Article Contents
| Korean J Pediatr > Volume 56(12); 2013 |
Abstract
Purpose
Electroencephalography (EEG) is frequently ordered for patients with febrile seizures despite its unclear diagnostic value. We evaluated the prevalence of abnormal EEGs, the association between clinical findings and abnormal EEGs, and the predictive value of EEG for the recurrence of febrile seizures.
Methods
Data were collected on 230 children who were treated for febrile seizures at Kyung Hee University Medical Center from 2005 to 2009. EEGs were recorded after 1-2 days of hospitalization when children became afebrile. EEG patterns were categorized as normal, epileptiform, or nonspecific relative to abnormalities. The patients' medical records were reviewed, and telephone interviews with the families of the children were conducted to inquire about seizure recurrence. The relationships between clinical variables, including seizure recurrence, and EEG abnormalities were evaluated.
Results
Of the 131 children included, 103 had simple and 28 had complex febrile seizures. EEG abnormalities were found in 41 children (31%). EEG abnormalities were more common in children with complex than simple febrile seizures (43% vs. 28%), but the difference was not statistically significant. Logistical regression analysis showed that having multiple seizures in a 24-hour period was significantly predictive of abnormal EEG (odds ratio, 2.98; 95% confidence interval, 1.0 to 88; P=0.048). The frequency of recurrence did not differ significantly in the normal (31%) and abnormal (23%) EEG groups.
Febrile seizures (FS) are the most common convulsive events in children less than 5 years old, with an incidence of 2.5%1,2). Simple febrile seizures (SFS) are defined as primary generalized seizures lasting for less than 15 minutes without recurrence within 24 hours, whereas complex febrile seizures (CFS) are defined as focal, prolonged (>15 minutes) or recurrent within 24 hours. In general, FS are benign, with most children demonstrating complete recovery without any neurological complications, whereas CFS is associated with an increased risk of epilepsy (4.1%) in later years2). Predicting FS recurrence, as well as the development of epilepsy, is an important issue for clinicians.
According to American Academy of Pediatrics guidelines, electroencephalography (EEG) is not indicated after SFS in healthy children3). Previous studies have reported that EEG is of limited value in the evaluation of children with FS4,5). The reported incidence of EEG abnormalities in children with FS varies from 2% to 86%6,7). Although EEG abnormalities may be present in children with FS, their clinical significance is unclear. Furthermore, there is no consensus that EEG findings can be used to predict FS recurrence or epilepsy development. To clarify the relationship between EEG findings and FS, we assessed the prevalence of abnormal EEG findings in patients with SFS and CFS. We also evaluated the ability of clinical variables to predict abnormal EEG in children with FS, and whether abnormal EEG was a predictor of FS recurrence.
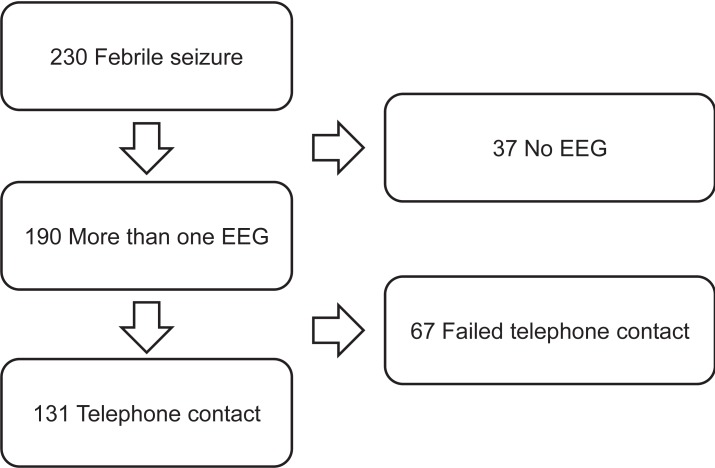
We retrospectively reviewed the clinical records of 230 children diagnosed with FS from September 2005 to July 2009 in the Department of Pediatrics at Kyung Hee University Medical Center. FS was defined as a seizure associated with body temperature ≥38℃ in the absence of central nervous system infection. Medical records were reviewed and telephone interviews were conducted with families of these children to inquire about seizure recurrence. Of these 230 patients, 193 neurologically normal patients had an EEG after FS; but 62 were excluded after failure of telephone contact. Thus, 131 patients were included in this study (Fig. 1). Children were diagnosed with CFS if they had experienced prolonged (>15 minutes in duration), focal, or repetitive (more than one seizure within 24 hours) seizures8). Patients with previous afebrile seizures, and those with acute or remote neurologic insults, were excluded.
All children with FS were hospitalized, with EEGs performed 1-2 days later, after they became afebrile; depending on individual circumstances, some patients underwent EEGs after discharge. Each EEG was interpreted independently by two pediatric neurologists. EEG findings were classified as normal, epileptiform, or nonspecific with regard to abnormalities. Paroxysms of spikes and spike-wave complexes, whether focal or generalized, were interpreted as epileptiform abnormalities (Fig. 2A). Nonspecific abnormalities included focal or generalized slow waves (Fig. 2B, C), generalized spike and waves (Fig. 2D), abnormal theta rhythms (Fig. 2E), and asymmetric backgrounds.
Groups were compared statistically using the chi-square test. All statistical analyses were performed using SPSS ver.12.0 (SPSS Inc., Chicago, IL, USA). A P value less than 0.05 was considered statistically significant.
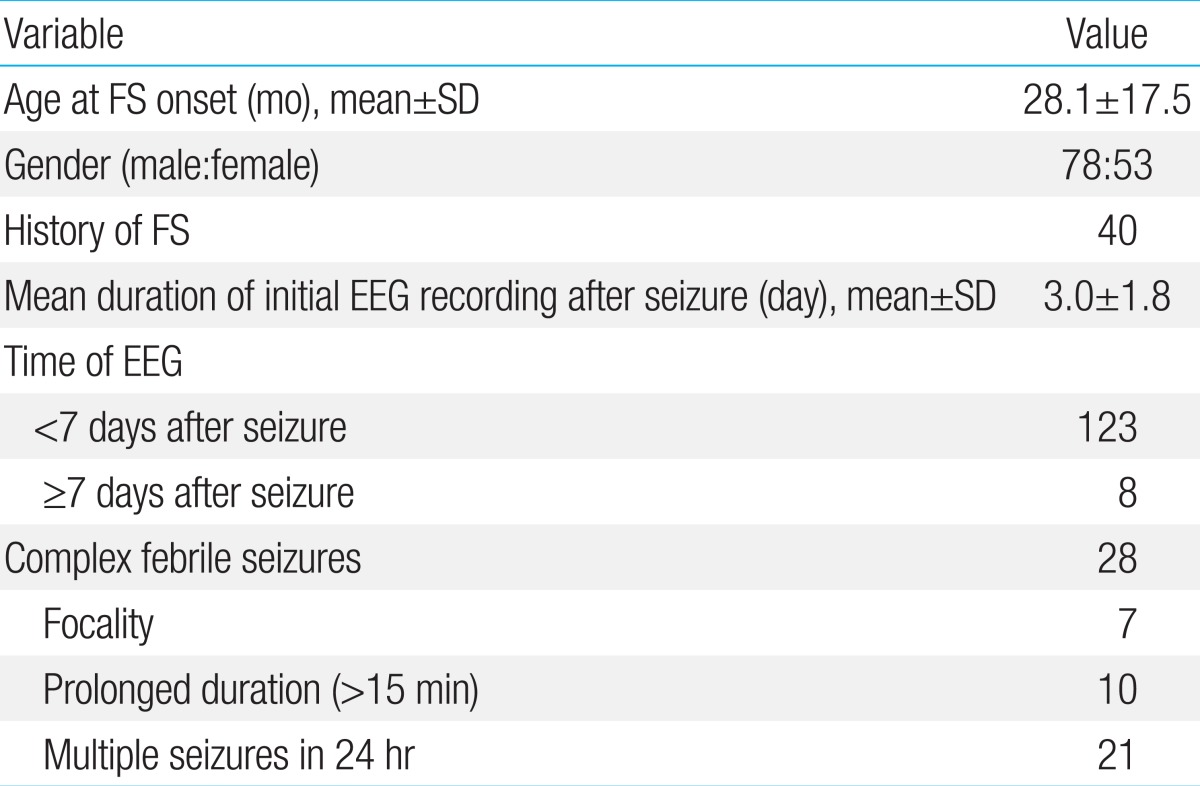
A total of 193 children met the inclusion criteria and underwent EEG. Of these, 131, including 78 males (59.5%) and 53 females (40.5%), were contacted by telephone. Mean patient age was 28.1±17.5 months, with 95 (73%) aged <36 months. The incident FS was the first in 91 children (69.5%), the second in 22 (16.8%), and the third or more in 18 (13.7%). Of these 131 patients, 103 (78.6%) were diagnosed with SFS and 28 (21.4%) with CFS. Clinical profiles of the 131 children are shown in Table 1.
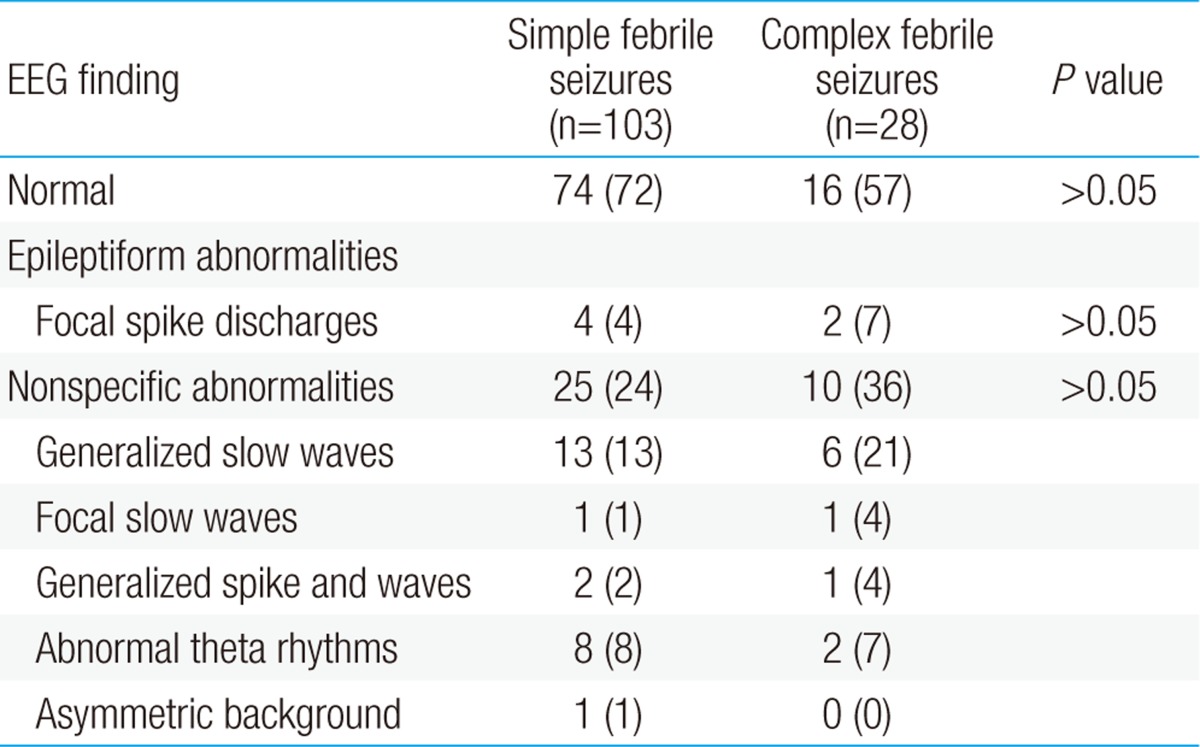
EEG recordings were performed an average 3.0±1.8 days after the FS and a mean 71.5 hours after the fever subsided. Initial EEGs were performed within 6 days after FS in 123 of the 131 children (94%). Background slowing were found in 34 (28%) in EEGs performed within 6 days, 1 (13%) in EEGs performed after 7 days. And epileptiform discharges were found in 5 (4%) in EEGs performed within 6 days, 1 (13%) in EEGs performed after 7 days. But the differences were not statistically significant (Table 2). EEG abnormalities were found in 41 children (31%), including 29 with SFS (28%) and 12 with CFS (43%). The most common EEG abnormalities were generalized slow waves, observed in 13 SFS (13%) and 6 CFS (21%) patients. Epileptiform discharges were observed in 6 children (5%), including 4 with SFS and 2 with CFS (Table 3). None of these differences between the SFS and CFS groups was statistically significant.
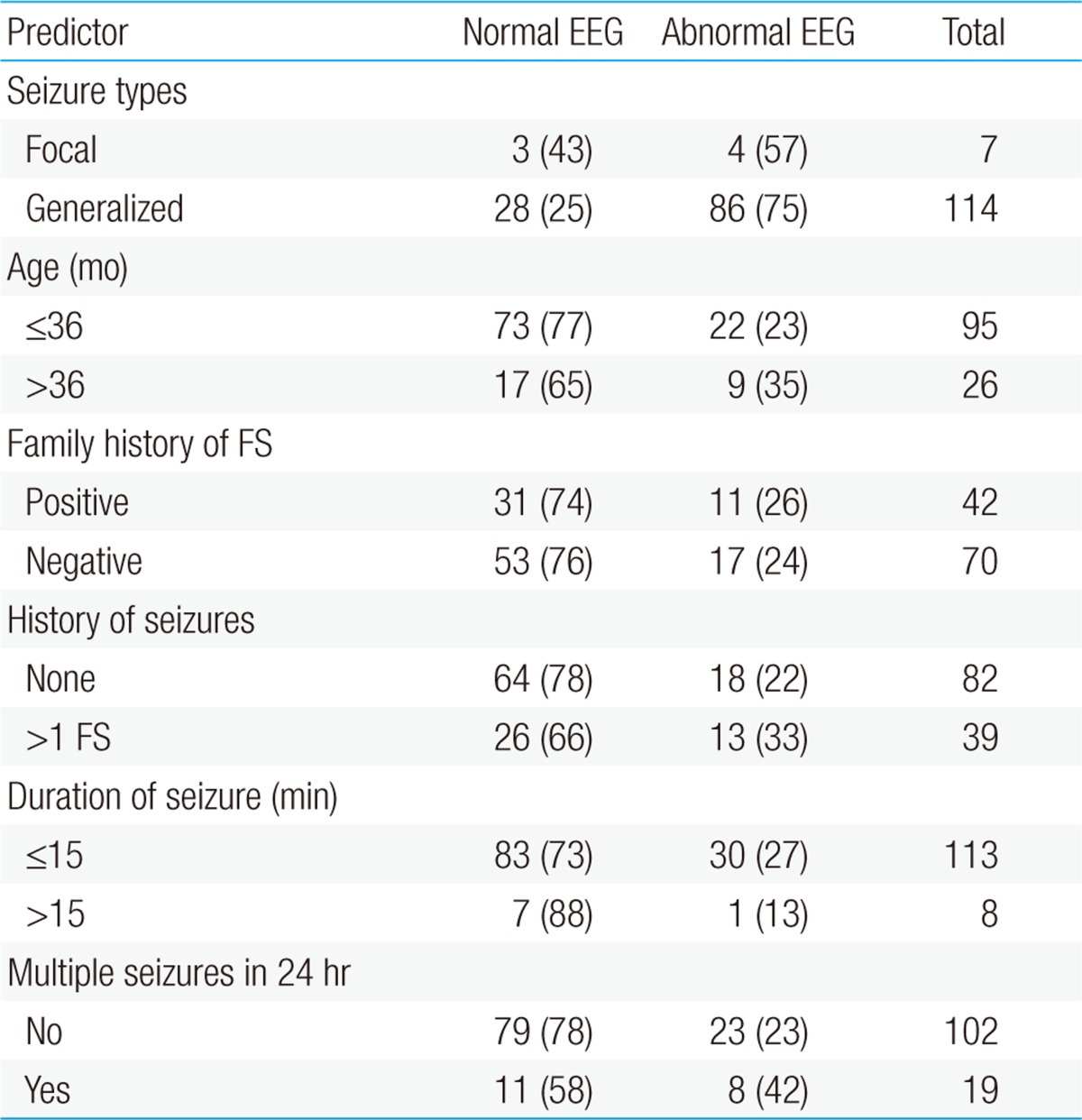
Clinical profiles of the normal and abnormal EEG groups with FS are shown in Table 4. Their clinical characteristics, including seizure type, age, family history of FS, previous seizure, duration, and seizure clustering, did not differ significantly in the normal and abnormal EEG groups. Multivariate logistic regression analysis showed that multiple seizures within a 24 hour period were significantly associated with an abnormal EEG (odds ratio [OR], 2.98; 95% confidence interval [CI], 1.0 to 88; P=0.048). None of the other factors, including age, seizure duration, and focality, was a significantly associated with abnormal EEGs.
Seizure recurrences were observed in 28 of the 90 patients in the normal EEG group (31%) and in 7 of 31 in the abnormal EEG group (23%). Among the latter, 3 had epileptiform discharges and 4 had nonspecific abnormalities (Table 5).
Epileptiform abnormalities on EEG were seen in one of five patients with two FS recurrences and in one of four with three recurrences. However, these findings were not statistically significant. FS recurrence was observed in 7 of 21 patients (33%) with multiple seizures within 24 hours and in 21 of 110 (26%) with single seizures, a difference that was not statistically significant.
Febrile seizures are benign in children and are usually associated with good prognosis. In general, an EEG is not recommended in the routine evaluation of a healthy child with SFS3). Despite a lack of convincing evidence regarding its benefit, EEGs are often performed by clinicians to evaluate FS, most often because of parental demand or to predict FS recurrence or epilepsy. The incidence of EEG abnormalities in children with FS has been reported to vary widely, from 2% to 86%9,10). This variability may be due to different definitions of EEG abnormalities, subject selection, and postseizure timing of the EEG. Because CFS is associated with increased risk of development of subsequent epilepsy, EEGs tend to be ordered more in these patients than in those with SFS. However, it is unclear whether EEG is sensitive enough to distinguish SFS from CFS. Although higher rates of paroxysmal discharges have been reported in children with CFS11), other studies have reported similar rates of paroxysmal abnormalities on immediate postictal EEGs of patients with SFS and CFS12,13). In our study, EEG abnormalities were found in 41 children (31%), 29 of 103 (28%) with SFS and 12 of 28 (43%) with CFS. Although the percentage of children with EEG abnormalities was slightly higher in the CFS group, the difference was not statistically significant.
EEG abnormalities in FS may depend on the timing of the EEG relative to the seizure. Epileptiform discharges are rarely reported and slowing is more prevalent within the first 7 days after FS14-16). For example, none of 33 patients with CFS showed evidence of paroxysmal abnormalities on EEG within one week after FS, whereas the true rate of abnormalities on early postictal EEGs in patients who have experienced CFS was calculated to be ≤8.6%15). Similarly, only 22.5% of EEG abnormalities were observed within 6 days of CFS, whereas EEGs performed 10 days after seizure were more likely to predict the development of epilepsy17). Of our 131 patients, 123 were evaluated by EEG within 6 days of seizure. The prevalence of background slowing was higher in EEGs performed earlier, and epileptiform discharges were more prevalent in EEGs performed after 7 days. Although the differences were not statistically significant, due to the small number of patients, these findings suggest that EEGs performed within 6 days reflect a postictal slowing effect after seizures.
We also investigated whether clinical variables are associated with abnormal EEGs in children with FS. We found that seizure type, age at onset, family history of FS, previous seizure, seizure duration, and seizure clustering did not differ between children with normal and abnormal EEGs, multiple logistic regression analysis found that multiple seizures within 24 hours were associated with EEG abnormalities. These findings differ somewhat from those of previous reports. For example, in children with CFS, the absence of family history of FS, age >3 years, and timing of the recording from ictus within 7 days were significantly associated with the likelihood of EEG abnormalities9). Similarly, in a study of 676 children with FS, focal seizures and seizures lasting >15 minutes were associated with abnormal EEG16). In addition, the number of previous FS and increased age at EEG were related to likelihood of paroxysmal EEG abnormalities. The discrepancies between these results and ours may be due to differences in timing of EEG and differences in patient selection. For example, in one study9), 66 children had EEGs within 7 days after seizures, whereas all patients in the other study16) had EEGs after 7 days. Considering the early timing of EEG in our study, the association between multiple seizures and EEG abnormalities was likely due to pronounced postictal effects in the patients.
FS recurrence is an important issue for parents and clinicians because it is a very frightening experience. Early age at onset, family history of FS and low temperature at the time of FS were reported predictors of FS recurrence18). A prospective study found that younger age at onset, recurrence within the same illness, frequent febrile episodes and maternal preponderance were significant prognostic markers for first FS recurrence19). However, the ability of EEG to predict FS recurrence or unprovoked seizures is likely low. For example, postictal EEG did not add useful information on the likelihood of FS recurrence20), and a study in 1991 found that EEGs in children with FS did not predict the later occurrence of febrile or afebrile seizures5). Despite not being practically useful, EEGs are frequently performed in children with FS. Although we found that a higher percentage of children with epileptiform discharges than those with normal EEGs experienced recurrent FS (50% vs. 31%), the difference was not statistically significant, likely because of the small number of patients. Nevertheless, our findings support previous results showing that EEGs in children with FS are generally not predictive of recurrence.
EEGs, however, may be useful in predicting subsequent epilepsy in some patients. A recent study found that focal epileptiform discharges were significantly more frequent in CSF patients who did than did not develop epilepsy (OR, 5.15)21). In addition, the risk of subsequent epilepsy was found to be higher (OR, 27.0) in 9 of 119 patients with CSF and frontal EEG paroxysms22). Prospective studies are therefore needed to assess the usefulness of EEGs in children with FS.
This study had several limitations, including its retrospective nature, the small patient sample size, and the early timing of EEG after FS. In real clinical practice, however, it is difficult to perform EEG after 10-14 days, when most patients have been discharged. We attempted to evaluate the clinical significance of early postictal EEGs performed in a real clinical setting.
In summary, we found that 31% of children with FS had EEG abnormalities, but the prevalence of these abnormalities did not differ significantly in groups of patients with SFS and CFS. Multiple seizures within 24 hours were associated with abnormal EEGs, but abnormal EEGs were not associated with FS recurrence.
These findings suggest that early postictal EEGs are of little predictive value in determining the risk of FS recurrence. Postictal slowing may have influenced our results because there was too little time for patients to manifest significant EEG findings given our study design. Therefore, we recommend that follow-up EEGs be performed in patients with early postictal EEG abnormalities.
References
1. Hauser WA, Kurland LT. The epidemiology of epilepsy in Rochester, Minnesota, 1935 through 1967. Epilepsia 1975;16:1–66.


2. Nelson KB, Ellenberg JH. Predictors of epilepsy in children who have experienced febrile seizures. N Engl J Med 1976;295:1029–1033.


3. Subcommittee on Febrile Seizures. American Academy of Pediatrics. Neurodiagnostic evaluation of the child with a simple febrile seizure. Pediatrics 2011;127:389–394.


4. Koyama A, Matsui T, Sugisawa T. Febrile convulsions in northern Japan: a quantitative and qualitative analysis of EEG and clinical findings. Acta Neurol Scand 1991;83:411–417.


5. Stores G. When does an EEG contribute to the management of febrile seizures? Arch Dis Child 1991;66:554–557.



6. Frantzen E, Lennox-Buchthal M, Nygaard A. Longitudinal EEG and clinical study of children with febrile convulsions. Electroencephalogr Clin Neurophysiol 1968;24:197–212.


8. Consensus statement. Febrile seizures: long-term management of children with fever-associated seizures. Pediatrics 1980;66:1009–1012.
9. Joshi C, Wawrykow T, Patrick J, Prasad A. Do clinical variables predict an abnormal EEG in patients with complex febrile seizures? Seizure 2005;14:429–434.


10. Thorn I. The significance of electroencephalography in febrile convulsions. Akimoto H, Kazamatsuri H, Seino M, Ward A, editors. Advances in epileptology: 13th Epilepsy International Symposium. New York: Raven Press, 1982;:93–95.
11. Yamatogi Y, Othahara S. Electroencephalographic aspects of febrile seizures. Jpn J Clin EEG 1983;25:401–408.
12. Rantala H, Uhari M, Tuokko H. Viral infections and recurrences of febrile convulsions. J Pediatr 1990;116:195–199.



13. Wo SB, Lee JH, Lee YJ, Sung TJ, Lee KH, Kim SK. Risk for developing epilepsy and epileptiform discharges on EEG in patients with febrile seizures. Brain Dev 2013;35:307–311.


14. Lennox-Buchthal MA. Febrile convulsions: a reappraisal. Electroencephalogr Clin Neurophysiol 1973;32(Suppl): 1–138.
15. Maytal J, Steele R, Eviatar L, Novak G. The value of early postictal EEG in children with complex febrile seizures. Epilepsia 2000;41:219–221.


16. Sofijanov N, Emoto S, Kuturec M, Dukovski M, Duma F, Ellenberg JH, et al. Febrile seizures: clinical characteristics and initial EEG. Epilepsia 1992;33:52–57.


17. Yücel O, Aka S, Yazicioglu L, Ceran O. Role of early EEG and neuroimaging in determination of prognosis in children with complex febrile seizure. Pediatr Int 2004;46:463–467.


18. Berg AT, Shinnar S, Darefsky AS, Holford TR, Shapiro ED, Salomon ME, et al. Predictors of recurrent febrile seizures: a prospective cohort study. Arch Pediatr Adolesc Med 1997;151:371–378.


19. Pavlidou E, Tzitiridou M, Kontopoulos E, Panteliadis CP. Which factors determine febrile seizure recurrence? A prospective study. Brain Dev 2008;30:7–13.


20. Kuturec M, Emoto SE, Sofijanov N, Dukovski M, Duma F, Ellenberg JH, et al. Febrile seizures: is the EEG a useful predictor of recurrences? Clin Pediatr (Phila) 1997;36:31–36.


Fig. 2
(A) Focal spike discharges from the right temporoparietal area in a 29-month-old boy who was having a simple febrile seizure (FS). FS recurrence was not observed over a 2-year follow-up period. (B, C). Generalized slow waves recorded the day after a febrile seizure in a 27-month-old girl. A follow-up electroencephalogram recorded 8 days later showed normal background activity. (D) Generalized spike and waves in a 51-month-old girl who experienced a second simple febrile seizure. The recurrence of a febrile or afebrile seizure was not noted until the age of 81 months. (E) Abnormal theta activities in an 11-month-old girl on the fourth day after a febrile seizure. The electroencephalogram was recorded during the waking state.
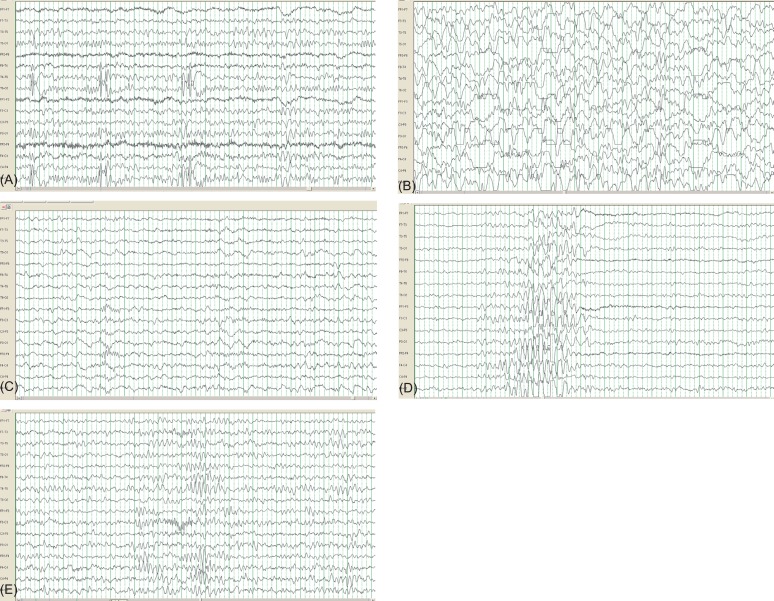
Table 3
Electroencephalographic characteristics of patients with simple and complex febrile seizures





 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


