< Previous Next >
Article Contents
| Korean J Pediatr > Volume 58(3); 2015 |
|
Abstract
Hand, foot, and mouth disease (HFMD) is an acute, mostly self-limiting infection. Patients usually recover without any sequelae. However, a few cases are life threatening, especially those caused by enterovirus 71 (EV71). A 12-month-old boy was admitted to a primary hospital with high fever and vesicular lesions of the mouth, hands, and feet. After 3 days, he experienced 3 seizure episodes and was referred to our hospital. On admission, he was conscious and his chest radiograph was normal. However, 6 hours later, he suddenly lost consciousness and had developed a massive pulmonary hemorrhage that continued until his death. He experienced several more intermittent seizures, and diffuse infiltration of both lung fields was observed on chest radiography. Intravenous immunoglobulin, dexamethasone, cefotaxime, leukocyte-depleted red blood cells, fresh frozen plasma, inotropics, vitamin K, and endotracheal epinephrine were administered. The patient died 9 hours after intubation, within 3 days from fever onset. EV71 subgenotype C4a was isolated retrospectively from serum and nasopharyngeal swab by real-time reverse transcription-polymerase chain reaction. Here, we report a fatal case of EV71-associated HFMD with sudden-onset massive pulmonary hemorrhage and suspected encephalitis.
Hand, foot, and mouth disease (HFMD) is a common, acute, and mostly self-limiting enteroviral infection that presents with fever and vesicular lesions on the hands, feet, mouth, and frequently buttocks as characteristic features1,2,3,4). Although patients with HFMD recover without any sequelae within about 1 week, severe complications occur in a few cases, some of which are life threatening. Severe complications accompanying HFMD include encephalitis, pulmonary edema/hemorrhage, cardiopulmonary collapse, and myocarditis1,2).
HFMD is most frequently caused by coxsackievirus A16 and enterovirus 71. Enterovirus 71 can often lead to a more severe clinical course accompanied by neurologic and cardiopulmonary complications and may rapidly lead to death1,2,3).
Enterovirus 71-induced HFMD developed in Korea in 2000 but it was not an epidemic and fatal until 20081,4,5). In the spring of 2009, a large nationwide outbreak of HFMD caused by enterovirus 71 occurred in Korea, and a few cases with encephalitis were fatal1,4,6). A multicenter study in Korea performed by the Enterovirus Complication Working Group revealed that encephalitis with cardiopulmonary complications of HFMD occurred in 2.4% of patients, including 2 patients (1.2%) who expired1). Pulmonary hemorrhage in enterovirus 71-infected HFMD patients has been reported in Taiwan, China, and Malaysia2,7,8,9). However, to our knowledge, no cases of HFMD with sudden-onset massive pulmonary hemorrhage have been reported in Korea. Here, we report a fatal case of enterovirus 71-induced HFMD that progressed rapidly with massive pulmonary hemorrhage and convulsion.
A 12-month-old, previously healthy boy with HFMD was referred to Inje University Ilsan Paik hospital because of 3 episodes febrile convulsions. He was born at full term weighing 3.87 kg through normal vaginal delivery by a Vietnamese mother. He was admitted to a primary hospital with high fever (up to 39℃), throat vesicles, and vesicular rashes on his hands and feet for 2 days. On the second hospital day, he had 3 generalized tonic-clonic and atonic convulsions with vertical eyeball deviation within 5 minutes of each other. He was consequently referred to our hospital.
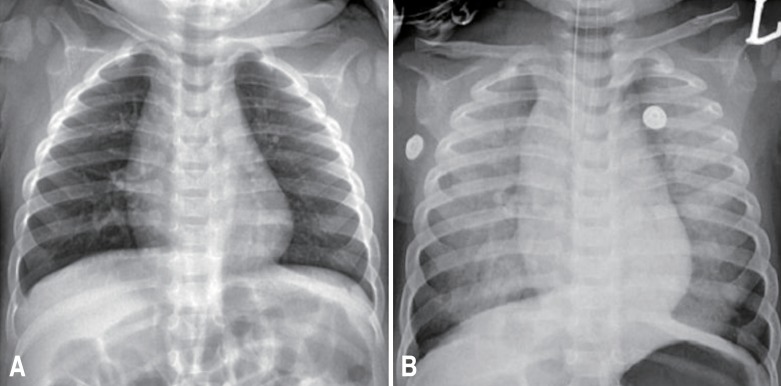
On admission, he appeared acutely ill and irritable. However, he was conscious and his vital signs were unremarkable. His chest radiography was normal. Within 5 hours after admission, he vomited 3 times after meals. Besides a body temperature of 38.3℃, his other vital signs were stable and he remained conscious. Desaturation to 76% percutaneous blood oxygen saturation (SpO2) and tachycardia to 176 beats/min accompanied by perioral cyanosis occurred suddenly 6 hours after admission. He moaned and exhibited slight subcostal retraction. His respiration rate was 34 breaths/min, and auscultation disclosed rales on the whole lung field. His consciousness decreased towards a coma state and bloody vomiting subsequently occurred. An endotracheal tube was inserted immediately, and a large amount of blood from a pulmonary hemorrhage came out through it. However, there were just small amounts of blood clots in the Levin tube. His convulsions subsequently restarted. Chest radiography immediately after desaturation revealed diffuse infiltrations on both lung fields (Fig. 1). Ventilator care was initiated in the intensive care unit.
Comparison of laboratory test results between his initial and pulmonary hemorrhagic status revealed that his hemoglobin decreased from 9.6 to 8.3 mg/dL and his white blood cell count increased from 13,090 to 17,450 cells/µL. However, platelet count was not decreased from 325,000 to 382,000 cells/µL. Blood glucose levels were increased from 125 to 465 mg/dL. Erythrocyte sedimentation rate was slightly increased from 15 to 32 mm/hr, but C-reactive protein level decreased from 1.06 to 0.2 mg/dL. Prothrombin time (PT) and PT international normalized ratio were slightly prolonged (16.5 and 1.39 seconds, respectively). Meanwhile, D-dimer (2,375.91 ng/mL) and fibrin degradation product (5-20 µg/mL) were elevated. Eight hours after admission, his blood pressure decreased to 82/41 mmHg and heart rate increased to 220 beats/min. His body temperature ranged from 38.8℃ to 40.2℃. The patient was diagnosed as fatal HFMD with massive pulmonary hemorrhage. Enterovirus 71 infection was suspected due to severe complications. Encephalitis was also suspected with seizure and loss of consciousness.
Intravenous immunoglobulin (IVIG, 1 g/kg/dose), dexamethasone (0.3 mg/kg/day in divided doses every 6 hours), and cefotaxime (150 mg/kg/day in divided doses every 6 hours) were administered immediately. In addition, leukocyte-depleted red blood cells and fresh frozen plasma were transfused, and intravenous vitamin K and endotracheal epinephrine were administered. Inotropics (dopamine and dobutamine) were also injected. Lumbar puncture was scheduled but was ultimately not performed because of the patient's unstable vital signs.
Massive pulmonary hemorrhage had continued till death. The patient expired 15 hours after admission, and it was within 3 days of fever onset and within 9 hours after intubation. We reported it to the Korea Centers for Disease Control and Prevention (KCDC), sending his urine, serum, nasopharyngeal swab, and endotracheal aspirate. Enterovirus genome detection was attempted by realtime reverse transcription-polymerase chain reaction (RT-PCR) using a TaqMan system (Applied Biosystems, Foster City, CA, USA). For genotyping, seminested RT-PCR was used to amplify part of the viral protein 1 (VP1) gene of enteroviruses according to the KCDC protocol, and sequencing analysis for the VP1 amplicon was performed by an automatic sequencer and the DNASTAR software package (DNASTAR Inc., Madison, WI, USA). The KCDC isolated enterovirus 71 with subgenotype C4a from the patient's serum and nasopharyngeal swab. Bocavirus was also isolated from his nasopharyngeal swab by real-time RT-PCR at our hospital.
To our knowledge, this is the first reported case of HFMD with sudden-onset massive pulmonary hemorrhage in Korea. Pulmonary edema/hemorrhage is the mysterious hallmark symptom of enteroviral HFMD and can kill a child within 1 day2). An epidemic study conducted in Taiwan from 1998 to 2005 reported that pulmonary edema/hemorrhage occurred in 43% of patients with severe culture-proven enteroviral HFMD/herpangina2). This discrepancy maybe due to different enterovirus virulence or subgenotypes, or differences in susceptibility with respect to ethnicity.
Pulmonary edema/hemorrhage is characterized by respiratory distress, tachypnea, tachycardia, hemoptysis, and rapidly progressing diffuse infiltration or congestion on a chest film2). A study on the outbreak in Taiwan in 1998 reported that enterovirus 71-infected HFMD patients with pulmonary edema had suddenonset tachypnea, tachycardia, and cyanosis within 1-3 days after disease onset, and pulmonary edema led to a rapid death within 12 hours after intubation10). The present patient exhibited a change in consciousness, respiratory distress, tachypnea, tachycardia, hemoptysis, rapidly progressing infiltration on a chest film, hypoxemia, and hypotension. This patient ultimately expired within 3 days of fever onset and within 9 hours after intubation. In this report, we had applied pulse oxymetry from the beginning, so we could find his desaturation immediately after pulmonary hemorrhage. We suggest saturation monitoring for HFMD patients with central nervous system (CNS) involvements for early detection.
The pathogenesis of pulmonary edema/hemorrhage due to enterovirus 71-induced HFMD remains unclear. It may be caused by increased pulmonary vascular permeability as a result of either brainstem encephalitis or a systemic inflammatory response caused by excessive cytokine release2,6).
CNS involvement of enterovirus 71-induced HFMD may also trigger a sympathetic storm, resulting in vasoconstriction with high systemic vascular resistance, passive pulmonary volume loading, and pulmonary edema/hemorrhage8). The present patient had several episodes of convulsions, lethargy, and loss of consciousness. Therefore, we suspected encephalitis, although we were unable to perform a cerebrospinal fluid study because of his unstable vital signs. His suspected encephalitis may be a possible cause of the pulmonary hemorrhage. However, pulmonary edema/hemorrhage without CNS involvement occurred in 11% of patients with severe culture-proven enteroviral HFMD/herpangina in an epidemic study in Taiwan from 1998 to 20052). Another possible explanation for the pathogenesis is coinfection with a second virus2), as bocavirus coinfection was detected in the patient. However, the presence of both viruses would not result in increased mortality due to HFMD, according to a previous study2).
A multicenter study performed by the Enterovirus Complication Working Group investigating enterovirus 71-induced HFMD in Korea in 2009 reported that an HFMD rash pattern, fever longer than 4 days, peak body temperature >39℃, vomiting, headache, neurologic signs, leukocytosis, serum glucose >100 mg/dL, and isolated enterovirus 71 may be indicative of poor prognosis during HFMD epidemic periods1,10). In addition, young age at disease onset and a history of lethargy are associated with an increased risk of severe HFMD11). The present patient was young and presented with HFMD rashes and vesicles, fever up to 39℃, vomiting, lethargy, and convulsions as neurologic signs as well as leukocytosis, high serum glucose level, and isolated enterovirus 71 as poor prognostic factors. HFMD patients are very frequently approximately 1 year of age, and deaths were reported to occur at around 1 year of age in other studies12,13,14,15). In the 2009 outbreak, 2 patients around 1 year of age expired, and the present patient was a 12-month-old baby.
The gold standard for the diagnosis of enteroviral infection is viral culture and isolation in 2 or more specimens of cerebrospinal fluid, blood, nasopharyngeal swab or secretion, vesicular fluid, or stool to increased diagnostic sensitivity1,4,6,11). In the present report, enterovirus 71 was isolated from both the patient's serum and nasopharyngeal swab. In the 2009 Korean outbreak, the subgenotype of enterovirus 71 was C4a, which was prevalent in China in 2008. In the present case, the subgenotype of enterovirus 71 was also C4a.
The main treatment for enterovirus 71-induced HFMD is supportive management. There are currently no established antiviral treatments or vaccine available for HFMD. The administration of high-dose IVIG is recommended for severe HFMD as optional treatment, because IVIG therapy was effective in many reports4,5,6,7,11). IVIG neutralizes the virus and has nonspecific antiinflammatory properties11); this would significantly reduce mortality by attenuating sympathetic activity and cytokine production5). Furthermore, fluid restriction, inotropic agents, and early intubation with positive-pressure mechanical ventilation are recommended for severe HFMD with pulmonary edema or hemorrhage8). The present patient was immediately treated with IVIG. Inotropics were also injected. Early intubation with positive-pressure mechanical ventilation was performed. Nevertheless, these treatments did not halt the rapid progression.
Enterovirus 71-infected HFMD became a national public health issue in Korea in 2009. Pulmonary hemorrhage in enterovirus 71-infected HFMD is very rare; the present case is likely the first to have occurred in Korea. The present case of fatal HFMD was rapidly aggravated and the patient expired within 3 days from fever onset. Therefore, all physicians should pay special attention to infants and young children with HFMD, particularly in enterovirus 71 epidemic areas such as Korea, to enable early detection and management of severe HFMD. Above all, keeping the infected child at home until full recovery and controlling the preschool with infected child are very important for reducing infection6).
In addition, the development of antiviral agents and vaccines is necessary especially for the control of severe HFMD with complication.
References
1. Kim SJ, Kim JH, Kang JH, Kim DS, Kim KH, Kim KH, et al. Risk factors for neurologic complications of hand, foot and mouth disease in the Republic of Korea, 2009. J Korean Med Sci 2013;28:120–127.



2. Chen KT, Chang HL, Wang ST, Cheng YT, Yang JY. Epidemiologic features of hand-foot-mouth disease and herpangina caused by enterovirus 71 in Taiwan, 1998-2005. Pediatrics 2007;120:e244–e252.


3. Zhang YC, Jiang SW, Gu WZ, Hu AR, Lu CT, Liang XY, et al. Clinicopathologic features and molecular analysis of enterovirus 71 infection: report of an autopsy case from the epidemic of hand, foot and mouth disease in China. Pathol Int 2012;62:565–570.


4. Choi CS, Choi YJ, Choi UY, Han JW, Jeong DC, Kim HH, et al. Clinical manifestations of CNS infections caused by enterovirus type 71. Korean J Pediatr 2011;54:11–16.



5. Ryu WS, Kang B, Hong J, Hwang S, Kim J, Cheon DS. Clinical and etiological characteristics of enterovirus 71-related diseases during a recent 2-year period in Korea. J Clin Microbiol 2010;48:2490–2494.



6. Kim KH. Enterovirus 71 infection: an experience in Korea, 2009. Korean J Pediatr 2010;53:616–622.



7. Liu JT, Peng D, Guan XH, Zou DD, Zhao DC. Clinical characteristics and treatment assessments of severe enterovirus 71 infected children. Zhonghua Er Ke Za Zhi 2010;48:24–28.

8. He SJ, Chen D, Zheng XQ, Wang CX, Huang AR, Jin YM, et al. Three cases of enterovirus 71 infection with pulmonary edema or pulmonary hemorrhage as the early clinical manifestation. Zhonghua Er Ke Za Zhi 2008;46:513–516.

9. Lum LC, Wong KT, Lam SK, Chua KB, Goh AY. Neurogenic pulmonary oedema and enterovirus 71 encephalomyelitis. Lancet 1998;352:1391

10. Chang LY, Lin TY, Hsu KH, Huang YC, Lin KL, Hsueh C, et al. Clinical features and risk factors of pulmonary oedema after enterovirus-71-related hand, foot, and mouth disease. Lancet 1999;354:1682–1686.


11. Ooi MH, Wong SC, Lewthwaite P, Cardosa MJ, Solomon T. Clinical features, diagnosis, and management of enterovirus 71. Lancet Neurol 2010;9:1097–1105.


12. Tseng FC, Huang HC, Chi CY, Lin TL, Liu CC, Jian JW, et al. Epidemiological survey of enterovirus infections occurring in Taiwan between 2000 and 2005: analysis of sentinel physician surveillance data. J Med Virol 2007;79:1850–1860.


13. Kung SH, Wang SF, Huang CW, Hsu CC, Liu HF, Yang JY. Genetic and antigenic analyses of enterovirus 71 isolates in Taiwan during 1998-2005. Clin Microbiol Infect 2007;13:782–787.





 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


