Article Contents
| Clin Exp Pediatr > Volume 69(3); 2026 |
|
Abstract
Background
We aimed to investigate differences in gut microbiota between patients with Hirschsprung disease (HSCR) and healthy children; assess longitudinal changes in the microbiota of patients with HSCR from diagnosis through postoperative period; and identify microbial markers predictive of postoperative HSCR-associated enterocolitis (HAEC).
Purpose
To investigate alterations in the gut microbiota of patients with HSCR by assessing longitudinal microbiome changes after surgery and identifying microbial signatures predictive of postoperative HAEC
Methods
A case-control study of 20 patients with HSCR and 20 controls was conducted at Maharaj Nakorn Chiang Mai Hospital. Fecal specimens were collected from patients with HSCR at initial diagnosis and from age-matched controls. Additional samples were obtained from patients intraoperatively and at 1 and 6 months postoperatively. A microbial analysis was performed using 16S rRNA gene sequencing (V3–V4 hypervariable regions).
Results
Compared to controls, patients with HSCR exhibited gut dysbiosis characterized by reduced microbial diversity and altered community composition as determined by Analysis of Compositions of Microbiomes with Bias Correction. Increased relative abundances of Robinsoniella, Fusobacterium, Cutibacterium, Citrobacter, and Eubacterium fissicatena were observed in patients with HSCR, whereas NK4A214, Lachnospiraceae XPB1014 groups, Acinetobacter and Acetitomaculum were decreased (q< 0.05). Alpha diversity in patients with HSCR was significantly increased at 6 months postoperatively versus at theinitial diagnosis (P<0.05). Longitudinal changes in Eubacterium and Eubacteriales suggest their potential use as markers of treatment efficacy. In patients who developed postoperative HAEC, Olsenella was enriched in the proximal intestine, whereas Holdemanella, Corynebacterium, Collinsella, and CAG-352 were elevated in the distal intestine (q<0.05).
Conclusion
Patients with HSCR exhibited distinct alterations in the gut microbiota, with significant shifts observed between the pretreatment period and 6 months postoperatively. Specific bacterial taxa were identified as potential markers for HAEC development. Future microbiome-targeted interventions to prevent HAEC need to be explored.
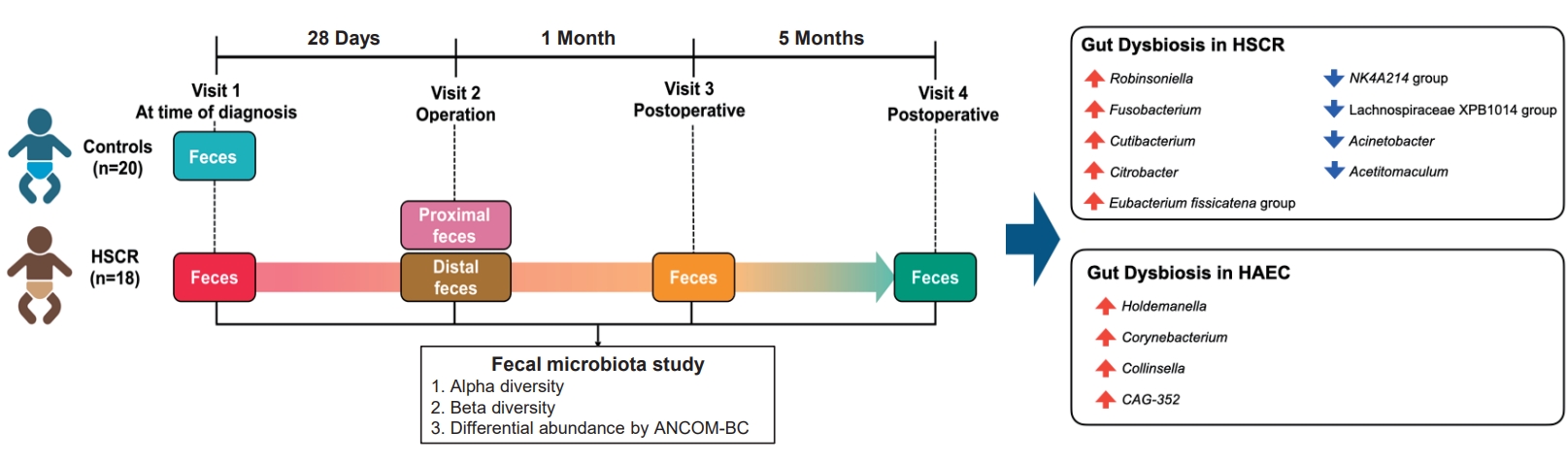
Graphical abstract. Visit 1, diagnosis; visit 2, during surgery from resected distal and proximal segments; visit 3, 1-month postsurgery; and visit 4, 6-month postsurgery; HSCR, Hirschsprung disease; HAEC, HSCR-associated enterocolitis; ANCOM-BC, Analysis of Compositions of Microbiomes with Bias Correction.
Hirschsprung disease (HSCR) is a congenital disorder characterized by the absence of ganglion cells in the distal intestine, leading to functional obstruction. Surgical removal of the aganglionic segment and restoration of bowel continuity to the anus are the primary treatments [1]. Despite surgical advancements, approximately one-third of patients experience postoperative complications [1]. Hirschsprung-associated enterocolitis (HAEC) remains a common and life-threatening complication in patients with HSCR, with a reported incidence of 25%–37% following definitive treatment [2]. Several studies have proposed multiple mechanisms predisposing to HAEC, including the disturbance of gut microbiota [1,3-7].
The gut microbiota plays a crucial role in maintaining intestinal homeostasis, and its composition has been linked to various pediatric gastrointestinal disorders, including inflammatory bowel disease (IBD) and short bowel syndrome [8]. However, detailed investigations of the gut microbiota in HSCR remain limited. Studies in animal models have demonstrated an increase in microbial diversity, with a predominance of the phyla Proteobacteria and Bacteroidetes [9,10]. In patients with HSCR, studies reported distinct differences in the gut microbiome between patients with and without HAEC [4,6,11]. Several studies in patients with HAEC found an increase in proteobacteria [6,11,12], especially Escherichia [12,13] and Enterobacteriaceae [11,14]. with a decrease in Bifidobacterium [12]. However, a detailed understanding of the gut microbiota in HSCR remains limited. Only a few studies have examined the longitudinal changes of gut microbiome in patients with HSCR before and after definitive surgery [15]. Therefore, this study aimed to: (1) compare gut microbiota profiles between children with HSCR and controls; (2) characterize longitudinal changes in the gut microbiota following definitive surgery; and (3) identify specific microbial features predictive of postoperative HAEC in high-risk patients.
Following these aims, this study was divided into 3 parts. First, we compared the fecal microbiota in 20 patients with 20 matched controls. Second, we characterized temporal alterations in the gut microbiota by analyzing samples collected at diagnosis, during surgery, 1-month, and 6-month postsurgery. Third, we collected intestinal content specimens from both the proximal and distal colon during surgery and then monitored clinical presentations of HAEC over 1 year to identify microbiome signatures predictive of postoperative HAEC.
This was a prospective, observational, matched case-control study and was approved by the Research Ethics Committee, Faculty of Medicine, Chiang Mai University (study code: SUR-2564-08226). The study included patients and matched controls aged 0–36 months who were treated at Maharaj Nakorn Chiang Mai Hospital, from August 25, 2021, to October 31, 2024. Informed consent was obtained from the parents or legal guardians.
As no prior studies had compared gut microbiota in untreated HSCR patients and normal children, this study was considered a pilot. We recruited 20 patients diagnosed with HSCR from a combination of clinical presentations, findings from contrast enema (particularly the presence of a transitional zone), and confirmation by histopathological examination of rectal biopsy specimens demonstrating absence of ganglion cells. Patients with concurrent congenital disorders, such as Down syndrome, and patients requiring an initial ostomy due to failed colonic surgery were excluded. Controlled participants were recruited by matching with similar ages (±20%), mode of delivery, and/or diets. Children with gastrointestinal infections or those who had taken probiotics, prebiotics, or antibiotics within 14 days were also excluded.
Postoperative HAEC was diagnosed based on definitions in previous studies [2,16,17], in patients presenting with at least one of the following: (1) explosive diarrhea or vomiting, (2) abdominal distension, (3) fever, or (4) leukocytosis. Diagnosis also required a Pastor’s HAEC score exceeding 4 and treatment with antibiotics [18]. Further details of the pre- and perioperative management are provided in Supplementary methods.
Fecal samples were collected from patients with HSCR at 4 timepoints: diagnosis (visit 1), during surgery from resected distal and proximal segments (visit 2), 1-month postsurgery (visit 3), and 6-month postsurgery (visit 4). Control samples were collected using a similar approach. Full protocols for specimen handling and preservation are provided in Supplementary methods and Supplementary Fig. 1A.
Bacterial genomic DNA was extracted and subjected to 16S rRNA gene sequencing targeting the V3–V4 region using the Illumina MiSeq platform. Sequence processing and microbial diversity analysis were performed using QIIME 2 (v2023.5), with taxonomic assignment based on the SILVA v138 database. Amplicon sequence variants were generated using the q2-dada2 plugin, and a phylogenetic tree was constructed using MAFFT. Differential abundance was analyzed using Analysis of Compositions of Microbiomes with Bias Correction (ANCOM-BC). Visualization was performed using the qiime2R package in R ver. 2023.06.1+524 (R Foundation for Statistical Computing, Austria).
Metabolic pathways were inferred using PICRUSt2 (v2.5.2) against the MetaCyc database. Only pathways with P value<0.05 were included in the results. Further details of the specimen handling and preservation, microbiota analysis, and metabolic pathway inference are provided in Supplementary methods.
Demographic and disease characteristics were collected and analyzed using Stata 16.0 (SAS Institute Inc., USA). Continuous variables were reported as mean±standard devatioan or median (interquartile range), categorical variables as frequencies and percentages. Fisher exact test and Student t test were used for group comparisons. Known HAEC confounders were adjusted using multivariable logistic regression. A complete case analysis was performed for missing data.
During the study period, 31 children presented with clinical symptoms suggestive of HSCR. After the performance of contrast studies and/or rectal biopsies, 20 patients were diagnosed with HSCR. One patient was excluded due to Down syndrome, and another due to incomplete sample extraction. Eleven children, suspected to have HSCR, presenting with constipation, and/or abdominal distension, experienced spontaneous resolution without requiring treatment were included in the control group, matched by age, mode of delivery, and/or diet. Additionally, 9 healthy children were enrolled as the controls, also matched by age, mode of delivery, and/or diet with the HSCR group. Details of all participants are shown in Supplementary Fig. 1B. The median age of the first specimen collection was 35.0 (range, 18.0–77.0) days for HSCR individuals and 34.5 (range, 24.5–69.0) days for controls. There were no statistically significant differences in other clinical characteristics between the 2 groups (Supplementary Table 1).
Microbiota analysis revealed distinct gut microbiome profiles between patients with HSCR and controls with and without HSCR-like symptoms. Taxonomic bar plots showing the relative abundance of gut microbial composition for each subject are presented in Fig. 1A. Observed features and Faith’s phylogenetic diversity indices revealed a significant reduction in alpha diversity in patients with HSCR compared to controls, both with and without HSCR-like symptoms. Beta-diversity indices (Bray-Curtis and Jaccard) showed significant differences in composition of the gut microbiota between HSCR individuals and controls. ANCOM-BC analysis identified differentially abundant bacterial taxa between the 2 groups. The results revealed an increase in the Robinsoniella, Fusobacterium, Cutibacterium, Citrobacter, and Eubacterium fissicatena group, while there were reductions in the NK4A214 group, Lachnospiraceae XPB1014 group, Acinetobacter, and Acetitomaculum. These results are illustrated in Figs. 1B-F. However, in the controls, there were no significant differences in alpha diversity, beta diversity, and ANCOM-BC analysis between children with and without HSCR-like symptoms.
The majority of patients had aganglionosis confined to the rectosigmoid region, with one patient diagnosed with rectal aganglionosis and another with long-segment aganglionosis extending to the descending colon. Except for the patient with long-segment HSCR, all patients underwent transanal endorectal pull-through. The median age at surgery was 60.0 (range, 41.0–95.0) days.
Alpha diversity analysis (Fig. 2A and B) based on observed features revealed a significant decrease in patients with HSCR at diagnosis (visit 1) compared to controls, with no significant differences between subsequent time points. Faith’s phylogenetic diversity analysis revealed a significant decrease in alpha diversity in patients with HSCR at diagnosis (visit 1) and 1-month postsurgery (visit 3) compared to controls. A significant increase in alpha diversity was observed 6 months postsurgery (visit 4) compared to diagnosis (visit 1), though no differences were found between other time points.
Beta-diversity indices (Fig. 2C and D) revealed significant differences in the microbial composition between controls and across different time points in patients with HSCR. Analysis of beta diversity demonstrated that the gut microbial composition in patients with HSCR differed from that of the controls. Additionally, the intestinal bacterial composition in HSCR patients at 6-month postsurgery (visit 4) differed from that at diagnosis (visit 1), during surgery (visit 2), and 1-month postsurgery (visit 3). No noticeable difference in beta diversity was identified between the proximal (ganglionic) and distal (aganglionic) samples.
The differences in abundance from the ANCOM-BC analysis were significant in bacterial taxa when comparisons were made between samples from the HSCR patients taken at the first diagnosis (visit 1) and distal specimens at surgery (visit 2). At 6-month following surgery, a significant difference was noted in bacterial taxa compared with diagnosis (visit 1), distal specimens at surgery (visit 2), and 1 month after surgery. There were no significant differences in bacterial taxa between 1 month after surgery (visit 3) and diagnosis (visit 1) or distal specimens at surgery (visit 2). The details of the bacterial taxa are shown in Fig. 2E.
Predictive metabolic pathways were analyzed to explore the effect of surgery on gut microbial metabolites by comparing patients with HSCR at 6-month postsurgery (visit 4) with those at diagnosis (visit 1). Vitamin and metabolite biosynthesis pathways were increased at visit 4, while pathways related to amino acid and aromatic compound degradative, pathogenicity-related functions, and energy and membrane metabolism pathways were decreased (Fig. 3).
At the genus, order, and family levels, bacterial patterns showed similar trends. Notably, dynamic changes in Eubacterium and Eubacteriales were observed across time points (Fig. 4). There was an increase in Eubacterium and Eubacteriales at 6 months after surgery (visit 4) compared with diagnosis (visit 1), distal samples during surgery (visit 2), and 1 month after surgery (visit 3). At diagnosis (visit 1), distal samples during surgery (visit 2), and 1 month after surgery (visit 3), Eubacterium and Eubacteriales were decreased compared with controls.
These findings revealed progressive gut microbiota changes in patients with HSCR from diagnosis through 6 months postoperatively. By that time, microbial composition had shifted significantly, with increased alpha diversity compared to baseline. However, persistent differences from controls suggest either a gradual return to equilibrium or continued dysbiosis.
Five patients (27.8%) developed HAEC within 1 year after surgery. The patient demographics, disease characteristics, therapeutic details, and postoperative characteristics of patients with HSCR, with and without postoperative HAEC are summarized in Supplementary Table 2. Patients or disease characteristics did not differ significantly between the 2 groups. However, patients who developed postoperative HAEC were significantly younger at diagnosis and surgery than those who did not. Additionally, the weight at the time of definitive surgery was significantly lower in the HAEC group. Patients with postoperative HAEC experienced a significantly higher rate of postoperative obstruction than those without HAEC (odds ratio, 47.9; 95% confidence interval, 2.4–958.2; P=0.011). However, after adjustments using multivariable logistic regression analysis (Supplementary Table 3), no significant differences were found in known clinical risk factors for HAEC between the 2 groups. There were also no significant differences in biochemical characteristics at the time of surgery between patients with and without postoperative HAEC (Supplementary Table 4).
An analysis of the gut microbiota during surgery in patients with HSCR, with and without enterocolitis (visit 2) revealed a slight reduction in alpha diversity in the proximal ganglionic specimens, and a nonsignificant increase in alpha diversity in the distal aganglionic specimens of patients with enterocolitis. However, these differences were not statistically significant (Fig. 5A and C). Beta diversity analysis also indicated no significant difference in bacterial composition between patients with HSCR, with and without enterocolitis, in either the proximal or distal regions during surgery (visit 2) (Fig. 5B and D). In contrast, ANCOM-BC analysis revealed significant taxonomic differences between HSCR patients with and without enterocolitis. These differences were observed in both proximal and distal regions during surgery (visit 2). In the proximal region, patients with enterocolitis showed a significant increase in the number of the genus Olsenella and an overall enrichment of the Atopobiaceae family. In the distal region, ANCOM-BC analysis revealed a significant increase in Holdemanella, Corynebacterium (family Corynebacteriaceae), Collinsella (family Coriobacteriaceae), and CAG-352 (Fig. 5E).
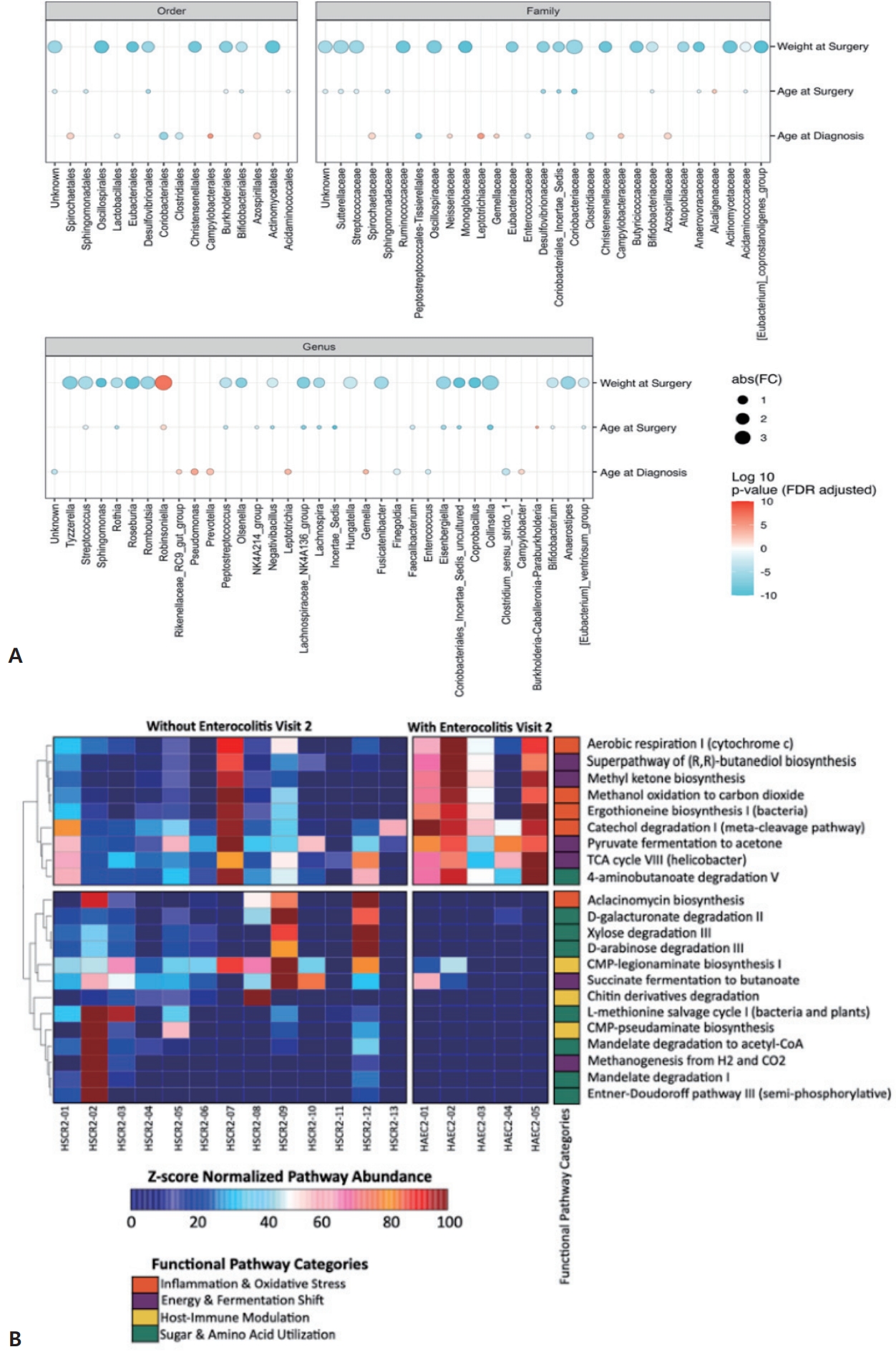
Comparison of predictive metabolic pathways between patients with enterocolitis revealed an increased abundance of pathways associated with inflammation, oxidative stress, and shifts in energy and fermentation metabolism. In contrast, pathways related to host immune modulation and sugar and amino acid utilization were reduced (Fig. 6A).
ANCOM-BC analysis (Fig. 6B) demonstrated correlations between clinical parameters and differentially abundant taxa in the distal colon of patients with enterocolitis during surgery (visit 2). Lower surgical weight was associated with decreased Robinsoniella and increased Sphingomonas, Roseburia, Lachnospiraceae NK4A136 group, and Coprobacillus. Younger age at surgery was associated with increased Incertae sedis and Collinsella. Similarly, a younger age at diagnosis was associated with decreased Pseudomonas, Leptotrichia, and Gemella.
HSCR is a functional intestinal obstruction in a distal aganglionic intestine. Consistent with previous animal and clinical studies, our findings confirm that HSCR is associated with gut dysbiosis, with significantly reduced alpha diversity and distinct beta diversity clustering when compared with age, delivery mode, and diet-matched controls [3,15,19]. While some animal models have reported increased microbial richness in obstructed segments due to delayed transit time and luminal stasis [10,20], our cohort showed compositional separation even against symptomatic children with HSCR-like constipation, supporting a disease-specific microbial signature beyond stasis alone. Although prior reports have described distinct microbiota profiles between proximal ganglionic and distal aganglionic segments [13,14], we did not observe marked regional beta diversity differences. This finding may be attributable to the preoperative bowel preparation protocol used at our center, which likely reduced the local microbial load and minimized segment-specific differences. Differential abundance analysis revealed enrichment of potentially pathogenic taxa, including Citrobacter and Fusobacterium, along with depletion of key butyrate-producing groups (NK4A214 group, Lachnospiraceae XPB1014 group) and the acetate producer Acetitomaculum. Notably, Citrobacter showed the highest log fold change among enriched taxa and is characterized as an enteric pathogenic bacterium [21]. This genus can produce glycoside hydrolases that break down the intestinal mucosal barrier, facilitating penetration by opportunistic bacteria and increasing the risk of local damage and infection [22]. Both clinical and preclinical studies have shown that increased abundance of Fusobacterium is linked to intestinal diseases, including IBDs, colitis, and colorectal cancer by promoting mucosal inflammation, releasing reactive oxygen species, and impairing the intestinal barrier [23,24]. Moreover, the depletion of butyrate and acetate-producing bacteria, well recognized for their anti-inflammatory and protective barrier roles, may further exacerbate intestinal pathology in HSCR by weakening mucosal integrity and amplifying local inflammatory responses [3,25].
Considering the changes in the gut microbiota of patients with HSCR from diagnosis to the postoperative period, we observed a significant decrease in alpha diversity at 1-month postsurgery compared to controls. This finding is consistent with a previous limited study comparing patients with HSCR and controls that revealed a decrease in microbiota richness in HSCR patients following surgery compared to controls [15]. Some studies have reported a reduction in the diversity of gut microbiota following rectal resection for rectal cancer, compared to the preoperative period and healthy adults [26]. This surgery is like the pull-through procedure in patients with HSCR, with a similar decrease in postoperative microbiota diversity. These findings suggest that surgery, particularly rectal resection, leads to significant alterations in bacterial communities. Additionally, 6 months after surgery, the gut microbiota differed from earlier time points, with an alpha diversity that had significantly increased compared to the pretreatment period, though it did not fully resemble the profiles of controls. This finding might indicate either a move towards equilibrium or persistent dysbiosis, as the gut microbiota at this stage still differed from that of the matched controls. These results are consistent with a previous report suggesting that persistent shift in gut microbiota after surgery may result from structural changes in the intestine, chronic inflammation and mucosal damage, the effects of perioperative antibiotics and other treatments, as well as dietary behaviors and environmental factors [15,27]. ANCOM-BC comparisons across time points revealed that significant differences in gut microbiota composition emerged only at 6 months postsurgery (visit 4), suggesting partial re-equilibration of the microbial community. This was characterized by enrichment of butyrate-producing genera, including Eubacterium within the order Eubacteriales, which gradually recovered by 6 months [28]. Prior studies have linked a reduction in butyrate-producing bacteria, such as Eubacterium, to increased intestinal inflammation and impaired gut barrier function in patients with IBD, as well as altered gut motility and stool consistency in children with functional constipation [29]. Therefore, the observed rebound in Eubacterium abundance may contribute to improved mucosal homeostasis and partial recovery of gut function in patients with HSCR. This is further supported by predictive functional analysis, which showed that at 6-month postsurgery (visit 4), there was a marked decrease in pathogenicity-related pathways, including enterobacterial toxin production, compared to diagnosis (visit 1) [11]. In parallel, pathways related to vitamin and metabolic biosynthesis increased, consistent with the recovery of butyrate-producing bacteria such as Eubacterium [25]. These results highlight that Eubacterium may act as a potential microbial biomarker to monitor postoperative gut re-equilibration and predict treatment success in HSCR.
HAEC is a serious complication. The incidence of postoperative HAEC in our study was 27.8%, aligned with previous reports (25%–37%) [2]. Several previous studies have proposed gut-microbial dysbiosis in patients with a history of HAEC and those predisposed to HAEC [3,13,14]. Nonetheless, the direct comparison of gut microbiota diversity across studies has been challenging due to variations in study design and specimen collection methods [3]. Focusing on studies with designs similar to ours, 2 studies investigated the microbiota of intestinal tissue during surgery and its association with the development of enterocolitis after surgery [5,11]. Due to differences in specimen collection and preparation, the results of these studies were discordant. Tang et al. [11] investigated fresh mucosal microbiota from the dilated colon in Chinese patients during surgery and discovered a reduction in alpha diversity among patients who later developed enterocolitis. In contrast, the study of Arbizu et al. [5] in American patients observed higher alpha diversity in aganglionic paraffin-fixed tissue at surgery in those who developed postoperative enterocolitis. In our study, which analyzed fecal gut microbiota during surgery, we observed a nonsignificant decrease in alpha diversity in the proximal ganglionic specimens and a nonsignificant increase in distal aganglionic specimens from patients who developed postoperative enterocolitis. These diversity patterns were in close alignment with previous research [5,11]. However, our results did not reach statistical significance. Beta diversity analysis revealed no significant differences in bacterial composition between the proximal and distal regions during surgery in patients with and without enterocolitis. Variations in gut microbiota diversity outcomes could be due to differences in specimen collection and preoperative bowel preparation protocols at our center. However, ANCOM-BC analysis identified significant taxa differences within the proximal and distal regions during surgery in HSCR patients who later developed enterocolitis and those who did not. We observed a significant increase in Olsenella in proximal ganglionic specimens. Elevated levels of Olsenella have been associated with inflammatory gut diseases, such as IBD, due to overproduction of lactic acid in the intestine [30]. In the distal aganglionic region, ANCOM-BC analysis showed a significant rise in the abundance of Holdemanella, Corynebacterium (family Corynebacteriaceae), Collinsella (family Coriobacteriaceae), and CAG-352. Consistent with previous studies on gastrointestinal inflammatory disorders, Holdemanella has been frequently detected in higher abundance among patients with IBD compared to the healthy controls [31]. Likewise, an increase in Collinsella has been observed in patients with IBD [30]. Mechanistically, Collinsella has been shown to alter bile acid metabolism and produce hydrogen sulfide, which can reduce colonic transit and increase gut permeability-factors that promote local stasis and mucosal inflammation [32,33]. Together, these features suggest that enrichment of these taxa may contribute to barrier dysfunction and an increased risk of postoperative HAEC. Comparison of ANCOM-BC results revealed that enriched taxa differed between the distal aganglionic and proximal ganglionic regions, reflecting niche-specific microbial dynamics. The distal colon, lacking enteric ganglion cells, exhibits impaired peristalsis, whereas the proximal colon maintains normal innervation and motility. These region-specific differences likely represent preoperative ecological variations that could influence host-microbe interactions beyond the resected segment. However, due to the scarcity of comparative studies, further large-scale prospective research is needed to validate these findings and investigate their potential implications for future therapeutic strategies.
These ecological shifts in the distal region were further supported by correlation analyses, which demonstrated that enriched pathogenic taxa were significantly linked to clinical parameters predisposing patients to postoperative HAEC. We also detected a correlation between previously known risk factors of postoperative HAEC and the distribution of gut microbiota. Lower weight at the time of surgery was associated with an increased abundance of Sphingomonas, Roseburia, Lachnospiraceae NK4A136 group, and Coprobacillus, and a reduction in Robinsoniella. Previous studies have proposed a correlation between lower weight at the time of surgery and early development of postoperative HAEC [34]. A previous study revealed a high incidence of HAEC at a younger age. Supporting this, our analysis detected a reduction in the abundance of Pseudomonas, Leptotrichia, and Gemella in patients diagnosed at a younger age [4]. Furthermore, our study found that a younger age at the time of surgery was associated with an increase in Incertae Sedis and Collinsella. Interestingly, an elevation in Collinsella was significantly correlated with postoperative obstruction, potentially due to its production of hydrogen sulfide, which reduces colonic transit [33]. Some studies have proposed a significant rise in the incidence of postoperative HAEC among patients with postoperative obstruction, which was also observed in our results [7]. However, after adjustment using multivariable logistic regression analysis, no significant differences were found in clinical parameters previously known to be risk factors for HAEC between the 2 groups. These findings may have been limited by the small sample size.
Predictive analysis indicated that patients with HSCR with enterocolitis exhibited increased pathways associated with inflammation and oxidative stress, and microbial energy metabolism. This elevation is consistent with previous reports and supports the notion that the gut microbiota in HAEC may actively promote mucosal inflammation while simultaneously initiating a compensatory antioxidant response to preserve the integrity of the mucosal barrier [3,35].
This study has several limitations. First, the small sample size limited statistical power, rendering some results inconclusive and lacking statistical significance. Second, the analysis of gut microbiota changes was confined to a 6-month postoperative period. Although no consensus exists regarding an optimal follow-up duration, this point was chosen based on clinical experience, as most patients with HSCR typically regain normal bowel function by 6-month postsurgery. At this stage, we observed significant alterations in gut microbial composition, although differences from controls persisted. This may reflect either progression toward microbial equilibrium or ongoing dysbiosis. Therefore, extended longitudinal studies beyond 6 months are necessary to determine whether these changes represent restoration of microbial homeostasis or continued dysregulation. Finally, consistent with many gut microbiome studies, interpretation is constrained by variability in study designs and sample collections. Additionally, factors such as race, environment, and geography substantially influence microbial composition and diversity, potentially limiting the generalizability of our findings. Variations in diet, lifestyle, and genetic background—particularly in Southeast Asia, where microbiome data remain limited—may contribute to the distinct microbial signatures observed in our cohort. To mitigate this, our study focused on microbiota analysis at the time of surgery to identify predictive microbial markers of postoperative HAEC and guide future preventive strategies.
These findings support the potential utility of microbial biomarkers—particularly Eubacterium and Eubacteriales—in monitoring postoperative recovery and predicting HAEC. Although overall microbial diversity did not significantly differ between patients with and without postoperative HAEC, differential taxa in proximal and distal segments suggest region-specific microbial dysbiosis. Collectively, these results highlight the role of gut microbiota in HAEC pathogenesis and underscore the need for larger, longitudinal studies to guide microbiota-based preventive strategies.
Supplementary materials
Supplementary Methods, Supplementary Tables 1-4, and Supplementary Fig. 1 are available at https://doi.org/10.3345/cep.2025.01886.
Supplementary Fig. 1.
Overview of sample collection timeline and study design. (A) The time points of fecal collection in this study. (B) Flow-chart of this study. Visit 1, diagnosis; visit 2, during surgery from resected distal and proximal segments; visit 3, 1-month postsurgery; and visit 4, 6-month postsurgery; HSCR, Hirschsprung disease; HAEC, Hirschsprung-associated enterocolitis.
Supplementary Table 1.
Comparison baseline characteristics of HSCR patients and healthy children in this study (total N=38)
Supplementary Table 2.
The patient and disease characteristics, therapeutic characteristics, and postoperative characteristics of HSCR patients with and without postoperative HAEC
Supplementary Table 3.
Univariable and multivariable logistic regression analysis of clinical parameters associated with development of postoperative HAEC
Supplementary Table 4.
Biochemical characteristics of HSCR patients with and without postoperative HAEC after surgery
Footnotes
Fig. 1.
Alterations in the gut microbiota of controls versus patients with Hirschsprung disease (HSCR). This figure summarizes the gut microbiome study comparing the control group (n=20) and the HSCR group at the time of diagnosis (visit 1) (n=18). The results demonstrated distinct gut microbiome profiles in the 2 groups. (A) Taxonomic bar plots illustrating the relative abundance of the gut microbial composition for each subject. (B and C) Alpha-diversity indices indicating a significant change in the diversity of the gut microbiota in patients with HSCR versus healthy controls. *P<0.05, Kruskal-Wallis test with Dunn post hoc test. (D and E) Beta-diversity indices revealing significant differences in the composition of the gut microbiota between patients with HSCR and healthy controls. *P<0.05 versus controls, PERMANOVA, 999 permutations. (F) Analysis of Compositions of Microbiomes with Bias Correction identified specific taxa that were differentially abundant between patients with HSCR and healthy controls. Visit 1, diagnosis; visit 2, during surgery from resected distal and proximal segments; visit 3, 1-month postsurgery; and visit 4, 6-month postsurgery. PERMANOVA, permutational multivariate analysis of variance; ASV, amplicon sequence variant; PC, principal coordinate.

Fig. 2.
Alterations in the gut microbiota over time in patients with Hirschsprung disease (HSCR). Controls=20; HSCR at the time of diagnosis (visit 1)=18; HSCR at the time of surgery (visit 2) - proximal region =16; HSCR at the time of surgery (Visit 2) - distal region=18; HSCR at 1 month postoperative (visit 3)=18; HSCR at 6 months postoperative (visit 4)=18. (A and B) Changes in alpha diversity across the time points. The dots represent individual samples. *P<0.05 vs. controls. †P<0.05 vs. HSCR at the time of diagnosis (Visit 1), Kruskal-Wallis test with Dunn post hoc test. (C and D) Beta diversity indices revealing significant differences in the composition of the gut microbiota between controls and across different time points in patients with HSCR. Beta-diversity analysis showing that the gut microbial composition differed between patients with HSCR and controls. Additionally, the gut bacterial composition of HSCR at 6 months postoperative (visit 4) differed from that at the time of diagnosis (visit 1), at the time of surgery (visit 2) (proximal and distal), and at 1 month postoperative (visit 3). *P<0.05, controls versus patients with HSCR. **P<0.05, visit 4 versus visits 1–3, PERMANOVA, 999 permutations. (E) Analysis of Compositions of Microbiomes with Bias Correction analysis comparing the gut microbiome between visits. PERMANOVA, permutational multivariate analysis of variance; PD, phylogenic diversity; PC, principal Coordinate.
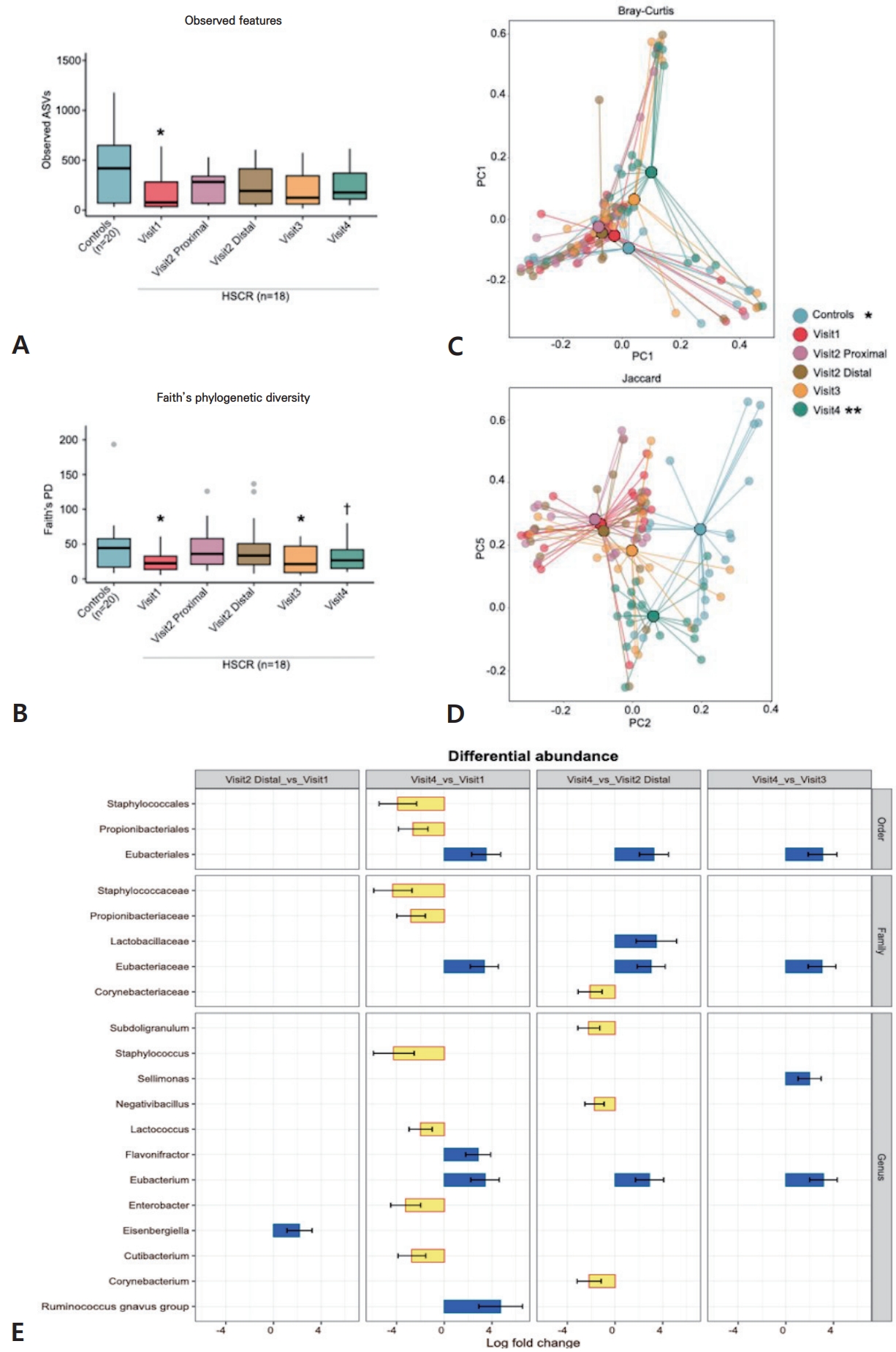
Fig. 3.
Predicted metabolic pathways analyzed by PICRUSt2 comparing Hirschsprung disease (HSCR) visit 4 and HSCR visit 1 indicating potential postoperative microbiome shifts. Visit 1, diagnosis; visit 4, 6-month postsurgery.

Fig. 4.
Dynamics of Eubacterium and Eubacteriales across the time points. A similar pattern is evident at the genus and order levels with an increase in sequencing of both Eubacterium and Eubacteriales in the Hirschsprung disease (HSCR) at 6 months postoperative (visit 4). *P<0.05 versus controls #P<0.05 versus HSCR at the time of diagnosis (visit 1) †P<0.05 versus HSCR at the time of surgery (visit 2). ‡P<0.05 versus HSCR at 1 month postoperative (visit 3). Kruskal-Wallis test with Dunn post hoc test.

Fig. 5.
Comparison of gut microbiota between patients with Hirschsprung disease (HSCR) and enterocolitis (n=5) versus HSCR without enterocolitis (n=13) at the time of surgery (visit 2). (A) Alpha-diversity indices indicating no significant change in gut microbiota diversity in the proximal region at the time of surgery (visit 2) between patients with HSCR with versus without enterocolitis. ns, Kruskal-Wallis test with Dunn post hoc test. (B) Beta-diversity analysis indicating no significant differences in bacterial composition in the proximal region at the time of surgery (visit 2) between patients with HSCR with versus without enterocolitis. ns, PERMANOVA, 999 permutations. (C) Alpha-diversity indices indicating no significant change in the gut microbiota diversity in the distal region at the time of surgery (visit 2) between patients with HSCR with versus without enterocolitis. ns, Kruskal-Wallis test with Dunn post hoc test. (D) Beta-diversity analysis indicating no significant difference in bacterial composition in the distal region at the time of surgery (visit 2) between patients with HSCR with versus without enterocolitis. ns, PERMANOVA, 999 permutations. (E) Results of Analysis of Compositions of Microbiomes with Bias Correction analysis showing the differential taxa in patients with HSCR with versus without enterocolitis. Significant differential taxa are evident in the proximal region at the time of surgery (visit 2) of patients with HSCR with versus without enterocolitis as well as the distal region at the time of surgery (visit 2) of patients with HSCR with versus without enterocolitis. Visit 2, during surgery from resected distal and proximal segments; PERMANOVA, permutational multivariate analysis of variance; ASV, amplicon sequence variant; PD, phylogenic diversity; PC, principal coordinate.
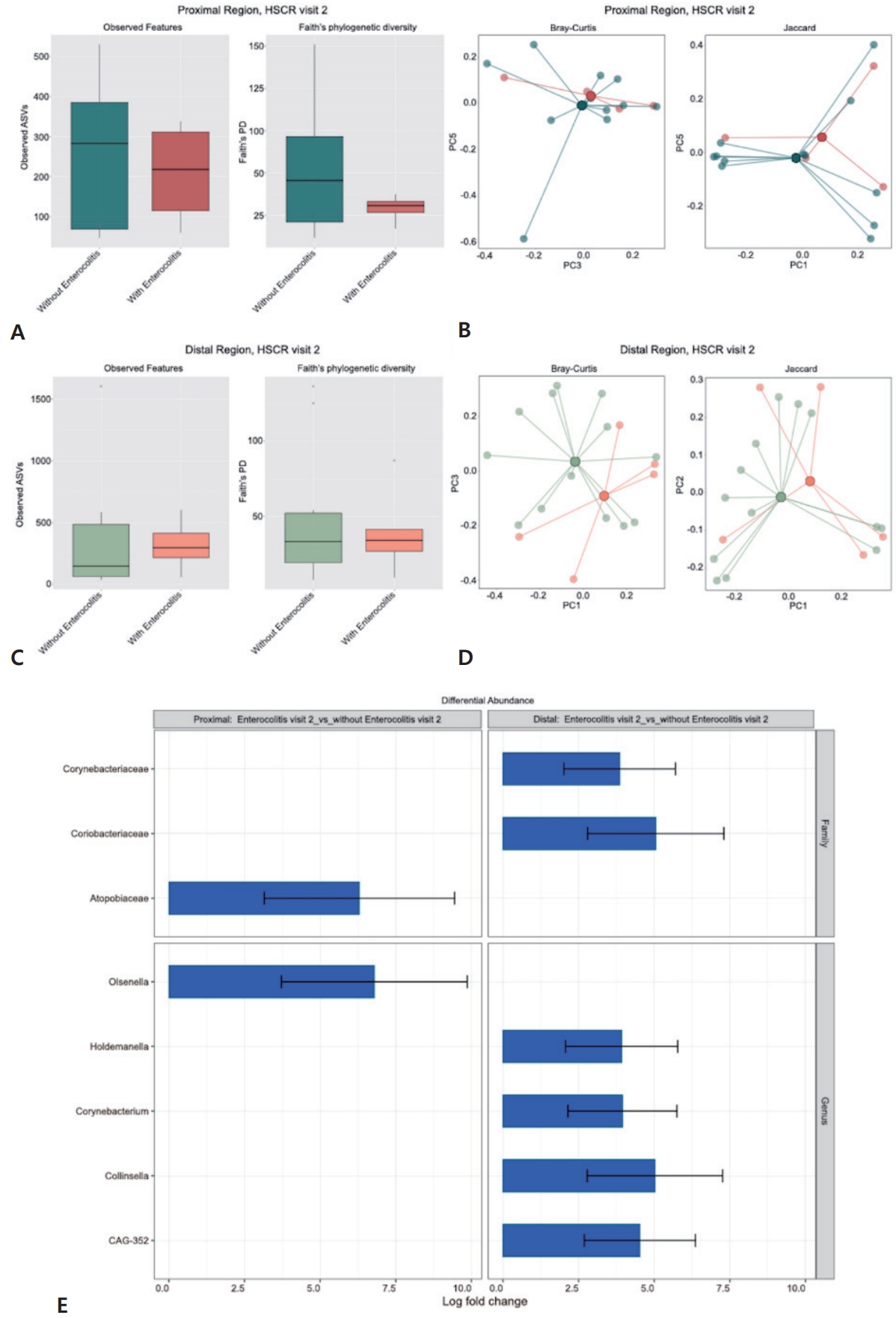
Fig. 6.
Associations between clinical parameters, microbial taxa, and predicted metabolic pathways in the distal colon of a patient with Hirschsprung disease (HSCR) and enterocolitis at the time of surgery. (A) Correlation between clinical parameters and differential abundance of taxa in the distal colon region of a patient with HSCR and enterocolitis at the time of surgery (visit 2). Analysis of Compositions of Microbiomes with Bias Correction analysis revealing that a lower weight at surgery was associated with a reduction in Robinsoniella and an increase in Sphingomonas, Roseburia, Lachnospiraceae NK4A136 group, and Coprobacillus. A younger age at surgery was correlated with increased Incertae sedis and Collinsella, while a younger age at diagnosis was linked to reduced Pseudomonas, Leptotrichia, and Gemella species. (B) Predicted metabolic pathways (PICRUSt2 analysis) comparing patients with HSCR with versus without enterocolitis at the time of surgery suggesting potential microbial functional shifts following the surgical intervention. Visit 2, during surgery from resected distal and proximal segments; FC, fold change; FDR, false discovery rate.
References
1. Gosain A. Established and emerging concepts in Hirschsprung's-associated enterocolitis. Pediatr Surg Int 2016;32:313-20.




2. Gosain A, Frykman PK, Cowles RA, Horton J, Levitt M, Rothstein DH, et al. Guidelines for the diagnosis and management of Hirschsprung-associated enterocolitis. Pediatr Surg Int 2017;33:517-21.




3. Chantakhow S, Khorana J, Tepmalai K, Boonchooduang N, Chattipakorn N, Chattipakorn SC. Alterations of gut bacteria in Hirschsprung disease and Hirschsprung-associated enterocolitis. Microorganisms 2021;9:2241.



4. Arnaud AP, Cousin I, Schmitt F, Petit T, Parmentier B, Levard G, et al. Different fecal microbiota in Hirschsprung's patients with and without associated enterocolitis. Front Microbiol 2022;13:904758.



5. Arbizu RA, Collins D, Wilson RC, Alekseyenko AV. Evidence for differentiation of colon tissue microbiota in patients with and without postoperative Hirschsprung's associated enterocolitis: a pilot study. Pediatr Gastroenterol Hepatol Nutr 2021;24:30-7.




6. Biassoni R, Di Marco E, Squillario M, Ugolotti E, Mosconi M, Faticato MG, et al. Pathways and microbiome modifications related to surgery and enterocolitis in Hirschsprung disease. Pediatr Surg Int 2022;38:83-98.



7. Jiao CL, Chen XY, Feng JX. Novel insights into the pathogenesis of Hirschsprung's-associated enterocolitis. Chin Med J (Engl) 2016;129:1491-7.



8. Till H, Castellani C, Moissl-Eichinger C, Gorkiewicz G, Singer G. Disruptions of the intestinal microbiome in necrotizing enterocolitis, short bowel syndrome, and Hirschsprung's associated enterocolitis. Front Microbiol 2015;6:1154.



9. Touré AM, Landry M, Souchkova O, Kembel SW, Pilon N. Gut microbiota-mediated gene-environment interaction in the TashT mouse model of Hirschsprung disease. Sci Rep 2019;9:492.


10. Pierre JF, Barlow-Anacker AJ, Erickson CS, Heneghan AF, Leverson GE, Dowd SE, et al. Intestinal dysbiosis and bacterial enteroinvasion in a murine model of Hirschsprung's disease. J Pediatr Surg 2014;49:1242-51.



11. Tang W, Su Y, Yuan C, Zhang Y, Zhou L, Peng L, et al. Prospective study reveals a microbiome signature that predicts the occurrence of post-operative enterocolitis in Hirschsprung disease (HSCR) patients. Gut Microbes 2020;11:842-54.



12. Singer G, Kashofer K, Castellani C, Till H. Hirschsprung's associated enterocolitis (HAEC) personalized treatment with probiotics based on gene sequencing analysis of the fecal microbiome. Case Rep Pediatr 2018;2018:3292309.




13. Li Y, Poroyko V, Yan Z, Pan L, Feng Y, Zhao P, et al. Characterization of intestinal microbiomes of Hirschsprung's disease patients with or without enterocolitis using Illumina-MiSeq high-throughput sequencing. PLoS One 2016;11:e0162079.



14. Yan Z, Poroyko V, Gu S, Zhang Z, Pan L, Wang J, et al. Characterization of the intestinal microbiome of Hirschsprung's disease with and without enterocolitis. Biochem Biophys Res Commun 2014;445:269-74.


15. Neuvonen MI, Korpela K, Kyrklund K, Salonen A, de Vos W, Rintala RJ, et al. Intestinal microbiota in Hirschsprung disease. J Pediatr Gastroenterol Nutr 2018;67:594-600.


16. Chung PHY, Yu MON, Wong KKY, Tam PKH. Risk factors for the development of post-operative enterocolitis in short segment Hirschsprung's disease. Pediatr Surg Int 2019;35:187-91.



17. Chantakhow S, Tepmalai K, Singhavejsakul J, Tantraworasin A, Khorana J. Prognostic factors of postoperative Hirschsprung-associated enterocolitis: a cohort study. Pediatr Surg Int 2023;39:77.



18. Frykman PK, Kim S, Wester T, Nordenskjöld A, Kawaguchi A, Hui TT, et al. Critical evaluation of the Hirschsprung-associated enterocolitis (HAEC) score: a multicenter study of 116 children with Hirschsprung disease. J Pediatr Surg 2018;53:708-17.



19. Cheng Z, Zhao L, Dhall D, Ruegger PM, Borneman J, Frykman PK. Bacterial microbiome dynamics in post pullthrough Hirschsprung-associated enterocolitis (HAEC): an experimental study employing the endothelin receptor B-null mouse model. Front Surg 2018;5:30.



20. Vandeputte D, Falony G, Vieira-Silva S, Tito RY, Joossens M, Raes J. Stool consistency is strongly associated with gut microbiota richness and composition, enterotypes and bacterial growth rates. Gut 2016;65:57-62.



21. Yuan C, Yin Z, Wang J, Qian C, Wei Y, Zhang S, et al. Comparative genomic analysis of Citrobacter and key genes essential for the pathogenicity of Citrobacter koseri. Front Microbiol 2019;10:2774.



22. Chen Y, Gilliland A, Liang Q, Han X, Yang H, Chan J, et al. Defining enteric bacterial pathogenesis using organoids: Citrobacter rodentium uses EspC, an atypical mucinolytic protease, to penetrate mouse colonic mucus. Gut Microbes 2025;17:2494717.



23. Weng YJ, Gan HY, Li X, Huang Y, Li ZC, Deng HM, et al. Correlation of diet, microbiota and metabolite networks in inflammatory bowel disease. J Dig Dis 2019;20:447-59.



24. Jeepipalli S, C A, Duncan W, Krishna VM, Sahay B, Chan EKL, et al. Exploring the complex interplay between oral Fusobacterium nucleatum infection, periodontitis, and robust microRNA induction, including multiple known oncogenic miRNAs. mSystems 2025;10:e0173224.




25. Demehri FR, Frykman PK, Cheng Z, Ruan C, Wester T, Nordenskjöld A, et al. Altered fecal short chain fatty acid composition in children with a history of Hirschsprung-associated enterocolitis. J Pediatr Surg 2016;51:81-6.



26. Cong J, Zhu H, Liu D, Li T, Zhang C, Zhu J, et al. A pilot study: changes of gut microbiota in post-surgery colorectal cancer patients. Front Microbiol 2018;9:2777.



27. Plekhova V, De Paepe E, Van Renterghem K, Van Winckel M, Hemeryck LY, Vanhaecke L. Disparities in the gut metabolome of post-operative Hirschsprung's disease patients. Sci Rep 2021;11:16167.




28. Cohen J, Opal SM, Powderly WG, editors. Infectious diseases. 3rd ed. London: Mosby; 2010. Chapter 173, Anaerobic bacteria; p. 1757-76.
29. Carding S, Verbeke K, Vipond DT, Corfe BM, Owen LJ. Dysbiosis of the gut microbiota in disease. Microb Ecol Health Dis 2015;26:26191.



30. Teofani A, Marafini I, Laudisi F, Pietrucci D, Salvatori S, Unida V, et al. Intestinal taxa abundance and diversity in inflammatory bowel disease patients: an analysis including covariates and confounders. Nutrients 2022;14:260.



31. Wang H, Wang Y, Yang L, Feng J, Tian S, Chen L, et al. Integrated 16S rRNA sequencing and metagenomics insights into microbial dysbiosis and distinct virulence factors in inflammatory bowel disease. Front Microbiol 2024;15:1375804.



32. Dordević D, Jančíková S, Vítězová M, Kushkevych I. Hydrogen sulfide toxicity in the gut environment: meta-analysis of sulfate-reducing and lactic acid bacteria in inflammatory processes. J Adv Res 2021;27:55-69.



33. Scaldaferri F, Nardone O, Lopetuso LR, Petito V, Bibbò S, Laterza L, et al. Intestinal gas production and gastrointestinal symptoms: from pathogenesis to clinical implication. Eur Rev Med Pharmacol Sci 2013;17 Suppl 2:2-10.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


