Sex | ||
| ŌĆāBoy | 1,008 (58.6) | 5,083 (50.8) |
| ŌĆāGirl | 711 (41.4) | 4,917 (49.2) |
| Birth residencea) | ||
| ŌĆāSeoul | 472 (27.5) | 2,572 (25.7) |
| ŌĆāMetropolitan | 391 (22.8) | 2,254 (22.5) |
| ŌĆāCity | 599 (34.9) | 3,786 (37.9) |
| ŌĆāRural | 241 (14.0) | 1,283 (12.8) |
| ŌĆāMissing | 16 (0.9) | 105 (1.05) |
| Income quintileb) | ||
| ŌĆā1 (lowest) | 110 (6.4) | 820 (0.8) |
| ŌĆā2 | 221 (12.9) | 1,457 (14.6) |
| ŌĆā3 (middle) | 432 (25.1) | 2,502 (25.0) |
| ŌĆā4 | 610 (35.5) | 3,131 (31.3) |
| ŌĆā5 (highest) | 295 (17.2) | 1,702 (17.00) |
| ŌĆāMissing | 51 (3.0) | 388 (3.9) |
| Type of milk feedingc) | ||
| ŌĆāBreast | 436 (25.4) | 2,253 (22.5) |
| ŌĆāBottle | 421 (24.5) | 1,820 (18.2) |
| ŌĆāMixed | 219 (12.7) | 948 (9.5) |
| ŌĆāSpecial formula | 4 (0.2) | 20 (0.2) |
| ŌĆāMissing | 6 (0.4) | 4,959 (49.6) |
| Birth year | ||
| ŌĆā2008 | 857 (49.9) | 5,173 (51.7) |
| ŌĆā2009 | 862 (50.2) | 4827 (48.3) |
| Prematurity | ||
| ŌĆāNo | 1,537 (89.4) | 8,695 (87.0) |
| ŌĆāYes | 113 (6.6) | 504 (5.0) |
| ŌĆāMissing | 69 (4.0) | 801 (8.0) |
Table┬Ā2.
Associated comorbidities in children with congenital muscular torticollis
| Comorbiditiesa) | Congenital muscular torticollis (n=1,719) | General population (n=10,000) | P value |
|---|---|---|---|
| Congenital malformations of the nervous system (Q00ŌĆōQ07) | 17 (1.0) | 0 (0.0) | |
| Congenital deformities of hip (Q65.*) | 78 (4.5) | 2 (0.02) | <0.001 |
| Congenital deformities of feet (Q66.*) | 42 (2.4) | 1 (0.01) | 0.075 |
| Congenital musculoskeletal deformities of head (Q67.0ŌĆōQ67.4) | 44 (2.6) | 1 (0.01) | <0.001 |
| Congenital deformity of spine (Q67.5) | 44 (2.6) | 1 (0.01) | <0.001 |
| Other congenital musculoskeletal deformities (Q68.X, except 68.0) | 13 (0.8) | 0 (0) | |
| Polydactyly/Syndactyly (Q69.*ŌĆōQ70.*) | 4 (0.2) | 0 (0) | |
| Reduction of defect of upper, lower, unspecified limb (Q71.*ŌĆōQ74.*) | 21 (1.2) | 0 (0) | |
| Other congenital malformations of skull and face bones (Q75.*) | 44 (2.6) | 1 (0.01) | <0.001 |
| Congenital malformations of spine (Q76.0ŌĆōQ76.4) | 10 (0.6) | 0 (0) | |
| Osteochondrodysplasia (Q77.*ŌĆōQ78.*) | 3 (0.2) | 0 (0) | |
| Congenital malformation of musculoskeletal system, other (Q79.*) | 7 (0.4) | 1 (0.01) | <0.001 |
| Chromosomal abnormalities, not elsewhere classified (Q90ŌĆōQ99) | 5 (0.3) | 0 (0) |
Table┬Ā3.
Incidence of comorbidities in children with congenital muscular torticollis
| No. of comorbidities | Congenital muscular torticollis (n=1,719) |
|---|---|
| 1,486 (86.4) | |
| 1 | 165 (9.6) |
| 2 | 22 (1.3) |
| Ōēź3 | 46 (2.7) |
Table┬Ā4.
Outcomes of the 5thŌĆō7th developmental screening sessions in children with congenital muscular torticollis
| Developmental areaa) | Nontreatment group (n=914) | Treatment group (n=805) | P value | RR (95% CI) | Adjusted RRb) (95% CI) | |
|---|---|---|---|---|---|---|
| Total | 0.10 | 0.98 (0.95ŌĆō1.01) | 0.97 (0.93ŌĆō0.99) | |||
| ŌĆā | Appropriate | 706 (94.4) | 643 (96.3) | |||
| Need for evaluationc) | 42 (5.6) | 25 (3.7) | ||||
| Communication | 0.37 | 0.71 (0.31ŌĆō1.63) | 0.41 (0.14ŌĆō1.24) | |||
| Appropriate | 741 (99.6) | 674 (99.9) | ||||
| Need for follow-up | 3 (0.4) | 1 (0.2) | ||||
| Gross motor | 0.56 | 0.70 (0.30ŌĆō1.61) | 0.56 (0.20ŌĆō1.58) | |||
| Appropriate | 730 (98.1) | 665 (98.5) | ||||
| Need for follow-up | 14 (1.9) | 10 (1.5) | ||||
| Fine motor | 0.92 | 1.01 (0.48ŌĆō2.10) | 0.77 (0.26ŌĆō1.56) | |||
| Appropriate | 728 (97.9) | 661 (97.9) | ||||
| Need for follow-up | 16 (2.2) | 14 (2.1) | ||||
| Problem-solving | 0.79 | 0.88 (0.42ŌĆō1.82) | 0.58 (0.21ŌĆō1.56) | |||
| Appropriate | 730 (97.7) | 661 (97.9) | ||||
| Need for follow-up | 17 (2.3) | 14 (2.1) | ||||
| Personal/social | 0.53 | 0.68 (0.33ŌĆō1.39) | 0.57 (0.21ŌĆō1.51) | |||
| Appropriate | 729 (97.6) | 662 (98.1) | ||||
| Need for follow-up | 18 (2.4) | 13 (1.9) | ||||
References
1. Oledzka MM, Sweeney JK, Evans-Rogers DL, Coulter C, Kaplan SL. Experiences of parents of infants diagnosed with mild or severe grades of congenital muscular torticollis. Pediatr Phys Ther 2020;32:322ŌĆō9.


2. Carenzio G, Carlisi E, Morani I, Tinelli C, Barak M, Bejor M, et al. Early rehabilitation treatment in newborns with congenital muscular torticollis. Eur J Phys Rehabil Med 2015;51:539ŌĆō45.

4. Do TT. Congenital muscular torticollis: current concepts and review of treatment. Curr Opin Pediatr 2006;18:26ŌĆō9.


5. Wei JL, Schwartz KM, Weaver AL, Orvidas LJ. Pseudotumor of infancy and congenital muscular torticollis: 170 cases. Laryngoscope 2001;111:688ŌĆō95.


6. Hussein MA, Yun IS, Lee DW, Park H, Oock KY. Cervical spine dysmorphism in congenital muscular torticollis. J Craniofac Surg 2018;29:925ŌĆō9.


7. Cheng J, Tang S, Chen T, Wong M, Wong E. The clinical presentation and outcome of treatment of congenital muscular torticollis in infantsŌĆöa study of 1,086 cases. J Pediatr Surg 2000;35:1091ŌĆō6.


8. Minihane KP, Grayhack JJ, Simmons TD, Seshadri R, Wysocki RW, Sarwark JF. Developmental dysplasia of the hip in infants with congenital muscular torticollis. Am J Orthop (Belle Mead NJ) 2008;37:E155ŌĆō8.

9. Schertz M, Zuk L, Zin S, Nadam L, Schwartz D, Bienkowski RS. Motor and cognitive development at one-year follow-up in infants with torticollis. Early Hum Dev 2008;84:9ŌĆō14.


10. Xiao Y, Chi Z, Yuan F, Zhu D, Ouyang X, Xu W, et al. Effectiveness and safety of massage in the treatment of the congenital muscular torticollis: a systematic review and meta-analysis protocol. Medicine (Baltimore) 2020;99:e21879.



11. ├¢hman A, Nilsson S, Lagerkvist AL, Beckung E. Are infants with torticollis at risk of a delay in early motor milestones compared with a control group of healthy infants? Dev Med Child Neurol 2009;51:545ŌĆō50.


12. Schertz M, Zuk L, Green D. Long-term neurodevelopmental follow-up of children with congenital muscular torticollis. J Child Neurol 2013;28:1215ŌĆō21.



13. Zhou X, Du Q. High risk of motor development delay in children with congenital muscular torticollis. Ann Phys Rehabil Med 2018;61:e299.

14. Amaral DM, Cadilha RP, Rocha JAG, Silva AIG, Parada F. Congenital muscular torticollis: where are we today? A retrospective analysis at a tertiary hospital. Porto Biomed J 2019;4:e36.



15. Kaplan SL, Coulter C, Fetters L. Physical therapy management of congenital muscular torticollis: an evidence-based clinical practice guideline from the section on pediatrics of the American Physical Therapy Association. Pediatr Phys Ther 2013;25:348ŌĆō94.

16. Lee K, Chung E, Lee BH. A comparison of outcomes of asymmetry in infants with congenital muscular torticollis according to age upon starting treatment. J Phys Ther Sci 2017;29:543ŌĆō7.



17. Cheng JC, Wong MW, Tang SP, Chen TM, Shum SL, Wong EM. Clinical determinants of the outcome of manual stretching in the treatment of congenital muscular torticollis in infants: a prospective study of eight hundred and twenty-one cases. J Bone Joint Surg Am 2001;83:679ŌĆō87.


18. Moon JS. Review of National Health Screening Program for infant and children in Korea. J Korean Med Assoc 2010;53:377ŌĆō85.

19. Kim JH, Lee JE, Shim SM, Ha EK, Yon DK, Kim OH, et al. Cohort profile: National Investigation of Birth Cohort in Korea study 2008 (NICKs-2008). Clin Exp Pediatr 2021;64:480ŌĆō8.




20. Stellwagen L, Hubbard E, Chambers C, Jones KL. Torticollis, facial asymmetry and plagiocephaly in normal newborns. Arch Dis Child 2008;93:827ŌĆō31.


21. Hylton N. Infants with torticollis: the relationship between asymmetric head and neck positioning and postural development. Phys Occup Ther Pediatr 1997;17:91ŌĆō117.

22. Boere-Boonekamp MM, van der Linden-Kuiper LT. Positional preference: prevalence in infants and follow-up after two years. Pediatrics 2001;107:339ŌĆō43.



23. Ho B, Lee E, Singh K. Epidemiology, presentation and management of congenital muscular torticollis. Singapore Med J 1999;40:675ŌĆō9.

24. Davids J, Wenger D, Mubarak S. Congenital muscular torticollis: sequela of intrauterine or perinatal compartment syndrome. J Pediatr Orthop 1993;13:141ŌĆō7.

25. Hardgrib N, Rahbek O, M├Ėller-Madsen B, Maimburg R. Do obstetric risk factors truly influence the etiopathogenesis of congenital muscular torticollis? J Orthop Traumatol 2017;18:359ŌĆō64.




26. Chen HX, Tang SP, Gao FT, Xu JL, Jiang XP, Cao J, et al. Fibrosis, adipogenesis, and muscle atrophy in congenital muscular torticollis. Medicine (Baltimore) 2014;93:e138.



27. Lee SJ, Han JD, Lee HB, Hwang JH, Kim SY, Park MC, et al. Comparison of clinical severity of congenital muscular torticollis based on the method of child birth. Ann Rehabil Med 2011;35:641ŌĆō7.



28. Li D, Wang K, Zhang W, Wang J. Expression of Bax/Bcl-2, TGF-╬▓1, and type III collagen fiber in congenital muscular torticollis. Med Sci Monit 2018;24:7869ŌĆō74.



29. Hussein MA, Yun IS, Park H, Kim YO. Cervical spine deformity in longstanding, untreated congenital muscular torticollis. J Craniofac Surg 2017;28:46ŌĆō50.


30. Cheng JC, Au AW. Infantile torticollis: a review of 624 cases. J Pediatr Orthop 1994;14:802ŌĆō8.


31. ├¢hman A, Beckung E. Children who had congenital torticollis as infants are not at higher risk for a delay in motor development at preschool age. PM R 2013;5:850ŌĆō5.


32. Michel GF, Harkins DA. Postural and lateral asymmetries in the ontogeny of handedness during infancy. Dev Psychobiol 1986;19:247ŌĆō58.







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


