Article Contents
| Clin Exp Pediatr > Volume 69(3); 2026 |
|
Abstract
Background
Purpose
Methods
Results
Supplementary materials
Supplementary Text 1.
Supplementary Table 1.
Supplementary Table 3.
Supplementary Table 4.
Supplementary Table 5.
Supplementary Table 5.1.
Supplementary Table 7.
Supplementary Table 9.
Supplementary Table 10.
Supplementary Table 11.
Supplementary Table 12.
Supplementary Table 13.
Supplementary Table 14.
Supplementary Table 15.
Supplementary Table 16.
Supplementary Table 17.
Supplementary Table 18.
Supplementary Table 19.
Supplementary Table 20.
Supplementary Table 21.
Supplementary Table 22.
Supplementary Figure 1.
Supplementary Figure 2.
Supplementary Figure 3.
Supplementary Figure 4.
Supplementary Figure 5.
Supplementary Figure 6.
Supplementary Figure 7.
Supplementary Figure 8.
Supplementary Figure 9.
Supplementary Figure 10.
Supplementary Figure 11.
Supplementary Figure 12.
Footnotes
Acknowledgments
We would like to extend our sincere gratitude to the patients and their families who contributed to this study. We are deeply thankful to Assistant Professor Dr. Janejira Kittivorapart for her invaluable guidance and support in providing materials and expertise related to GTs. We also express our appreciation to Assistant Professor Dr. Chulaluk Komoltri,Miss Nerisa Thornsri, and Dr. Kernfan Puthikitakawiwong for their exceptional contributions to the statistical analysis, which were instrumental in the completion of this study. Their dedication and insights were invaluable in ensuring the accuracy and rigor of this work.
Fig. 1.
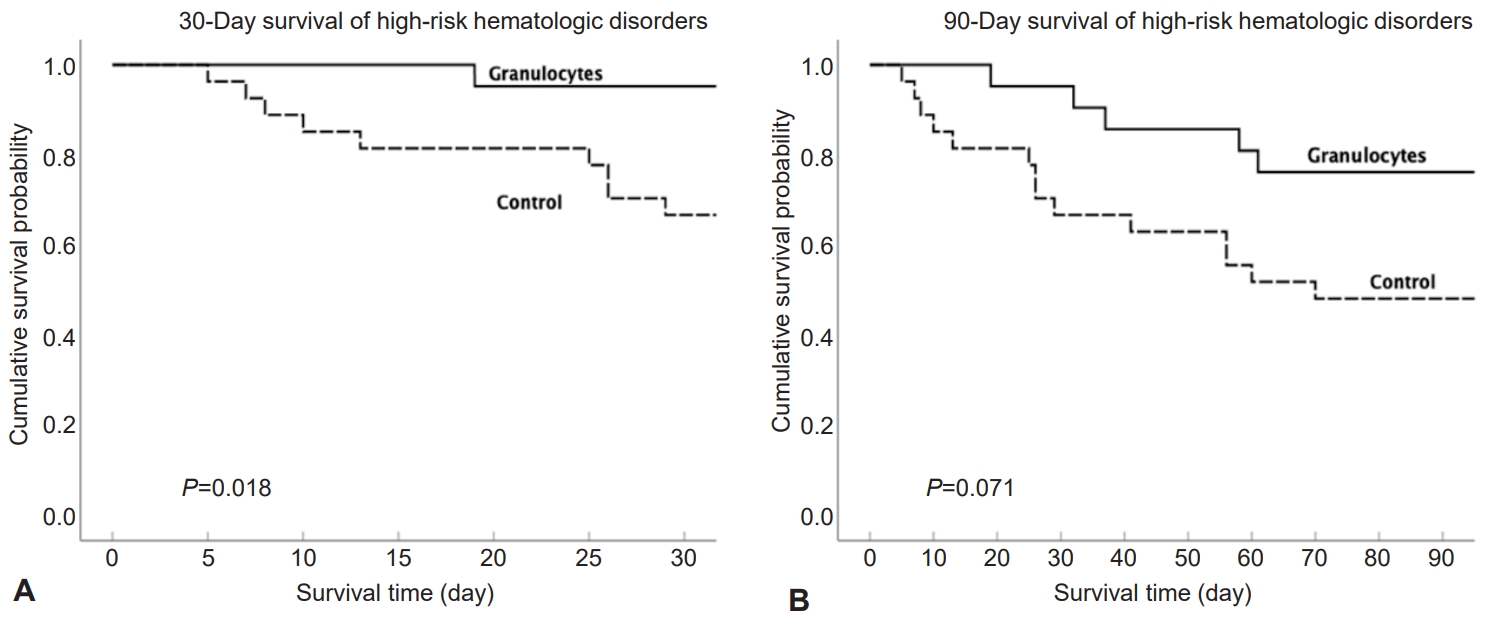
Fig. 2.
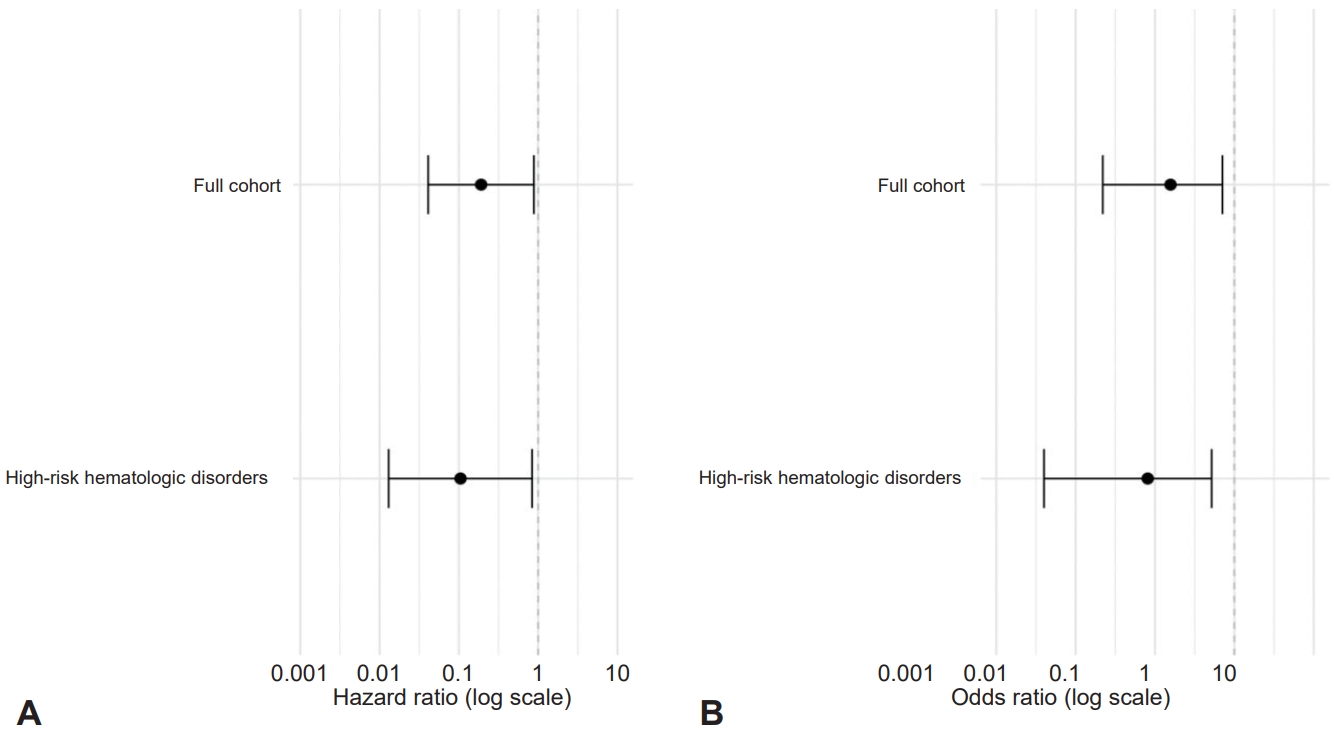
Table 1.
Values are presented as number (%) or median (range).
BSA, body surface area; ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; BAL, biphenotypic acute leukemia; HLH, hemophagocytic lymphohistiocytosis; ATRT, atypical teratoid rhabdoid tumor; HSCT, hematopoietic stem cell transplantation; ANC, absolute neutrophil count.
Granulocyte colony-stimulating factor (G-CSF) use was defined as administration during or after the febrile neutropenia episode.
Table 2.
Values are presented as number (%).
MDR, multidrug-resistant.
The sources of infection are detailed in Supplementary Table 23.
Table 3.
CI, confidence interval; OR, odds ratio; MDR, multidrug resistance; ANC, absolute neutrophil count; HR, hazard ratio; GT, granulocyte transfusion; IPTW, inverse probability of treatment weighting.
See Supplementary Table 12 for the doubly robust IPTW Cox regression results for the high-risk hematologic condition subgroup.
Multivariate logistic regression, Cox regression, and inverse probability of treatment weighting (IPTW) models were used to evaluate the association between GT and clinical outcomes, including 30- and 90-day survivals, and composite outcomes (death or no recovery by day 30). Each model estimated the independent effects of GT while adjusting for relevant confounders.
The IPTW models used stabilized weights derived from baseline propensity scores to balance the covariates between treatment groups. “GT (weighted)” indicates that GT was the exposure variable used to construct the weighted sample. The covariates listed in each row represent the variables included in the multivariate model.
Table 4.
Values are presented as median (interquartile range).
GT, granulocyte transfusion; FN, febrile neutropenia; ANC, absolute neutrophil count.
GT recipients demonstrated significantly faster clinical recovery, including reduced time to fever resolution, FN recovery, and ANC recovery compared with controls.
The Kaplan-Meier curves are shown in Supplementary Fig. 10.
Boldface indicates a statistically significant difference with P<0.05.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


