Article Contents
| Clin Exp Pediatr > Volume 69(3); 2026 |
|
Abstract
Background
Purpose
Methods
Results
Footnotes
Fig. 1.
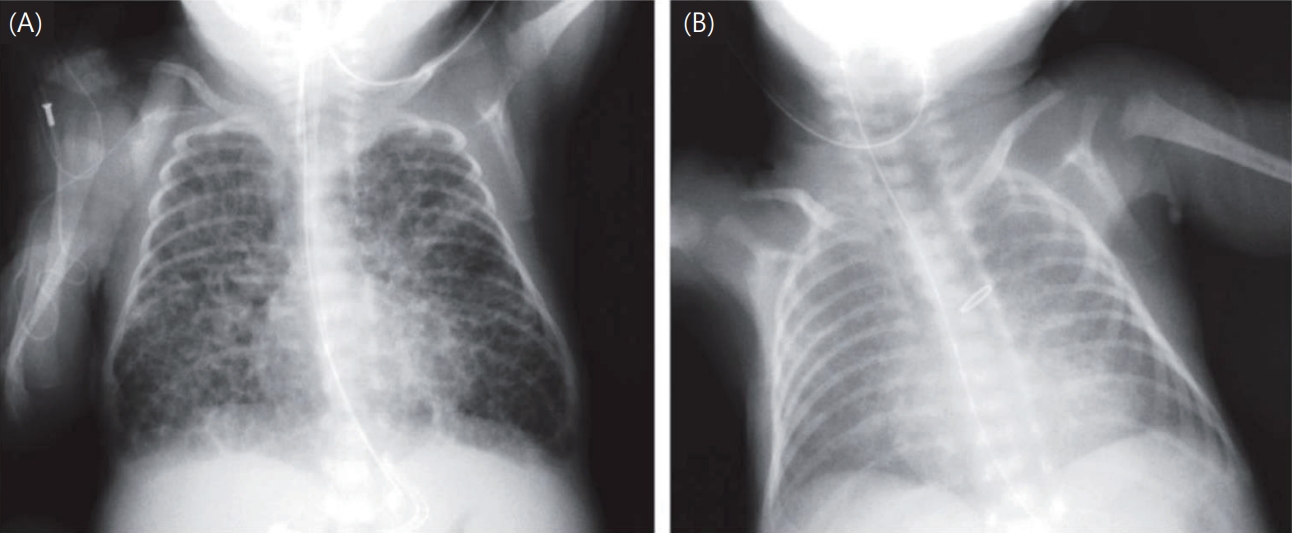
Table 1.
| Type (a) | Histological CAM (c) | Bubbly/cystic appearance on chest x-ray (d) |
|---|---|---|
| I (s) (b) | − | + |
| II (s) (b) | − | − |
| III (s) (b) | + | + |
| IV (s) (b) | + | − |
| V | Unclassifiable (e) | |
BPD, bronchopulmonary dysplasia; MHLW, Ministry of Health, Labour and Welfare (Japan); CAM, chorioamnionitis; SGA, small for gestational age.
(a) BPD is classified into 5 categories based on 3 criteria: histological CAM, bubbly/cystic appearance on frontal chest x-ray, and SGA status. The BPD type is presented. (b) SGA is defined as a birth weight below the 10th percentile, and “s” is included alongside the BPD type in cases of SGA (BPD types without SGA are designated I, II, III, and IV, while those with SGA are designated Is, IIs, IIIs, and IVs). (c) CAM should be diagnosed histologically, notwithstanding its stage. (d) X-ray changes are evaluated within the first 28 days of life, and the lungs are split into 4 areas, first at the level of the right and left lungs and then at the level of the upper and lower sections within each lung. A diffuse bubbly/cystic appearance is characterized by the presence of bubbles or cysts with a diameter of 1.0–10.0 mm in 3 of 4 areas. (e) Placental histology is advised; however, if histological findings are unknown, the case is classified as type V regardless of the chest x-ray results.
Table 2.
Values are presented as mean±standard deviation or number (%).
BPD, bronchopulmonary dysplasia; PDA, patent ductus arteriosus; RDS, respiratory distress syndrome; CAM, chorioamnionitis.
P values indicate differences among groups. Boldface indicates a statistically significant difference with P<0.05.
Table 3.
Table 4.
| Factor | Adjusted OR | 95% CI | P value |
|---|---|---|---|
| SGA | 3.32 | 1.16–9.48 | 0.032 |
| Chest x-ray bubbly/cystic findings | 10.88 | 4.43–26.72 | <0.01 |
| Placental pathology | 1.45 | 0.72–2.94 | 0.30 |
A multivariate logistic regression analysis was conducted to adjust for potential confounding variables (gestational age, birth weight, sex, Apgar score at 5 min, antenatal steroid use, RDS, and PDA).
BPD, bronchopulmonary dysplasia; PMA, postmenstrual age; OR, odds ratio; CI, confidence interval; RDS, respiratory distress syndrome; PDA, patent ductus arteriosus; SGA, small for gestational age.
Boldface indicates a statistically significant difference with P<0.05.
Table 5.
| BPD subtype | Severe BPD at 36-wk PMA | OR (unadjusted) | 95% CI | P value | OR (adjusted)a) | 95% CI | P value |
|---|---|---|---|---|---|---|---|
| I (n=29; CAM [-], bubbly/cystic chest x-ray [+]) | 22 (75.9) | 5.41 | 1.93–15.16 | <0.01 | 6.21 | 1.93–14.36 | <0.01 |
| II (n=49; CAM [-], bubbly/cystic chest x-ray [-]) | 18 (36.7) | 1.0 (ref) | - | - | 1.0 (ref) | - | - |
| III (n=14; CAM [+], bubbly/cystic chest x-ray [+]) | 14 (87.5) | 12.06 | 2.46–59.20 | <0.01 | 15.32 | 2.48–46.32 | <0.01 |
| IV (n=20; CAM [+], bubbly/cystic chest x-ray [-]) | 8 (40.0) | 1.15 | 0.40–3.34 | 0.80 | 1.30 | 0.36–4.69 | 0.69 |
| V (n=31; unclassifiable) | 18 (58.1) | 2.39 | 0.95–5.98 | 0.06 | 2.63 | 0.86–6.32 | 0.09 |
Values are presented as number (%) of infants with severe BPD at 36 weeks' PMA.
BPD, bronchopulmonary dysplasia; PMA, postmenstrual age; OR, odds ratio; CI, confidence interval; CAM, chorioamnionitis; PDA, patent ductus arteriosus; RDS, respiratory distress syndrome.
Table 6.
| BPD subtype | Severe BPD at 36-wk PMA | OR (unadjusted) | 95% CI | P value | OR (adjusted)a) | 95% CI | P value |
|---|---|---|---|---|---|---|---|
| I (n=24; CAM [-], bubbly/cystic chest x-ray [+]) | 16 (66.7) | 3.01 | 1.03 – 9.24 | 0.042 | 4.88 | 1.23 – 19.3 | 0.026 |
| II (n=33; CAM [-], bubbly/cystic chest x-ray [-]) | 13 (39.4) | 1.0 (ref) | - | - | 1.0 (ref) | - | - |
| III (n=14; CAM [+], bubbly/cystic chest x-ray [+]) | 13 (92.9) | 21.54 | 2.52–184.01 | <0.01 | 20.8 | 1.80–165.32 | 0.018 |
| IV (n=16; CAM [+], bubbly/cystic chest x-ray [-]) | 8 (50.0) | 1.54 | 0.46–5.33 | 0.48 | 1.48 | 0.38–5.76 | 0.57 |
| V (n=23; unclassifiable) | 14 (60.9) | 2.39 | 0.80–7.81 | 0.10 | 3.22 | 0.89–11.7 | 0.07 |






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


