Article Contents
| Clin Exp Pediatr > Volume 65(12); 2022 |
|
Abstract
Footnotes
Funding
The surveillance study of the Korean Childhood Community-Acquired Pneumonia Study Group (KoC-CAPS) was supported by a grant from the Korea Disease Control and Prevention Agency, Republic of Korea (grant number: 4800- 4821-304). This fund provides financial support in the investigation, design of the study, data collection, data analysis, and interpretation of data.
Acknowledgments
Fig.┬Ā1.
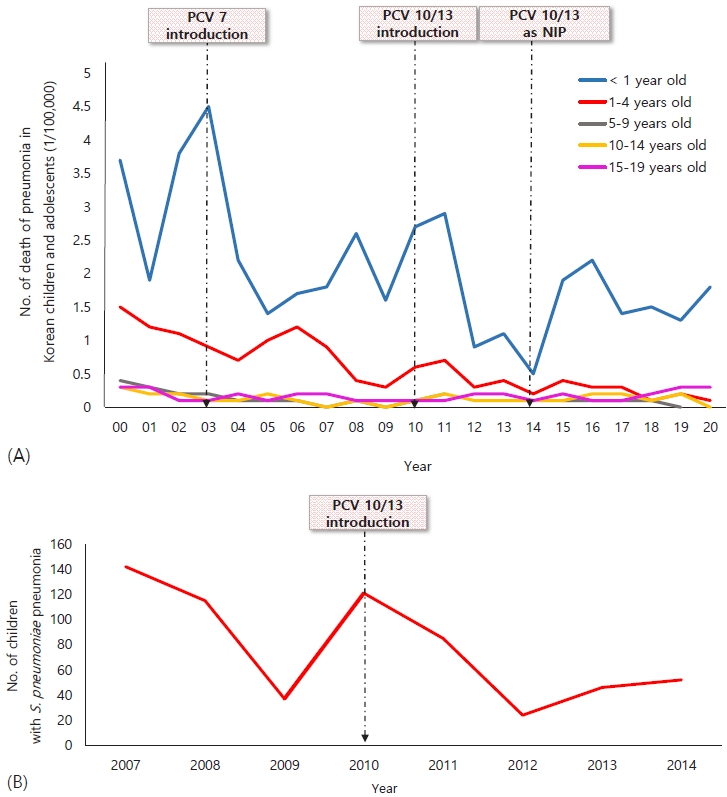
Fig.┬Ā2.
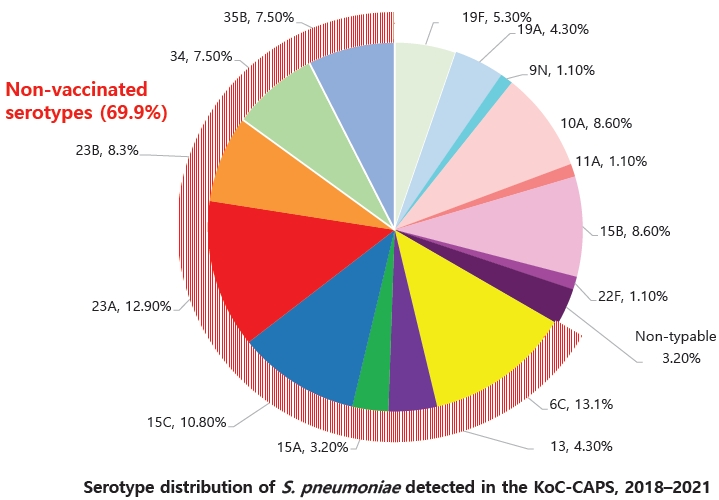
Fig.┬Ā4.
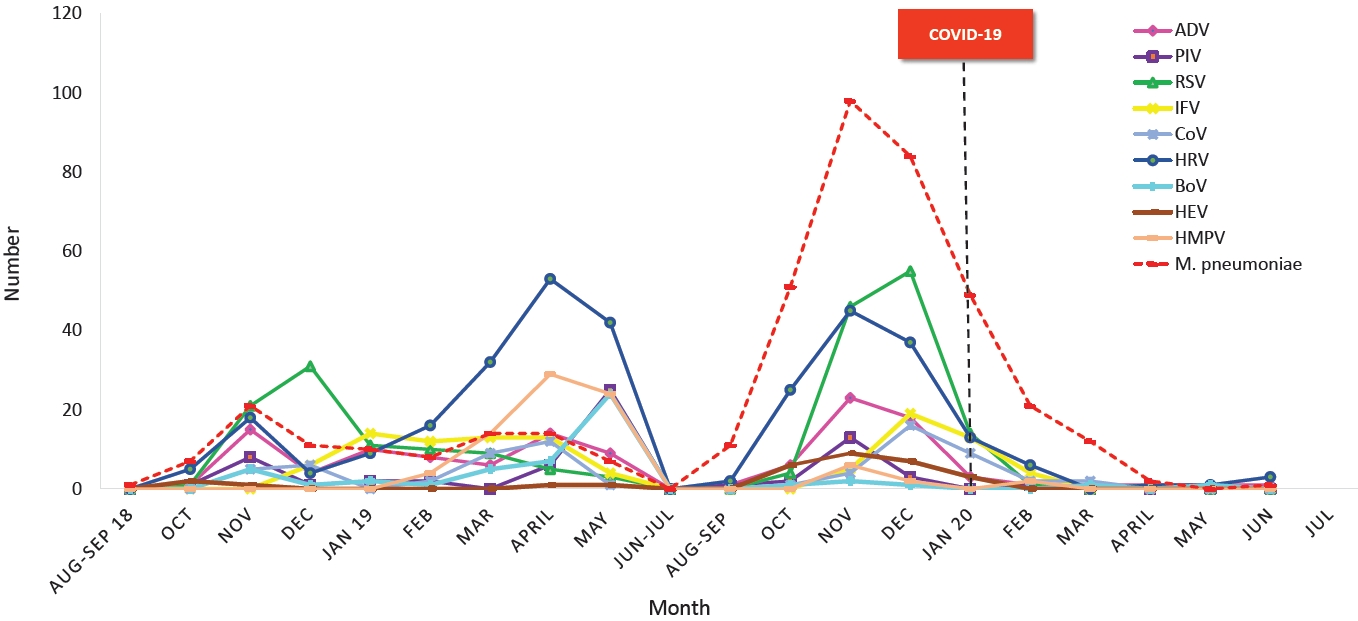
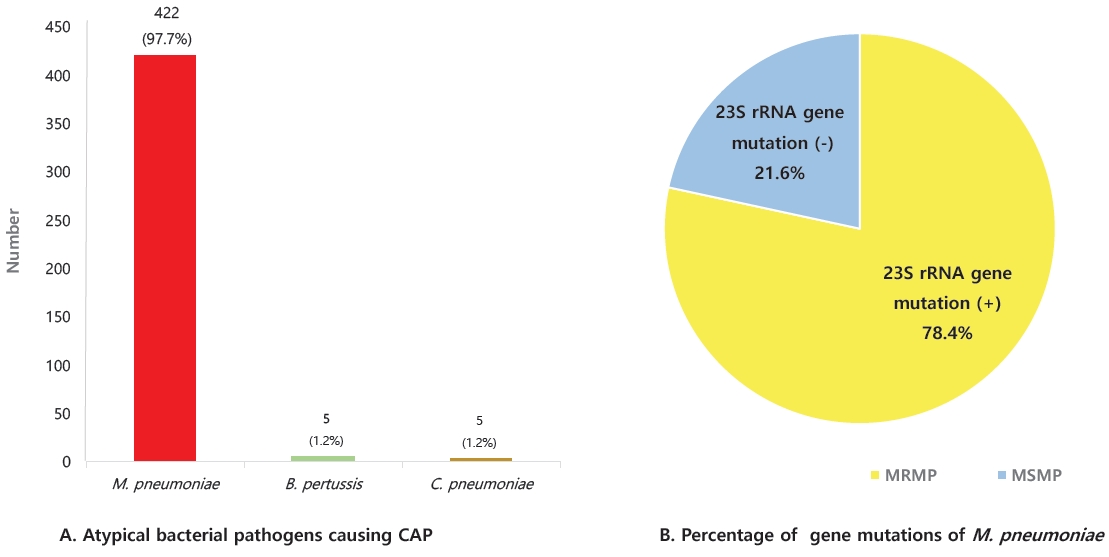
Fig.┬Ā5.
Table┬Ā1.
| Country | Study | No. of institutions | Origin of samples | Research period | No. of subjects | Age group (yr) | Population | Bacterial pathogen | Atypical bacterial pathogen | Viral pathogen |
|---|---|---|---|---|---|---|---|---|---|---|
| US | Jain et al. [12] | Multicenter (3 centers) | Naso/oropharyngeal swab | 2010ŌĆō2012 | 2,358 | 0ŌĆō18 | Hospitalized with CAP | 175 (8%) | 178 (8%) | 1,462 (66%) |
| BAL | S. pneumoniae (M/C) | M. pneumonia | RSV (M/C) | |||||||
| Sputum | ||||||||||
| PF | ||||||||||
| ET aspirate | ||||||||||
| Blood culture | ||||||||||
| Blood PCR | ||||||||||
| Singapore | Chiang et al. [48] | Single center | Sputum | For 3 yr | 1,702 | 0ŌĆō18 | Hospitalized with CAP | 175 (10.3%) | 350 (20.6%) | 94 (5.5%) |
| Nasopharyngeal aspirates | S. pneumoniae (M/C) | M. pneumoniae | RSV (M/C) | |||||||
| PF | ||||||||||
| Blood culture | ||||||||||
| Taiwan | Chi et al. [49] | Multicenter (8 centers) | Blood culture | 2010ŌĆō2013 | 1,032 | 0ŌĆō18 | Hospitalized with CAP | 326 (31.6%) | 233 (22.6%) | 180 (17.4%) |
| PF | S. pneumoniae (M/C) | M. pneumoniae | ADV (M/C) | |||||||
| Nasopharyngeal swab | ||||||||||
| China | Oumei et al. [50] | Blood culture | 2015 | 1,500 | 0ŌĆō18 | Hospitalized with CAP | 486 (32.4%) | 291 (33.5%) | ||
| Oropharyngeal swab | M. pneumoniae | RSV (M/C) | ||||||||
| Peru | del Valle-Mendoza et al. [51] | Single center | Nasopharyngeal swab | 2009ŌĆō2010 | 146 | 0ŌĆō18 | Hospitalized with CAP | 58 (39.7%) | 52 (35.6%) | |
| M. pneumoniae (M/C) | RSV (M/C) | |||||||||
| Australia | Bhuiyan et al. [52] | Multicenter | Nasopharyngeal swab | 2015ŌĆō2017 | 230/230 | 0ŌĆō17 | Hospitalized with CAP/attended clinic without URI | 19 (8.2%) | 130 (56.5%) | |
| M. pneumoniae | RSV (M/C) |
CAP, community-acquired pneumonia; BAL, bronchoalveolar lavage; PF, pleural fluid; ET, endotracheal; PCR, polymerase chain reaction; S. pneumoniae, Streptococcus pneumoniae; M. pneumoniae , Mycoplasma pneumoniae; M/C, most common; RSV, respiratory syncytial virus; ADV, adenovirus; URI, upper respiratory tract infections.
Table┬Ā2.
| Study | No. of institutions | Origin of samples | Research period | No. of subjects | Age group | Population | Pneumonia incidence | Bacterial pathogen | Atypical bacterial pathogen | Viral pathogen |
|---|---|---|---|---|---|---|---|---|---|---|
| Moon et al. [53] | 2 Centers | Nasopharyngealaspirates | 2000ŌĆō2001 | 796 | 0ŌĆō18 yr | Hospitalized with ALRI | 75 (36%) | 208 (26.1%) | ||
| ADV (Seoul) | ||||||||||
| RSV (Masan) | ||||||||||
| Choi et al. [54] | 2 Centers | Nasopharyngealaspirates | 2000ŌĆō2005 | 2,198 | 0ŌĆō5 yr | Hospitalized with ALRI | RSV (M/C) | |||
| Cheong et al. [57] | Single center | Nasopharyngealaspirates | 2005 | 654 | 0ŌĆō15 yr | Hospitalized with ALRI | 169 (72.2%) | 234 (35.4%) | ||
| ADV (M/C) | ||||||||||
| Eun et al. [59] | Single center | Serologic diagnosis | 1986ŌĆō2004 | 2,405 | 0ŌĆō18 yr | Diagnosed with CAP | 568 (23.6%) | |||
| M. pneumoniae | ||||||||||
| Kwon et al. [58] | Single center | Nasopharyngeal aspirates | 2002ŌĆō2006 | 3,854 | 0ŌĆō17 yr | Hospitalized with ALRI | 276 (73%) | 98 (35.5%) | ||
| PIV3 (M/C) | ||||||||||
| Chun et al. [55] | Single center | Nasopharyngeal aspirates | Nov 2007ŌĆōApril 2008 | 297 | 0ŌĆō5 yr | Hospitalized with ALRI | 222 (75%) | 120 (54%) | ||
| RSV (M/C) | ||||||||||
| Kim et al. [56] | 3 Centers | Nasopharyngeal aspirates | 2008ŌĆō2009 | 418 | 0ŌĆō5 yr | Hospitalized with ALRI | 225 (53.8%) | 154 (68.4%) | ||
| RSV (M/C) | ||||||||||
| Lee et al. [60] | Single center | Blood culture | 2006ŌĆō2008 | 288 | 0ŌĆō15 yr | Hospitalized with CAP (lobar/lobular form) | 27 (9.4%) | 146 (50.7%) | 17 (5.9%) | |
| Nasopharyngeal aspirates | S. pneumoniae (M/C) | M. pneumoniae | ||||||||
| Sputum | ||||||||||
| Urinary S. pneumoniae | ||||||||||
| Ag | ||||||||||
| Kim [61] | 2 Centers | Nasopharyngeal aspirates | 2010ŌĆō2011 | 1,520 | 0ŌĆō18 yr | Hospitalized with ALRI | 595 (52.3%) | 210 (67.7%) | ||
| RSV (M/C) | ||||||||||
| Lee et al. [62] | Multicenter (146 EDs) | 2012 | 38,415 | 0ŌĆō18 yr | Diagnosed with CAP from ED | 2,039 (5.3%) | 1,732 (4.5%) | 11,146 (29%) | ||
| M. pneumoniae | IFV (M/C) | |||||||||
| Shin et al. [63] | Multicenter (117 EDs) | 2007ŌĆō2014 | 329,380 | 1 moŌĆō18 yr | Diagnosed with CAP from ED | 4,316 (1.3%) | 12,635 (3.8%) | 27,607 (8.4%) | ||
| S. pneumoniae (M/C) | M. pneumoniae | IFV (M/C) | ||||||||
| An et al. [65] | Multicenter (5 centers) | Nasopharyngeal aspirates | 2015ŌĆō2016 | 428 | 0ŌĆō18 yr | Hospitalized with CAP | 298 (69.6%) | 261 (61%) | ||
| Blood culture | M. pneumoniae | RV, RSV | ||||||||
| Lee et al. [64] | Multicenter (23 centers) | Nasopharyngeal aspirates | 2010ŌĆō2015 | 30,944 | 0ŌĆō18 yr | Hospitalized with CAP | 9,183 (29.6%) | 16,895 (54.5%) | ||
| Blood culture | M. pneumoniae | RSV (M/C) | ||||||||
| Roh et al. [66] | Multicenter (27 centers) | Nasopharynx | 2018ŌĆō2020 | 1,023 | 1 moŌĆō18 yr | Hospitalized or ambula- tory with CAP | 264 (25.8%) | 432 (42.2%) | 65.70% | |
| Nasal swab | S. aureus (M/C) | M. pneumoniae (M/C)) | RSV (M/C) | |||||||
| Throat swab | ||||||||||
| Sputum | ||||||||||
| BAL | ||||||||||
| Transtracheal aspirates |
CAP, community-acquired pneumonia; ALRI, acute lower respiratory tract infection; ADV, adenovirus; RSV, respiratory syncytial virus; M. pneumoniae , Mycoplasma pneumoniae; PIV3, parainfluenza virus type 3; M/C, most common; S. pneumoniae , Streptococcus pneumoniae; EDs, emergency departments; IFV, influenza virus; BAL, bronchoalveolar lavage; S. aureus, Staphylococcus aureus.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


