Article Contents
| Korean J Pediatr > Volume 56(9); 2013 |
Abstract
Kawasaki disease (KD) is an acute febrile illness that is the predominant cause of pediatric acquired heart disease in infants and young children. Because the diagnosis of KD depends on clinical manifestations, incomplete cases are difficult to diagnose, especially in infants younger than 1 year. Incomplete clinical manifestations in infants are related with the development of KD-associated coronary artery abnormalities. Because the diagnosis of infantile KD is difficult and complications are numerous, early suspicion and evaluation are necessary.
Kawasaki disease (KD) is an acute febrile vasculitis. The peak incidence of KD is from 6 months to 2 years of age, which includes approximately 50% of all KD patients1,2). The proportion of KD patients younger than 6 months of age in relation to all KD patients is approximately 10%3,4), which is similar to the 11.2% in Japan5) and 7.7% in Korea6). In addition, the incidence of KD patients younger than 3 months of age was 1.7% in Japan5) and 2.2% in Korea6). The incidence of KD is very low in patients younger than 3 months of age3,4) or older than 8 years of age7), and few cases of KD in the neonatal period have been reported8).
Incomplete KD is more common in infants than in older children. Additionally, because of their higher risk for developing coronary artery abnormalities, precise diagnosis is important4). The diagnosis of infantile KD can be particularly difficult due to obscure clinical manifestations. This paper is a review with a special focus on infantile KD.
KD is the predominant cause of pediatric acquired heart disease in developed countries9). Cardiac lesions, such as coronary artery aneurysms, are a hallmark of KD10-12). Coronary artery complications develop in up to 25% of affected children if the disease is left untreated13,14), and these lead to mortality in approximately 2% of cases15,16). Prevention of aneurysms is the primary target for pediatricians treating patients with KD. Prompt diagnosis and the administration of intravenous immunoglobulin (IVIG) can reduce the incidence of coronary artery abnormalities from 25% to 5%17).
To establish a diagnosis, physicians depend on the diagnostic criteria, based on the typical combination of clinical presentations, formulated by the American Heart Association (AHA) (Table 1)18) or the Japanese Kawasaki Disease Research Committee (Table 2)19). However, it is becoming evident that all criteria for KD need not be present at the same time and can appear in sequence. For this reason, the diagnosis of KD is based on a high index of clinical suspicion in cases that lack the typical criteria (incomplete KD). Because the diagnosis of KD is based upon nonspecific clinical signs and because definitive diagnostic test exists, timely identification is challenging.
KD should therefore be considered in the differential diagnosis of every child with fever of at least several days' duration, rash, and nonpurulent conjunctivitis, especially in children younger than 1 year old and in adolescents, in whom the diagnosis is frequently missed.
Experienced clinicians who have treated numerous KD cases can establish the diagnosis before day 4. Fever duration of less than 5 days, which can be achieved by early IVIG treatment, can be considered a principal symptom according to the Japanese criteria20).
Several studies have attempted to define risk factors for the development of coronary artery abnormalities in children with KD. Identified risk factors include incomplete presentation, delay in diagnosis, duration of fever before treatment, male sex, young and older age, and IVIG resistance21,22). Incomplete clinical manifestations in infants and IVIG nonresponsiveness in the older children are particularly associated with the development of KD-associated coronary artery abnormalities23). Most studies also point to the timing of therapy as a critical factor in the development of coronary artery abnormalities.
Infants younger than 1 year tend to show fewer clinical manifestations of the disease, and various features are common by age group. Some studies found that typical mucosal and lymph node changes were absent in infants younger than 6 months. Patients younger than 6 months also had a lower incidence of strawberry tongue and indurative edema of the palms and soles compared with older patients24,25).
The diagnosis of KD is difficult in infants because its presentation is usually atypical and similar to that of other diseases. In our experience, distinguishing between KD and enteroviral meningitis in febrile infants with cerebrospinal fluid pleocytosis poses a diagnostic challenge26). We identified longer duration of fever before admission, higher absolute neutrophil count, increased C-reactive protein (CRP) level, pyuria, and less prominent cerebrospinal fluid pleocytosis as initial features of infants finally diagnosed with KD26). We also encountered the unusual case of infantile parotitis unresponsive to antibiotics and accompanied by prolonged fever that was diagnosed as incomplete KD with coronary complications27).
The current guidelines recommend that infants aged 6 months or younger with fever for 7 days or longer without other explanation should undergo laboratory investigation for indicators of a systemic vascular response (i.e., erythrocyte sedimentation rate [ESR], CRP level)28).
KD is frequently associated with elevation of inflammatory markers including ESR, CRP, and platelet count. Other laboratory findings such as high white blood cell (WBC) count (neutrophilic type), nonseptic pyuria, low sodium levels, hypoalbuminemia, or elevated liver enzymes may supplement the diagnosis29).
KD is rare in infants younger than 3 months of age. The presumptive explanations for this phenomenon include the protective effect of passive immunity transmitted from the mother and the lower possibility of exposure to unknown airborne pathogens because the infants live indoors.
Some studies have reported that infants younger than 6 months old take the longest time to diagnose, are the least likely to fulfill the major clinical criteria, and have the least favorable laboratory results, all of which are risk factors for developing coronary artery abnormalities3,4,30). Coronary artery aneurysm is more frequently observed in infants younger than 6 months, although not statistically significant. Patients younger than 1 year old have more frequent high WBC count and sterile pyuria, but less neck lympadenopathy30). Because many infants present atypically, KD should be considered in all children of 1 year or less with prolonged fever, extreme platelet count elevation, and no compelling alternative diagnosis30,31).
Witt et al.32) concluded that coronary artery abnormalities were more common in cases who did not meet the criteria (20%) compared with those who had the complete clinical presentation (7%). Despite the potential risks of overdiagnosis and overtreatment, this practice seems justified because the complete criteria are an insensitive indicator of having or developing coronary artery abnormalities.
In a retrospective study of 562 patients diagnosed with KD at 8 North American centers, 16 percent were diagnosed after the first 10 days of the illness (i.e., late diagnosis)33). Predictors of delayed diagnosis of KD included age below 6 months, clinical presentation of incomplete KD, greater distance from a tertiary center, and variability between clinical centers. The results of this study underscore the need for a high index of suspicion of KD, especially in infants and patients who present with incomplete KD, in order to identify and treat patients in a timely manner.
These age-specific characteristics could aid customization of the diagnostic and therapeutic strategies in KD, thereby helping to improve the outcome of the disease.
The term "atypical KD" has recently been used to describe patients with an incomplete presentation of the disease, regardless of the presence of coronary complications34,35), and is interchangeable with "incomplete KD." In the absence of a specific test, the diagnosis of clinically incomplete KD based on the history, physical examination, and clinical laboratory results can be challenging.
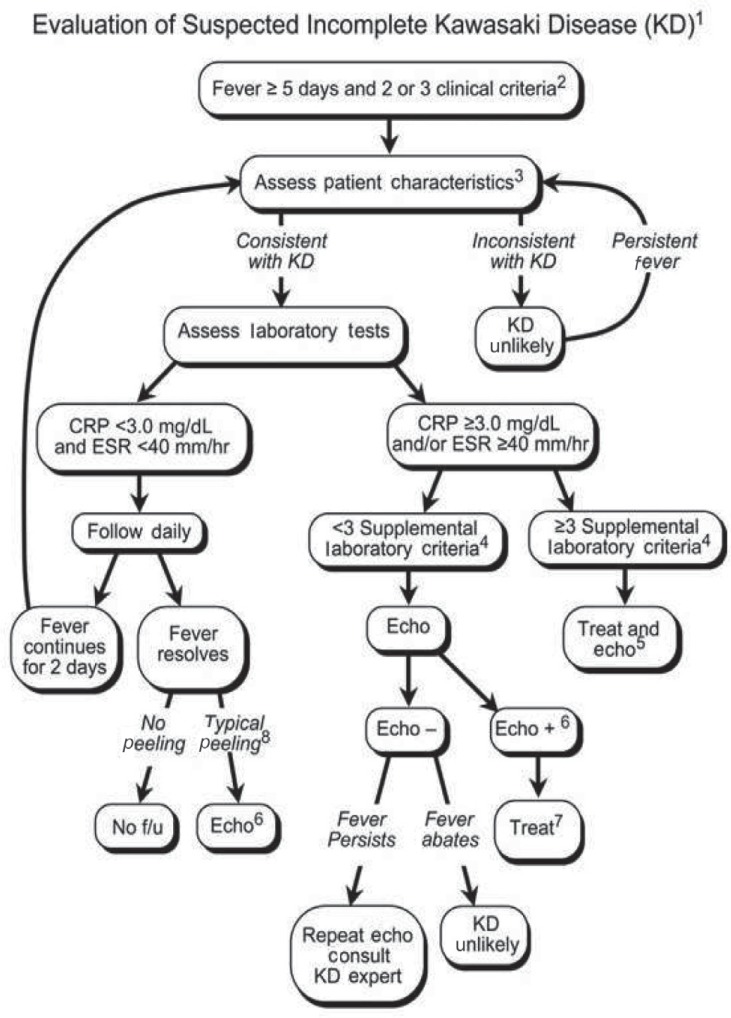
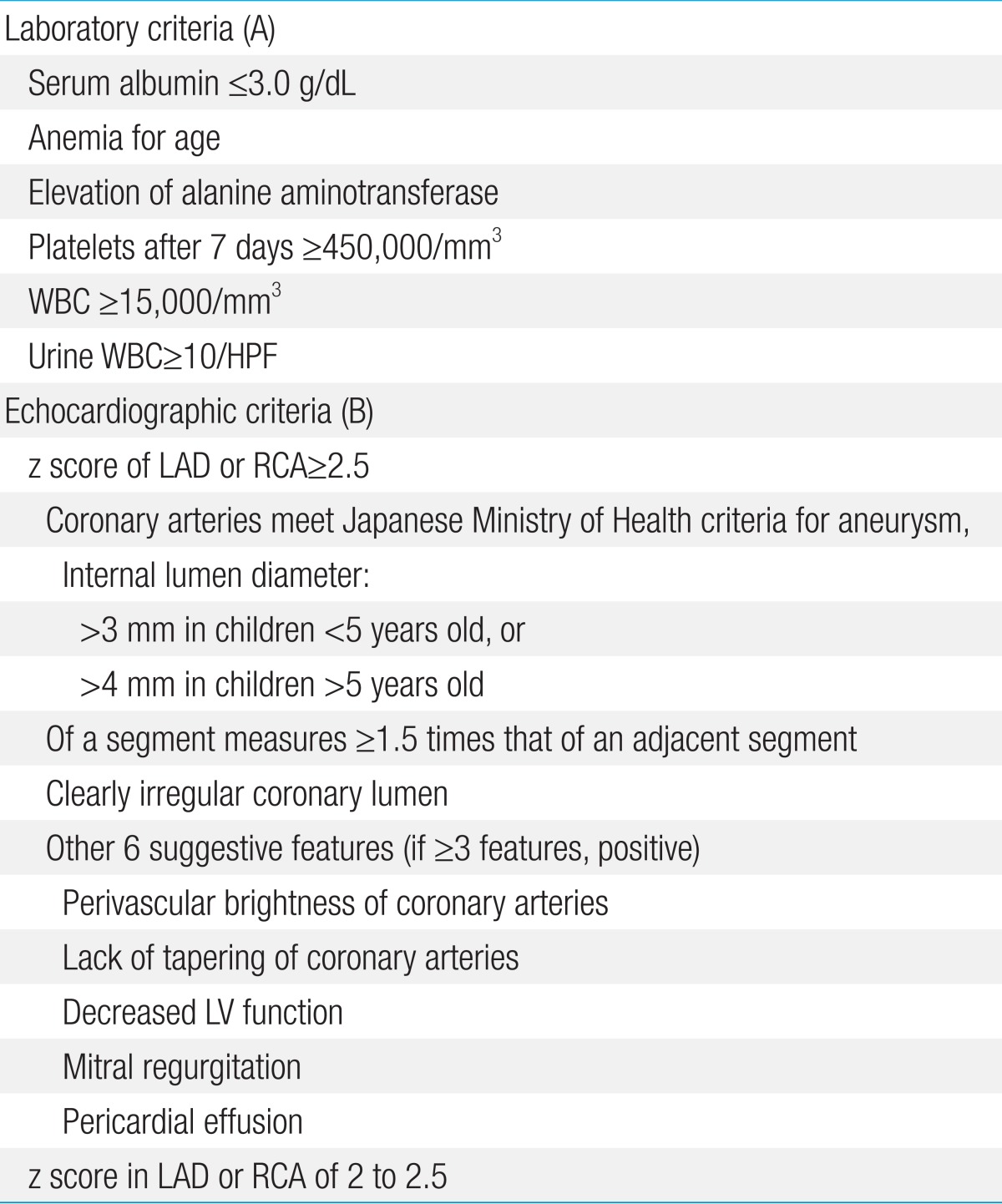
Using the algorithm of the AHA for the diagnosis of incomplete KD (Fig. 1)14), echocardiography should be performed in presentations with evidence of systemic inflammation, even if there are no clinical criteria of KD. This algorithm is offered as guidance to clinicians until an evidence-based algorithm or a specific diagnostic test for KD becomes available. Echocardiography and IVIG administration should be considered if the CRP level is 3 mg/dL or higher or if the ESR is 40 mm/hr or greater14). The AHA recommended a diagnostic algorithm of incomplete KD, which consists of 6 supplemental laboratory and echocardiographic criteria (Table 3)14,36). The presence of more than 3 laboratory criteria supports the diagnosis of incomplete KD (Table 3A). An echocardiogram is considered diagnostically positive if any of the 3 conditions mentioned in Table 3B are met.
A recent report describes some problems with an excessively restrictive definition of incomplete presentation36). Patients who had fever with less than 4 characteristic manifestations were diagnosed with incomplete KD when 2-dimensional echocardiography detected coronary artery involvement. A delay of management may be induced by postponing diagnosis of incomplete presentations until coronary artery abnormality is confirmed. It is now recognized that a large number of patients with KD do not fulfill full clinical KD criteria (i.e., incomplete KD) and that up to 20% of patients who are treated for KD are incomplete cases5,14,37,38).
Chang et al.4) showed higher coronary complications in infants less than 6 months old diagnosed as incomplete KD. Park et al.6) also reported differences between patients with KD younger and older than 6 months in the incidence of coronary artery abnormalities (21.0% vs. 18.7%) and coronary artery aneurysms (4.7% vs. 3.1%), respectively.
However, Lee et al.39) studied the incidence of coronary artery lesions in KD patients younger than 3 months of age and older than 3 months of age and found no significant difference in the prevalence of coronary artery dilatation (19.9% vs. 18.7%) or coronary artery aneurysms (3.4% vs. 2.6%), respectively. They speculated that this unexpected result was because of the very small number of patients younger than 3 months of age among all KD patients, because the majority of pediatricians have a high index of suspicion for incomplete KD, and because early diagnosis can reduce the incidence of incomplete KD in Korea.
Currently, the diagnosis of incomplete KD can be made in cases with fewer classical diagnostic criteria and with several compatible clinical, laboratory, or echocardiographic findings after the exclusion of other febrile illnesses.
Some studies state that almost half of the patients with coronary artery abnormalities were incomplete cases14,40). The high incidence of incomplete KD raises the concern that many cases of KD may be undiagnosed and untreated. The risk of delayed diagnosis and management in young infants with an incomplete presentation has been indicated previously41-44).
Risk factors for incomplete KD are not well established. However, males and children of Asian ancestry appear to have the highest risk, which is consistent with the risk factors for complete presentation. Sudo et al.28) compared clinical risk factors for coronary artery lesions in patients with both incomplete and complete presentations of KD and showed that late IVIG administration (Ōēź7 days of illness) was a significant risk factor for the development of coronary artery lesions in both groups. Infantile KD patients also may have more severe coronary abnormalities and a slower resolution than older children. In addition, diastolic function might be vulnerable to impairment in the infant group based on mitral inflow and tissue Doppler echocardiographic studies45).
The higher rate of incomplete KD in infants may originate from their weakened responses to vasculitis due to an immature immune response, neutralization of superantigen by maternal antibody transferred through the placenta, and cross-reaction to antibody produced by frequent active immunity4). KD should therefore be considered in any infant or child, primarily if younger than 6 months old, with persistent and unexplained fever and laboratory evidence of systemic inflammation, even without more clinical criteria suggestive of the disease, because early recognition and treatment may prevent development of coronary artery dilatation and aneurysm formation. The clinical challenge lies in distinguishing cases of KD that do not fully meet the diagnostic criteria from those that strongly resemble a variety of common childhood disorders.
The majority of coronary artery abnormalities in children with KD have been identified at initial echocardiography during the first week of illness. KD must be considered when prolonged fever is present, mainly in young infants in whom the incomplete forms of the disease are more frequent. A diagnostic approach that includes a high index of suspicion for very young infants who have a high fever with no definite clinical presentations of KD is therefore crucial to prevent coronary artery abnormalities. The majority of the "high-risk" patients (i.e., those who develop coronary artery abnormalities) might be identifiable by echocardiography at the time of initiation of therapy.
References
3. Burns JC, Wiggins JW Jr, Toews WH, Newburger JW, Leung DY, Wilson H, et al. Clinical spectrum of Kawasaki disease in infants younger than 6 months of age. J Pediatr 1986;109:759ŌĆō763.


4. Chang FY, Hwang B, Chen SJ, Lee PC, Meng CC, Lu JH. Characteristics of Kawasaki disease in infants younger than six months of age. Pediatr Infect Dis J 2006;25:241ŌĆō244.


5. Yanagawa H, Nakamura Y, Yashiro M, Ojima T, Tanihara S, Oki I, et al. Results of the nationwide epidemiologic survey of Kawasaki disease in 1995 and 1996 in Japan. Pediatrics 1998;102:E65


6. Park YW, Han JW, Park IS, Kim CH, Cha SH, Ma JS, et al. Epidemiologic study of Kawasaki disease in 6 months old and younger infants. Korean J Pediatr 2008;51:1320ŌĆō1323.

7. Park YW, Han JW, Park IS, Kim CH, Cha SH, Ma JS, et al. Clinical and epidemiologic study of Kawasaki disease in children 8 years of age and older. Korean J Pediatr 2005;48:1139ŌĆō1142.
8. Iino M, Shiraishi H, Igarashi H, Honma Y, Momoi MY. Case of Kawasaki disease in NICU. Pediatr Int 2003;45:580ŌĆō583.


9. Yellen ES, Gauvreau K, Takahashi M, Burns JC, Shulman S, Baker AL, et al. Performance of 2004 American Heart Association recommendations for treatment of Kawasaki disease. Pediatrics 2010;125:e234ŌĆōe241.



10. Neuwirth CA, Singh H. Intercostal artery aneurysm in a child with Kawasaki disease and known coronary artery aneurysms. J Vasc Interv Radiol 2010;21:952ŌĆō953.


11. JCS Joint Working Group. Guidelines for diagnosis and management of cardiovascular sequelae in Kawasaki disease (JCS 2008): digest version. Circ J 2010;74:1989ŌĆō2020.


12. Fukazawa R. Long-term prognosis of Kawasaki disease: increased cardiovascular risk? Curr Opin Pediatr 2010;22:587ŌĆō592.


13. Rowley AH, Gonzalez-Crussi F, Shulman ST. Kawasaki syndrome. Curr Probl Pediatr 1991;21:387ŌĆō405.


14. Newburger JW, Takahashi M, Gerber MA, Gewitz MH, Tani LY, Burns JC, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Pediatrics 2004;114:1708ŌĆō1733.


15. Dajani AS, Taubert KA, Gerber MA, Shulman ST, Ferrieri P, Freed M, et al. Diagnosis and therapy of Kawasaki disease in children. Circulation 1993;87:1776ŌĆō1780.


16. Kato H, Sugimura T, Akagi T, Sato N, Hashino K, Maeno Y, et al. Long-term consequences of Kawasaki disease: a 10- to 21-year follow-up study of 594 patients. Circulation 1996;94:1379ŌĆō1385.


17. Kim SH, Kim KH, Kim DS. Clinical characteristics of Kawasaki disease according to age at diagnosis. Indian Pediatr 2009;46:585ŌĆō590.

18. Council on Cardiovascular Disease in the Young. Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease. American Heart Association. Diagnostic guidelines for Kawasaki disease. Circulation 2001;103:335ŌĆō336.


19. Japan Kawasaki Disease Research Center, Japan Kawasaki Disease Research Committee. Diagnostic guidelines of Kawasaki disease [Internet]. Tokyo: Japan Kawasaki Disease Research Center, Japan Kawasaki Disease Research Committee, c2012;cited 2011 Aug 15. Available from: http://Kawasaki-disease.org/diagnostic/.
20. Research Committee on Kawasaki Disease. Report of Subcommittee on Standardization of Diagnostic Criteria and Reporting of Coronary Artery Lesions in Kawasaki Disease. Tokyo: Ministry of Health and Welfare, 1984.
21. McCrindle BW, Li JS, Minich LL, Colan SD, Atz AM, Takahashi M, et al. Coronary artery involvement in children with Kawasaki disease: risk factors from analysis of serial normalized measurements. Circulation 2007;116:174ŌĆō179.


22. Belay ED, Maddox RA, Holman RC, Curns AT, Ballah K, Schonberger LB. Kawasaki syndrome and risk factors for coronary artery abnormalities: United States, 1994-2003. Pediatr Infect Dis J 2006;25:245ŌĆō249.


23. Song D, Yeo Y, Ha K, Jang G, Lee J, Lee K, et al. Risk factors for Kawasaki disease-associated coronary abnormalities differ depending on age. Eur J Pediatr 2009;168:1315ŌĆō1321.


24. Limbach HG, lindinger A. Das Kawasaki-syndrome bei Sauglingen im ersten Leganshalbjahr. Klin Padiatr 1991;203:133ŌĆō136.


25. Hsiao JY, Chen MR, Huang FY, Kao HA, Sung TC. Clinical analysis of Kawasaki disease in infants below 6 months of age. Zhonghua Min Guo Xiao Er Ke Yi Xue Hui Za Zhi 1988;29:318ŌĆō323.

26. Yeom JS, Park JS, Seo JH, Park ES, Lim JY, Park CH, et al. Initial characteristics of Kawasaki disease with cerebrospinal fluid pleocytosis in febrile infants. Pediatr Neurol 2012;47:259ŌĆō262.


27. Do HJ, Baek JG, Kim HJ, Yeom JS, Park JS, Park ES, et al. Kawasaki disease presenting as parotitis in a 3-month-old infant. Korean Circ J 2009;39:502ŌĆō504.



28. Sudo D, Monobe Y, Yashiro M, Mieno MN, Uehara R, Tsuchiya K, et al. Coronary artery lesions of incomplete Kawasaki disease: a nationwide survey in Japan. Eur J Pediatr 2012;171:651ŌĆō656.


29. Sundel R. Kawasaki disease: clinical features and diagnosis [Internet]. Waltham: UpToDate Inc., 2008;cited 2011 Aug 15. Available from: http://www.uptodate.com/contents/kawasaki-disease-clinical-features-and-diagnosis?detectedLanguage=en&source=search_result&search=Clinical+manifestations+and+diagnosis+of+Kawasaki+disease&selectedTitle=1%7E150&provider=noProvider.
30. Liu HC, Lo CW, Hwang B, Lee PC. Clinical manifestations vary with different age spectrums in infants with Kawasaki disease. ScientificWorldJournal 2012;2012:210382


31. Nigrovic LE, Nigrovic PA, Harper MB, Chiang VW. Extreme thrombocytosis predicts Kawasaki disease in infants. Clin Pediatr (Phila) 2006;45:446ŌĆō452.


32. Witt MT, Minich LL, Bohnsack JF, Young PC. Kawasaki disease: more patients are being diagnosed who do not meet American Heart Association criteria. Pediatrics 1999;104:e10


33. Minich LL, Sleeper LA, Atz AM, McCrindle BW, Lu M, Colan SD, et al. Delayed diagnosis of Kawasaki disease: what are the risk factors? Pediatrics 2007;120:e1434ŌĆōe1440.


34. Burns JC, Mason WH, Glode MP, Shulman ST, Melish ME, Meissner C, et al. United States Multicenter Kawasaki Disease Study Group. Clinical and epidemiologic characteristics of patients referred for evaluation of possible Kawasaki disease. J Pediatr 1991;118:680ŌĆō686.


35. Forsey J, Mertens L. Atypical Kawasaki disease: a clinical challenge. Eur J Pediatr 2012;171:609ŌĆō611.


37. Yanagawa H, Nakamura Y, Yashiro M, Oki I, Hirata S, Zhang T, et al. Incidence survey of Kawasaki disease in 1997 and 1998 in Japan. Pediatrics 2001;107:E33


38. Dominguez SR, Anderson MS, El-Adawy M, Glode MP. Preventing coronary artery abnormalities: a need for earlier diagnosis and treatment of Kawasaki disease. Pediatr Infect Dis J 2012;31:1217ŌĆō1220.


39. Lee EJ, Park YW, Hong YM, Lee JS, Han JW. Epidemiology of Kawasaki disease in infants 3 months of age and younger. Korean J Pediatr 2012;55:202ŌĆō205.



41. Manlhiot C, Christie E, McCrindle BW, Rosenberg H, Chahal N, Yeung RS. Complete and incomplete Kawasaki disease: two sides of the same coin. Eur J Pediatr 2012;171:657ŌĆō662.


42. Sonobe T, Kiyosawa N, Tsuchiya K, Aso S, Imada Y, Imai Y, et al. Prevalence of coronary artery abnormality in incomplete Kawasaki disease. Pediatr Int 2007;49:421ŌĆō426.


43. Chuang CH, Hsiao MH, Chiu CH, Huang YC, Lin TY. Kawasaki disease in infants three months of age or younger. J Microbiol Immunol Infect 2006;39:387ŌĆō391.

Fig.┬Ā1
Evaluation of suspected incomplete Kawasaki disease. 1) In the absence of a gold standard for Kawasaki disease diagnosis, this algorithm cannot be evidence based but rather represents the informed opinion of the experts committee. Consultation with an expert should be sought anytime assistance is needed. 2) Infants Ōēż6 months old with fever for Ōēź7 days without any explanation should undergo laboratory testing, and if evidence of systemic inflammation is found, echocardiography should be performed, even if the infants have no clinical criteria. 3) Patient characteristics suggestive of Kawasaki disease are listed in Table 118,36). Characteristics suggestive of a disease other than Kawasaki disease include exudative conjunctivitis, exudative pharyngitis, discrete intraoral lesions, bullous or vesicular rash, or generalized adenopathy. An alternative diagnosis should be considered. 4) Supplemental laboratory criteria include albumin Ōēż3.0 g/dL, anemia, elevation of alanine aminotransferase, platelet count Ōēź450,000/mm3 after 7 days, WBC count Ōēź15,000/mm3, and WBC in urine Ōēź10/high-power field. 5) Treatment can be undertaken before performing echocardiography. 6) The echocardiogram is considered positive for the purpose of this algorithm if any of the following 3 conditions are met: (1) z score of LAD or RCA Ōēź2.5; (2) the coronary arteries meet the Japanese Ministry of Health criteria for aneurysms; and (3) Ōēź3 other suggestive features exist, including perivascular brightness, lack of tapering, decreased LV function, mitral regurgitation, pericardial effusion, or z scores in LAD or RCA of 2-2.5 (Table 3)14,36). 7) If the echocardiogram is positive, treatment should be given to children within 10 days of fever onset and those with fever beyond day 10 with clinical and laboratory signs (CRP, ESR) of ongoing inflammation. 8) Typical peeling begins under the nail bed of the fingers and then the toes. WBC, white blood cell; LAD, left anterior descending coronary artery; RCA, right coronary artery; LV, left ventricular; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; f/u, follow-up. Reproduced from Newburger JW, et al. Pediatrics 2004;114:1708-33, with permission of American Academy of Pediatrics14).
Table┬Ā2
Six principal symptoms in the diagnostic guidelines for Kawasaki disease
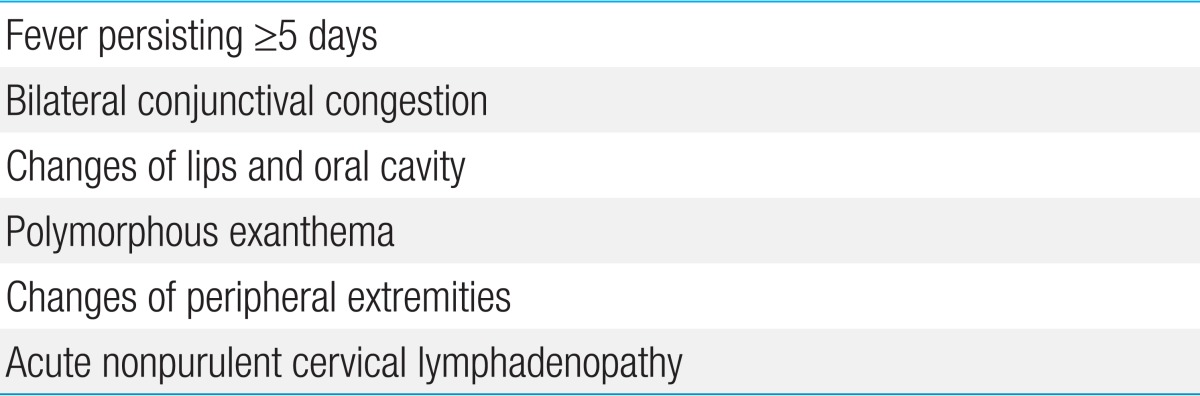
Adapted from Japan Kawasaki Disease Research Center, Japan Kawasaki Disease Research Committee. Diagnostic guidelines of Kawasaki disease [Internet]. Available from: http://Kawasaki-disease.org/diagnostic/19) and Yu JJ. Korean J Pediatr 2012;55:83-736).




 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


