Article Contents
| Clin Exp Pediatr > Volume 65(11); 2022 |
|
Abstract
Seizures are the most common neurological disorders in newborns. Managing neonatal seizures is challenging, especially for neurologists who are not neonatal specialists. Acute brain injury during ischemic insult is a key component of seizure occurrence, while genetic and metabolic disorders play less prevalent but more severe roles. The diagnosis of neonatal seizure is ambiguous, as the subjective differentiation between seizure and nonepileptic events is difficult; therefore, electrographic recording is the gold standard for diagnosis. The detection of electrographic seizures by neonatologists is currently facilitated by amplitude-integrated electroencephalography availability in many neonatal intensive care units. Although it is less sensitive than conventional electroencephalography, it is better to record all risky neonates to filter the abnormal events as early as possible to enable the initiation of dedicated therapy at proper dose and time and facilitate the initial response to antiepileptic drugs. This, in turn, helps maintain the balance between unnecessary drug use and their neurotoxic effects. Moreover, the early treatment of electrographic seizures plays a vital role in the suppression of subsequent abnormal brain electricity (status epilepticus) and shortening the hospital stay. An explicit understanding of seizure etiology and pathophysiology should direct attention to the proper prescription of short- and long-term antiepileptic medications to solve the challenging issue of whether neonatal seizures progress to postneonatal epilepsy and long-term cognitive deficits. This review addresses recent updates in different aspects of neonatal seizures, particularly electrographic discharge, including their definition, etiology, classification, diagnosis, management, and neurodevelopmental outcomes.
Graphical abstract
Seizure is the most common and critical neurological disorder in newborns [1]. Its incidence ranges from 1/1,000 to 5/1,000 live births in population-based studies [2-5] and 8.6/1000 live births in a neonatal intensive care unit (NICU)–based study [6]. Approximately 80% of neonatal seizures occur in the first week of life [7]. However, this subject remains a mystery due to dissimilarities and special characteristics of neonates versus older age groups and the fact that most seizures are subclinical without overt clinical correlates [8].
Neonatal seizure is a paroxysmal stereotypic repetitive event due to abnormal electrical activity of the brain caused mainly by ischemic or hemorrhagic insult [9]. The duration of this electrographic event remains under debate, as the American Clinical Neurophysiology Society defines neonatal seizures as an abnormal repetitive evolving (in frequency, voltage, and morphology) electrographic pattern lasting for at least 10 seconds [10], while brief rhythmic discharges lasting less than 10 seconds cause special concern as they have worse outcomes than electrographic events [11]. Therefore, the International League Against Epilepsy (ILAE) defines seizure as an abnormal evolving electrical signal with definite onset and offset; its duration is not necessary 10 seconds but should be long enough to detect changes in signal frequency, morphology, and resolution [12].
Despite the silent nature of electrographic seizures, they have gained special consideration in recent years, as in the selected high-risk hypoxic ischemic encephalopathy (HIE) group, 50%–80% of seizures are electrographic only and the extent of seizure burden may be greatly underestimated [13-15]. Moreover, they may adversely affect the brain and increase morbidity and mortality with increased risk of death, severe cerebral palsy (CP), microcephaly, and failure to thrive. Long-term neurological follow-up of these patients with electroclinical and electrographic-only seizures is poor, with significant morbidity and mortality rates [14].
Therefore, it seems reasonable to depend on electroencephalography (EEG) to accurately diagnose neonatal seizures and overcome the dilemma of clinical epileptic and nonepileptic events, and polygraphic video EEG can help to evaluate any manifestations in question, such as autonomic features or automatisms, and decrease the risk of overdiagnosing common nonseizure events as epileptic [16].
Variability in the clinical presentation of neonatal seizures is the major factor of its misdiagnosis since it depends on clinical symptoms only, which may lead to overdiagnosis (excessive abnormal movement such as jitteriness, startle) or underdiagnosis (presence of electrical activity in areas away from the motor cortex) [17]. Therefore, the clinical suspicion of seizures should be verified by conventional/amplitude-integrated EEG (EEG/aEEG) recording whenever available. Of note, conventional EEG remains a cornerstone for diagnosis but cannot be used for long-term monitoring; thus, aEEG is a less sensitive but easily applicable and interpretable alternative in many centers [18].
A main obstacle is that EEG may miss some clinical events without recording due to the reduced montage of EEG in neonates, as only 9 electrodes are placed and focal seizures originating from subcortical cerebral areas such as the limbic and peri-limbic systems may be missed on EEG. However, studies have shown that the electrographic ictal pattern of epileptic seizures becomes apparent during prolonged EEG monitoring [14,19]. We must stress that clinical episodes that have no ictal EEG correlation are not seizures. The ambiguous “EEG-negative seizure” concept is eliminated [10] as seizures that do not originate from or migrate to the motor cortex do not result in obvious movements. Therefore, the placement of more EEG electrodes and continuous prolonged bedside electrographic monitoring are essential for interpreting EEG-negative seizures.
Another main obstacle is that EEG is not available in every NICU; therefore, it is better to follow the flowchart developed by the Brighton collaboration illustrating different degrees of diagnostic certainties (definite, probable, possible) [9] depending on the available tools. Therefore, a definite diagnosis is reached if an electrographic seizure is recorded by conventional EEG with or without clinical events, whereas probable certainty is reached if events are recorded on aEEG or focal clonic or focal tonic clinical events are witnessed. However, it is possible to ascertain whether other clinical events, such as automatisms, autonomic seizures, and seizures with behavioral arrest, are seen.
Notably, particular attention should be paid to the role of EEG as a screening tool for high-risk newborns and its application as early as possible to detect electrographic events even before their clinical occurrence. This idea has been studied by Wusthoff et al. [20], who found that seizures in HIE often lack distinct clinical signs and that preemptive use of conventional EEG for seizure screening increases treatment success compared to confirmatory EEG after clinically suspected seizures occur. Therefore, it is important for future research to redirect the goal of using conventional EEG as a tool to screen for seizures among high-risk neonates to facilitate an earlier and more accurate diagnosis and thereby more effective treatment avoiding unnecessary medication for seizure mimics.
Several years ago, Volpe [21] tried to classify seizures based on semiology only into tonic, clonic, myoclonic, and subtle types (staring, sudden awakening and alerting, eyeball deviation, eye blinking, nystagmus, chewing, and limb movements such as swimming, rowing, and pedaling), both focal and generalized. After the integration of EEG for the diagnosis, seizures are divided into electroclinical or electrographic only [22].
Electrographic seizure involves the presence of an electrographic seizure observed on EEG that is not associated with any evident clinical signs (synonyms: clinically silent or subclinical seizures). The term electrographic-only is preferred, as it depends on the observational methods used and the seizure may not be truly subclinical [12]. The diagnosis of clinical events in neonates is challenging for many reasons [23,24]. First, clear clinical events occur in a small number of neonates due to their immature motor pathways and underdeveloped central nervous system (CNS) connections [25] in term and preterm neonates; thus, the seizures may not spread to certain areas such as the motor cortex. Moreover, seizures that remain focal in the nonmotor cortex do not generate clear clinical events, which may hinder accurate segregation between seizures and nonepileptic movements [26].
Second, since newborns are preverbal, they are unable to communicate sensory phenomena associated with seizures, while a seizure in the temporal cortex may manifest as interrupted speech or in the occipital cortex may manifest as visual events that cannot be assessed. Third, neonates develop pseudo-paroxysmal events, such as jitteriness, rapid eye movements, automatisms, and sleeprelated myoclonus. Fourth, electroclinical “uncoupling” occurs after antiepileptic drug administration as clinical events stop but electrographic events continue, which may lead to premature discontinuation of antiepileptic drugs [27]. Finally, clinical observation of neonates in the NICU is often inhibited by isolette coverings, dark lighting, and other environmental features of modern neurocritical care. Therefore, the clinical signs of neonatal seizures are always subtle or even absent, and even the most attentive observer may miss them.
To our knowledge, the ILAE [12] recently proposed a diagnostic framework for the classification of neonatal seizures that consists of 4 zones: (1) clinical presentation: risk factor or clinically suspicious events; (2) diagnosis: EEG recording; (3) manifestation: with or without clinical events; (4) seizure types: motor: automatisms, clonic, epileptic spasms, myoclonic, sequential, and tonic; and nonmotor: autonomic and behavioral arrest; unclassified or electrographic only. The special consideration for this classification is that all neonatal seizures start with focal onset with special concern about unique sequential type, clinically only events are eliminated, and an impaired or preserved level of consciousness is unnecessary.
Etiological drives for neonatal seizures are related to acute brain insult, such as stroke, hemorrhage, infection, or electrolyte disturbance. Their onset usually occurs within one week after a dedicated event and are considered acute symptomatic/provoked seizures [28] or related to neonatal structural, metabolic, or genetic epileptic syndromes [29] that occur in the absence of a potentially causative clinical condition or beyond the interval of an acute event [30]. These represent approximately 15% of causes and are known as unprovoked seizures [31]. Coronavirus disease 2019 (COVID-19) infection remains uncommon in neonates, and little is known about its neurological complications. Neurological manifestations are mainly parainfectious and immune-mediated; Martin et al. [32] reported the first case of neonatal seizures due to COVID-19-induced encephalitis with good response to antiepileptic drugs and a normal short-term outcome. Although the clinical course is known to begin more with favorable outcomes than those of adults, one study showed an unusual presentation of COVID infection in full-term babies, including multisystem involvement and cerebral ischemic lesions [33]. In addition, 2 neonates exposed to prenatal infection developed ventriculomegaly, neurological dysfunction, and seizures [34]. A recent study emphasized that the COVID-19 pandemic has led to a widespread disruption in the delivery of developmental services to children with a history of neonatal seizures [35].
Of note, seizure onset and type are crucial to identifying the underlying etiology and hence tailoring early distinct therapeutic interventions aimed at reducing the detrimental effects of seizures on synaptic plasticity in the growing brain. HIE accounts for approximately two-thirds of acute symptomatic seizures occurring on the first day of life, whereas genetic disorders occur around the seventh day of life [36,37].
The clinical course of seizures beyond the neonatal period is challenging. There are 4 clinical scenarios (A, B, C, and D) according to brain injury etiology and severity [38]. The clinical courses of acute symptomatic seizures are completely within in the neonatal period (A), waking up in the postneonatal period within the first year of life [39,40] after the latent period of control (C), or continued without control, particularly in severe brain injury such as infectious encephalitis and severe HIE (D), whereas structural or genetic epilepsy is a remote cause of symptomatic seizures (B). To some extent, seizure onset in neonatal epilepsy overlaps with acute provoked events, as it may be superimposed onto acute brain injury [28].
Treatment protocols for neonatal seizures have not significantly changed in recent decades, although they only are initial responders [41]. First-line therapy that acts through the gammaaminobutyric acid (GABA) receptor, that is, phenobarbital and benzodiazepines. Phenytoin or levetiracetam as second-line treatment has not yielded superior results [42]. Levetiracetam is attractive for its neuroprotective effect [43], although a recent study suggested poor seizure termination efficacy compared to phenobarbital [44].
Moreover, many centers use midazolam infusion for persistent events [42]. Lidocaine is also an alternative in refractory cases if phenytoin has not been previously utilized [45]. Alternative drugs, such as topiramate, have more limited use because of the unavailability of an intravenous formulation despite its neuroprotective effect [46]. It acts by inhibiting α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid and kainate receptors and potentiating GABA signaling, resulting in dose-dependent brain injury reduction and neurobehavioral improvement. In addition, when combined with hypothermia, topiramate use was correlated with a lower seizure burden during therapeutic hypothermia, less need for drug titration, lower mortality rates in newborns with HIE, and a lower prevalence of epilepsy [47]. Table 1 summarizes the advantages and disadvantages of commonly used antiepileptic drugs for the treatment of neonatal seizures.
Clinicians and researchers have begun to widen the comfort zone for the treatment of neonatal seizures, weighing the balance between benefits and harms by reducing the seizure burden and drug neurotoxicity. Therefore, recent studies paid great attention to the treatment of electrographic seizures even without clinical events as it is the common presentation in neonates [26]. The treatment of electrographic seizures is ideally time-critical, as infants treated within 1 hour of seizure onset had the lowest seizure burden and fewer seizures over the subsequent 24 hours [48]. However, a recent study reported no difference in mortality or long-term morbidity in neonates treated after clinical or electrical events with some concern with worse cognitive function in those with electrographic seizures [49]. Briefly, the threshold for escalation of therapy to decrease seizure burden remains an area of active research.
The duration of drug continuation after seizure resolution remains variable [50] but accumulating evidence supports their earlier discontinuation before hospital discharge, although exact timing has not been reported [42] if acute symptomatology is confirmed, as it will not be associated with the risk of childhood epilepsy or worse neurodevelopmental outcomes [51]. Interestingly, Vegda et al. [52] designed an algorithm for medication discontinuation based on the risk of recurrence and availability of EEG/aEEG; therefore, seizures that are controlled with one drug and resolved within 2–3 days, it is reasonable to withdraw and stop the medication prior to discharge. If the seizures exceed 7 days, require more than 2 drugs for control, or are confirmed as epilepsy, it is reasonable to maintain the baby on 1 or 2 drugs at discharge and refer them for following by a neurologist to decide when to wean the medication.
Acute symptomatic seizures have been identified as a major risk factor for neurological and cognitive impairment as well as the development of epilepsy in children in developing countries [53]. The main prognostic factors associated with poor outcomes are prematurity, low birth weight, low Apgar score, severe HIE, high-grade intraventricular hemorrhage, abnormal EEG background in the form of burst suppression or long-term low voltage, seizure onset on the first day or that persisted after the third day of life, status epilepticus, CNS infection, and brain damage [9,54]. The most common neurological sequelae of neonatal seizures include developmental delay (30%–50%) [9], epilepsy (20%–35%) [9], and CP (15%–30%) [9]. Mortality correlates with the etiology of seizures (7%–25% in neonates with seizures) [9], the highest mortality rate is related to HIE, hemorrhage, and stroke [13].
Seizure etiology, burden, and response to medications are the key factors of outcomes (Table 2) [55]. The underlying causes of seizures is considered the critical determinant of neurological abnormalities later in life, as the relative risk for adverse outcomes in neonates with perinatal asphyxia was 8.41 while that for neonates with stroke and seizures was 4.95 compared to that of those with stroke but no seizures [56]. Of note, Oh [57] estimated the impact of neonatal seizures on subsequent neurological disorders, such as epilepsy, intellectual disability, psychiatric and behavioral disorders, and headache, independent of the putative underlying etiology. They found that the risk of developing epilepsy, intellectual disability, and headache among newborn infants with seizures was 32.7, 2.0, and 1.6 times that among those without neonatal seizures, respectively. Surprisingly, the risk of subsequent epilepsy was considerably higher in infants who were prescribed antiepileptic drugs.
Moreover, there is a positive relationship between seizure burden and brain injury severity [57]; a maximal seizure burden exceeding 13 min/hr is associated with neurological morbidities in HIE infants [58]. Further studies found that electrographic seizure burden exceeding 10 minutes is significantly associated with severe brain injuries [55,59,60]. Surprisingly, seizure burden is eliminated with an early start of dedicated therapy and antiepileptic drugs, resulting in a reduction in the development of status epilepticus [61,62] and better cognitive outcomes [60].
Of note, many studies reported a high incidence of seizures during cooling and rewarming periods in cooled HIE newborns [24,63-65]. Interestingly, Chalak et al. [66] found a higher seizure burden during rewarming and worse long-term outcomes at 2 years of age and highlighted the importance of continuous EEG monitoring during the rewarming period. In addition, a longer seizure duration is associated with a higher 1-year risk of epilepsy in children with perinatal asphyxia, and each 10-min increase in seizure burden is associated with a 5-fold increase in the risk of epilepsy, while having more than 10 seizures increases the risk of epilepsy by 30-fold compared to no seizures [67].
Moreover, worse outcomes and higher mortality rates were scored in neonates with electrographic-only seizures versus those with clinical or electroclinical seizures (P<0.002), and the mortality rate was doubled in neonates who did not respond initially to the loading dose of antiepileptic drugs [13]. The number of electrographic seizures (none, 1–75, >75) correlates with subsequent mortality and morbidity in a cohort of at-risk newborns and newborns with HIE [68]. Background EEG and electrographiconly seizures were independent predictors of a lack of response to phenobarbital in one study [69]; moderately and severely abnormal EEG findings were associated with poor response in an additional study, which also identified higher mean seizure score and higher degrees of brain magnetic resonance imaging injury (white matter, cortex, and watershed regions) were associated with a poor response to phenobarbital [70].
Briefly, the early declaration of this silent enemy is vital through the early use of EEG in any suspected or critically ill newborn with the aim of detecting any abnormal discharge and initiating antiepileptic drugs in curable doses that dampen the current and subsequent sparks. Moreover, it is reasonable to pause these drugs as soon as possible after seizure control is achieved due to their neurotoxic effects, with selective subsequent continuation of medication in non-responder babies. The flow chart in Fig. 1 illustrates the initiation and termination of antiepileptic drugs for electrographic seizures.
The threshold of neonatal seizure decreases; therefore, it is reasonable to consider EEG application as a confirmatory tool for clinical events as well as a screening tool for high-risk newborns. The approach to healthy brains after detrimental insult involves building a standardized framework allocating the classification of neonatal seizures based on their EEG signature, pathophysiologic-clinical-semiology features, and thus, selection of nominated antiepileptic drugs for a dedicated duration to overcome their adverse effects.
Footnotes
Fig. 1.
Flow chart of management of electrographic seizures. cEEG, conventional electroencephalogram; aEEG, amplitude integrated EEG; AED, antiepileptic drug; IV, Intravenous.
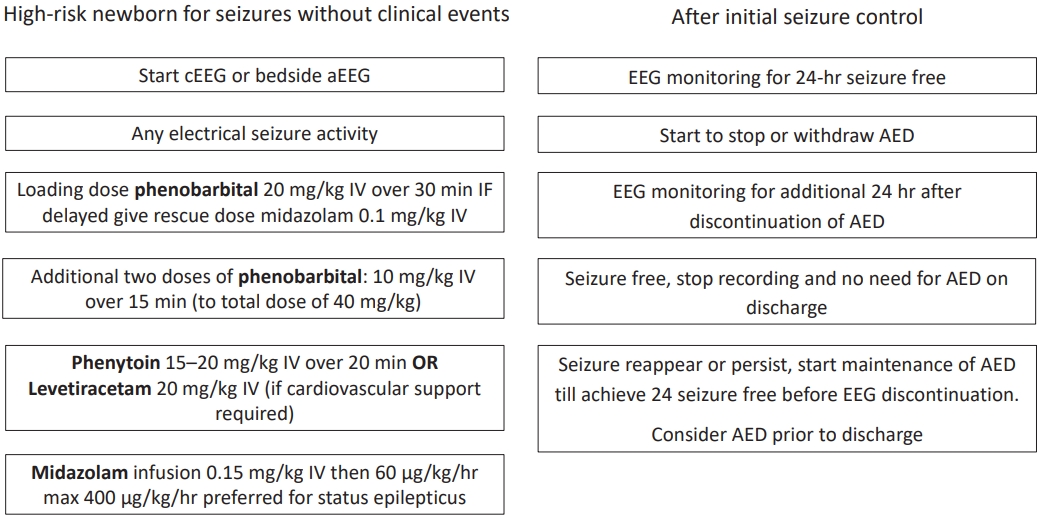
Table 1.
Summary of common antiepileptic drugs in neonates
| Drug | Special consideration | Efficacy | Superiority |
|---|---|---|---|
| Phenobarbital | 1st line of treatment | 43%–80% electrical seizures control [44,71] | Two recent studies showed no significant difference between phenobarbital and levetiracetam as 1st line in treatment of electrical seizures [72,73] while, one study showed superiority of phenobarbital [44] |
| S/E: hypotension, respiratory depression, neuronal apoptosis | |||
| Levetiracetam | 1st or 2nd line of treatment | 35%–86% seizure control[78,79] | |
| Effects: better neurodevelopmental outcome [44,74,75]; antiepileptogenic effect [76]; less reported adverse effects of phenobarbital [77] | |||
| Phenytoin | 1st or 2nd line of treatment | 45% seizure control [80] | |
| S/E: cardiac arrhythmia, hepatoxic | |||
| Midazolam | 2nd or 3rd line of treatment | Variable | One study demonstrated clearance of electrical activity by midazolam infusion within 1 hour after initial phenobarbital [81] |
| S/E: hypotension, respiratory depression | Other study demonstrated seizure control within 3 days by midazolam infusion after initial phenobarbital and phenytoin [82] |
Table 2.
Outcomes of electrographic seizures
| Study | Short-term outcome | Long-term outcome |
|---|---|---|
| Time of treatment of Ez [48] | Achievement of lowest seizure burden, fewer subsequent recurrence within 24 hours after the start of AED within 1 hour of seizure onset | N/A yet |
| Effect of treatment of Cz vs. Ez [49] | No significant difference regarding seizure duration, seizure burden, response to AED, time to reach full oral feeds. | No significant reduction in death or disability at 2 years with special consideration to worse cognitive score in Ez group |
| Little evidence of higher brain injury score of MRI in Ez group | ||
| Treatment of Ez [60] | Lower seizure burden in Ez group | No significant difference between Cz and Ez groups at 18–24 months |
| MSB exceeding 10 minutes is associated with severe brain injury | Of note, increasing seizure burden was associated with lower developmental scores | |
| Contemporary profile of seizures in neonates [13] | Higher seizure burden is associated with high mortality | |
| Seizure burden and neurodevelopmental outcome [58] | Abnormal outcome had been scored if TSB and MSB exceeding 40 and 13 minutes respectively | |
| Seizures burden alone is a predictor of poor outcome | ||
| Prolonged or recurrent seizures [67] | Each 10-minute increase in seizure burden is associated with a 5-fold risk increase of epilepsy, while having more than 10 seizures increases the risk of epilepsy by 30-fold compared to no seizures | |
| Seizure burden during rewarming [66] | High seizure burden during rewarming after 72 hours of cooling compared to 120 hours of cooling | Higher scores of death and severe disability at 2 years are reported in infants with Ez during rewarming |
| Electrographic predictor of AED failure [70] | Significantly higher seizure burden, abnormal EEG background and severe brain injury are seen among non-responders | |
| Role of neonatal seizures on outcome [57] | Epilepsy 32.7 times, intellectual disability 2.0 times and headache 1.6 times greater among newborn infants with seizure than those without neonatal seizures |
References
1. Volpe JJ. Neurology of the newborn. 5th ed. Philadelphia (PA): Saunders/Elsevier, 2008.
2. Glass HC, Pham TN, Danielsen B, Towner D, Glidden D, Wu YW. Antenatal and intrapartum risk factors for seizures in term newborns: a population-based study, California 1998-2002. J Pediatr 2009;154:24-8; e1.



3. Ronen GM, Penney S, Andrews W. The epidemiology of clinical neonatal seizures in Newfoundland: a population-based study. J Pediatr 1999;134:71-5.


4. Lanska MJ, Lanska DJ. Neonatal seizures in the United States: results of the National Hospital Discharge Survey, 1980-1991. Neuroepidemiology 1996;15:117-25.



5. Pisani F, Facini C, Bianchi E, Giussani G, Piccolo B, Beghi E. Incidence of neonatal seizures, perinatal risk factors for epilepsy and mortality after neonatal seizures in the province of Parma, Italy. Epilepsia 2018;59:1764-73.



6. Sheth RD, Hobbs GR, Mullett M. Neonatal seizures: incidence, onset, and etiology by gestational age. J Perinatol 1999;19:40-3.



7. Panayiotopoulos CP. The epilepsies: seizures, syndromes and management. Oxfordshire (UK): Bladon Medical Publishing, 2005;Chapter 2. Optimal use of the EEG in the diagnosis and management of epilepsies.
8. Lloyd RO, O'Toole JM, Pavlidis E, Filan PM, Boylan GB. Electrographic Seizures during the Early Postnatal Period in Preterm Infants. J Pediatr 2017;187:18-25.e2.


9. Pellegrin S, Munoz FM, Padula M, Heath PT, Meller L, Top K, et al. Neonatal seizures: Case definition & guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine 2019;37:7596-609.



10. Tsuchida TN, Wusthoff CJ, Shellhaas RA, Abend NS, Hahn CD, Sullivan JE, et al. American clinical neurophysiology society standardized EEG terminology and categorization for the description of continuous EEG monitoring in neonates: report of the American Clinical Neurophysiology Society critical care monitoring committee. J Clin Neurophysiol 2013;30:161-73.

11. Nagarajan L, Palumbo L, Ghosh S. Brief electroencephalography rhythmic discharges (BERDs) in the neonate with seizures: their significance and prognostic implications. J Child Neurol 2011;26:1529-33.



12. Pressler RM, Cilio MR, Mizrahi EM, Moshé SL, Nunes ML, Plouin P, et al. The ILAE classification of seizures and the epilepsies: modification for seizures in the neonate. Position paper by the ILAE Task Force on Neonatal Seizures. Epilepsia 2021;62:615-28.



13. Glass HC, Shellhaas RA, Wusthoff CJ, Chang T, Abend NS, Chu CJ, et al. Contemporary profile of seizures in neonates: a prospective cohort study. J Pediatr 2016;174:98-103.e1.



14. Boylan GB, Pressler RM, Rennie JM, Morton M, Leow PL, Hughes R, et al. Outcome of electroclinical, electrographic, and clinical seizures in the newborn infant. Dev Med Child Neurol 1999;41:819-25.


15. Boylan GB, Rennie JM, Pressler RM, Wilson G, Morton M, Binnie CD. Phenobarbitone, neonatal seizures, and video-EEG. Arch Dis Child Fetal Neonatal Ed 2002;86:F165-70.



16. Facini C, Spagnoli C, Pisani F. Epileptic and non-epileptic paroxysmal motor phenomena in newborns. J Matern Fetal Neonatal Med 2016;29:3652-9.


17. Nagarajan L, Palumbo L, Ghosh S. Classification of clinical semiology in epileptic seizures in neonates. Eur J Paediatr Neurol 2012;16:118-25.


18. Pisani F, Pavlidis E. The role of electroencephalogram in neonatal seizure detection. Expert Rev Neurother 2018;18:95-100.


19. Worden LT, Chinappen DM, Stoyell SM, Gold J, Paixao L, Krishnamoorthy K, et al. The probability of seizures during continuous EEG monitoring in high‐risk neonates. Epilepsia 2019;60:2508-18.




20. Wusthoff CJ, Sundaram V, Abend NS, Massey SL, Lemmon ME, Thomas C, et al. Seizure control in neonates undergoing screening vs confirmatory EEG monitoring. Neurology 2021;97:e587-96.



21. Volpe JJ. Neonatal seizures: current concepts and revised classification. Pediatrics 1989;84:422-8.



22. Malone A, Ryan CA, Fitzgerald A, Burgoyne L, Connolly S, Boylan GB. Interobserver agreement in neonatal seizure identification. Epilepsia 2009;50:2097-101.


23. Mizrahi EM, Kellaway P. Characterization and classification of neonatal seizures. Neurology 1987;37:1837-44.


24. Nash KB, Bonifacio SL, Glass HC, Sullivan JE, Barkovich AJ, Ferriero DM, et al. Video-EEG monitoring in newborns with hypoxic-ischemic encephalopathy treated with hypothermia. Neurology 2011;76:556-62.



25. Galanopoulou AS, Moshé SL. In search of epilepsy biomarkers in the immature brain: goals, challenges and strategies. Biomark Med 2011;5:615-28.



26. Murray DM, Boylan GB, Ali I, Ryan CA, Murphy BP, Connolly S. Defining the gap between electrographic seizure burden, clinical expression and staff recognition of neonatal seizures. Arch Dis Child Fetal Neonatal Ed 2008;93:F187-91.


27. Scher MS, Alvin J, Gaus L, Minnigh B, Painter MJ. Uncoupling of EEG-clinical neonatal seizures after antiepileptic drug use. Pediatr Neurol 2003;28:277-80.


28. Shellhaas RA, Wusthoff CJ, Tsuchida TN, Glass HC, Chu CJ, Massey SL, et al. Profile of neonatal epilepsies: characteristics of a prospective US cohort. Neurology 2017;89:893-9.



29. Ziobro J, Shellhaas RA. Neonatal seizures: diagnosis, etiologies, and management. Semin Neurol 2020;40:246-56.


30. Beghi E, Carpio A, Forsgren L, Hesdorffer DC, Malmgren K, Sander JW, et al. Recommendation for a definition of acute symptomatic seizure. Epilepsia 2010;51:671-5.


31. Gunawardane N, Fields M. Acute symptomatic seizures and provoked seizures: to treat or not to treat? Curr Treat Options Neurol 2018;20:41.



32. Martin PJ, Felker M, Radhakrishnan R. MR imaging findings in a neonate with COVID-19-Associated encephalitis. Pediatr Neurol 2021;119:48-9.



33. Brum AC, Glasman MP, De Luca MC, Rugilo CA, Urquizu Handal MI, Picon AO, et al. Ischemic lesions in the brain of a neonate with SARS-CoV-2 infection. Pediatr Infect Dis J 2021;40:e340-3.


34. Archuleta C, Wade C, Micetic B, Tian A, Mody K. Maternal COVID-19 infection and possible associated adverse neurological fetal outcomes, two case reports. Am J Perinatol 2022;39:1292-8.


35. Peyton C, Girvan O, Shellhaas RA, Lemmon ME, Rogers EE, Soul JS, et al. Impact of COVID-19 pandemic on developmental service delivery in children with a history of neonatal seizures. Pediatr Neurol 2022;129:14-8.



36. Grinton BE, Heron SE, Pelekanos JT, Zuberi SM, Kivity S, Afawi Z, et al. Familial neonatal seizures in 36 families: Clinical and genetic features correlate with outcome. Epilepsia 2015;56:1071-80.


37. Van Hove JL, Lohr NJ. Metabolic and monogenic causes of seizures in neonates and young infants. Mol Genet Metab 2011;104:214-30.


38. Pisani F, Spagnoli C. Neonatal seizures: a review of outcomes and outcome predictors. Neuropediatrics 2016;47:12-9.


39. Andreolli A, Turco EC, Pedrazzi G, Beghi E, Pisani F. Incidence of epilepsy after neonatal seizures: a population-based study. Neuroepidemiology 2019;52:144-51.



40. Pisani F, Facini C, Pavlidis E, Spagnoli C, Boylan G. Epilepsy after neonatal seizures: literature review. Eur J Paediatr Neurol 2015;19:6-14.


41. Glass HC, Soul JS, Chu CJ, Massey SL, Wusthoff CJ, Chang T, et al. Response to antiseizure medications in neonates with acute symptomatic seizures. Epilepsia 2019;60:e20-4.




42. Keene JC, Morgan LA, Abend NS, Bates SV, Huang SLB, Chang T, et al. Treatment of neonatal seizures: comparison of treatment pathways from 11 neonatal intensive care units. Pediatr Neurol 2022;128:67-74.


43. Khan O, Cipriani C, Wright C, Crisp E, Kirmani B. Role of intravenous levetiracetam for acute seizure management in preterm neonates. Pediatr Neurol 2013;49:340-3.


44. Sharpe C, Reiner GE, Davis SL, Nespeca M, Gold JJ, Rasmussen M, et al. Levetiracetam versus phenobarbital for neonatal seizures: a randomized controlled trial. Pediatrics 2020;145:e20193182.




45. Weeke LC, Toet MC, Van Rooij LG, Groenendaal F, Boylan GB, Pressler RM, et al. Lidocaine response rate in aEEG‐confirmed neonatal seizures: retrospective study of 413 full‐term and preterm infants. Epilepsia 2016;57:233-42.


46. Dixon BJ, Reis C, Ho WM, Tang J, Zhang JH. Neuroprotective strategies after neonatal hypoxic ischemic encephalopathy. Int J Mol Sci 2015;16:22368-401.



47. Nuñez-Ramiro A, Benavente-Fernández I, Valverde E, Cordeiro M, Blanco D, Boix H, et al. Topiramate plus cooling for hypoxic-ischemic encephalopathy: a randomized, controlled, multicenter, double-blinded trial. Neonatology 2019;116:76-84.



48. Pavel AM, Rennie JM, de Vries LS, Blennow M, Foran A, Shah DK, et al. Neonatal seizure management–is the timing of treatment critical? J Pediatr 2022;243:61-8.e2; .



49. Hunt RW, Liley HG, Wagh D, Schembri R, Lee KJ, Shearman AD, et al. Effect of treatment of clinical seizures vs electrographic seizures in full-term and near-term neonates: a randomized clinical trial. JAMA Netw Open 2021;4:e2139604.



50. Shellhaas RA, Chang T, Wusthoff CJ, Soul JS, Massey SL, Chu CJ, et al. Treatment duration after acute symptomatic seizures in neonates: a multicenter cohort study. J Pediatr 2017;181:298-301.e1.



51. Glass HC, Soul JS, Chang T, Wusthoff CJ, Chu CJ, Massey SL, et al. Safety of early discontinuation of antiseizure medication after acute symptomatic neonatal seizures. JAMA Neurol 2021;78:817-25.



52. Vegda H, Krishnan V, Variane G, Bagayi V, Ivain P, Pressler RM. Neonatal seizures-perspective in low-and middle-income countries. Indian J Pediatr 2022;89:245-53.




53. Feigin RD, McCracken GH Jr, Klein JO. Diagnosis and management of meningitis. Pediatr Infect Dis J 1992;11:785-814.


54. Glass HC, Grinspan ZM, Shellhaas RA. Outcomes after acute symptomatic seizures in neonates. Semin Fetal Neonatal Med 2018;23:218-22.


55. Pisani F, Copioli C, Di Gioia C, Turco E, Sisti L. Neonatal seizures: relation of ictal video-electroencephalography (EEG) findings with neurodevelopmental outcome. J Child Neurol 2008;23:394-8.



56. De Haan TR, Langeslag J, van der Lee JH, van Kaam AH. A systematic review comparing neurodevelopmental outcome in term infants with hypoxic and vascular brain injury with and without seizures. BMC Pediatr 2018;18:147.




57. Oh A, Thurman DJ, Kim H. Independent role of neonatal seizures in subsequent neurological outcomes: a population‐based study. Dev Med Child Neurol 2019;61:661-6.



58. Kharoshankaya L, Stevenson NJ, Livingstone V, Murray DM, Murphy BP, Ahearne CE, et al. Seizure burden and neurodevelopmental outcome in neonates with hypoxic–ischemic encephalopathy. Dev Med Child Neurol 2016;58:1242-8.




59. van Rooij LG, Toet MC, van Huffelen AC, Groenendaal F, Laan W, Zecic A, et al. Effect of treatment of subclinical neonatal seizures detected with aEEG: randomized, controlled trial. Pediatrics 2010;125:e358-66.



60. Srinivasakumar P, Zempel J, Trivedi S, Wallendorf M, Rao R, Smith B, et al. Treating EEG seizures in hypoxic ischemic encephalopathy: a randomized controlled trial. Pediatrics 2015;136:e1302-9.



61. Bashir RA, Espinoza L, Vayalthrikkovil S, Buchhalter J, Irvine L, Bello-Espinosa L, et al. Implementation of a neurocritical care program: improved seizure detection and decreased antiseizure medication at discharge in neonates with hypoxic-ischemic encephalopathy. Pediatr Neurol 2016;64:38-43.


62. Harris ML, Malloy KM, Lawson SN, Rose RS, Buss WF, Mietzsch U. Standardized treatment of neonatal status epilepticus improves outcome. J Child Neurol 2016;31:1546-54.



63. Glass HC, Wusthoff CJ, Shellhaas RA, Tsuchida TN, Bonifacio SL, Cordeiro M, et al. Risk factors for EEG seizures in neonates treated with hypothermia: a multicenter cohort study. Neurology 2014;82:1239-44.



64. Cornet MC, Pasupuleti A, Fang A, Gonzalez F, Shimotake T, Ferriero DM, et al. Predictive value of early EEG for seizures in neonates with hypoxicischemic encephalopathy undergoing therapeutic hypothermia. Pediatr Res 2018;84:399-402.



65. Gluckman PD, Wyatt JS, Azzopardi D, Ballard R, Edwards AD, Ferriero DM, et al. Selective head cooling with mild systemic hypothermia after neonatal encephalopathy: multicentre randomised trial. Lancet 2005;365:663-70.


66. Chalak LF, Pappas A, Tan S, Das A, Sánchez PJ, Laptook AR, et al. Association between increased seizures during rewarming after hypothermia for neonatal hypoxic ischemic encephalopathy and abnormal neurodevelopmental outcomes at 2-year follow-up: a nested multisite cohort study. JAMA Neurol 2021;78:1484-93.

67. Fox CK, Mackay MT, Dowling MM, Pergami P, Titomanlio L, Deveber G. Prolonged or recurrent acute seizures after pediatric arterial ischemic stroke are associated with increasing epilepsy risk. Dev Med Child Neurol 2017;59:38-44.




68. McBride MC, Laroia N, Guillet R. Electrographic seizures in neonates correlate with poor neurodevelopmental outcome. Neurology 2000;55:506-13.


69. Spagnoli C, Seri S, Pavlidis E, Mazzotta S, Pelosi A, Pisani F. Phenobarbital for neonatal seizures: response rate and predictors of refractoriness. Neuropediatrics 2016;47:318-26.


70. Dwivedi D, Lin N, Venkatesan C, Kline-Fath B, Holland K, Schapiro M. Clinical, neuroimaging, and electrographic predictors of phenobarbital failure in newborns with hypoxic ischemic encephalopathy and seizures. J Child Neurol 2019;34:458-63.



72. Wagner CB, Kreimer AM, Carrillo NP, Autry E, Schadler A, Cook AM, et al. Levetiracetam compared to phenobarbital as a first line therapy for neonatal seizures: an unexpected influence of benzodiazepines on seizure response. J Pediatr Pharmacol Ther 2021;26:144-50.




73. Thibault C, Naim MY, Abend NS, Licht DJ, Gaynor JW, Xiao R, et al. A retrospective comparison of phenobarbital and levetiracetam for the treatment of seizures following cardiac surgery in neonates. Epilepsia 2020;61:627-35.



74. Falsaperla R, Mauceri L, Pavone P, Barbagallo M, Vitaliti G, Ruggieri M, et al. Short-term neurodevelopmental outcome in term neonates treated with phenobarbital versus levetiracetam: a single-center experience. Behav Neurol 2019;2019:3683548.




75. Maitre NL, Smolinsky C, Slaughter JC, Stark AR. Adverse neurodevelopmental outcomes after exposure to phenobarbital and levetiracetam for the treatment of neonatal seizures. J Perinatol 2013;33:841-6.




76. Talos DM, Chang M, Kosaras B, Fitzgerald E, Murphy A, Folkerth RD, et al. Antiepileptic effects of levetiracetam in a rodent neonatal seizure model. Pediatr Res 2013;73:24-30.




77. McHugh DC, Lancaster S, Manganas LN. A systematic review of the efficacy of levetiracetam in neonatal seizures. Neuropediatrics 2018;49:12-7.


78. Abend NS, Gutierrez-Colina AM, Monk HM, Dlugos DJ, Clancy RR. Levetiracetam for treatment of neonatal seizures. J Child Neurol 2011;26:465-70.




79. Khan O, Chang E, Cipriani C, Wright C, Crisp E, Kirmani B. Use of intravenous levetiracetam for management of acute seizures in neonates. Pediatr Neurol 2011;44:265-9.


80. Painter MJ, Scher MS, Stein AD, Armatti S, Wang Z, Gardiner JC, et al. Phenobarbital compared with phenytoin for the treatment of neonatal seizures. N Engl J Med 1999;341:485-9.







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


