Article Contents
| Clin Exp Pediatr > Volume 66(10); 2023 |
|
Abstract
Background
Purpose
Methods
Results
Conclusion
Supplementary materials
Supplementary Table 2.
Footnotes
Table 1.
Table 2.
| Risk factor | Source (yr) | No. of population | No. of included studies | Study design | Effect metrics | Random-effect summary estimate | Credibility of evidence |
|---|---|---|---|---|---|---|---|
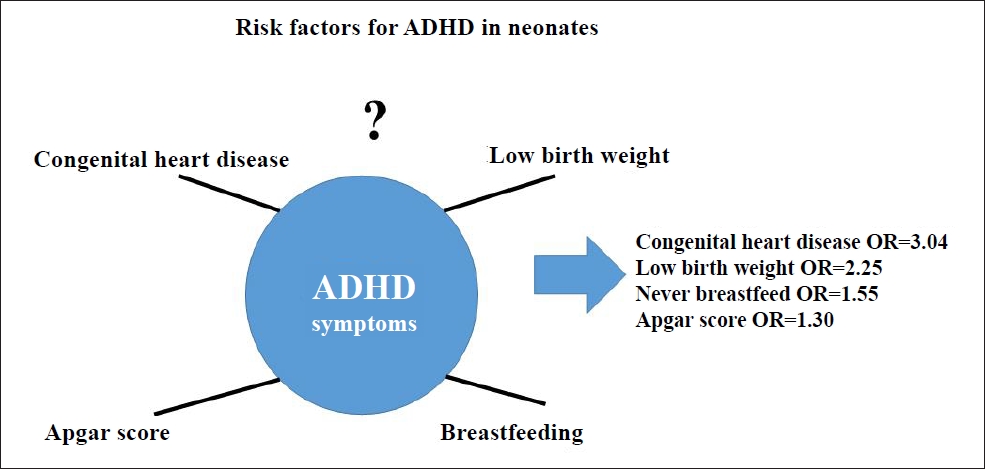
| Congenital heart disease | Jenabi et al. [8] (2022) | 21,551,625 | 5 | Case-control, cohort | Odds ratio | 3.04 (1.58–4.49) | Class IV |
| Neonatal intensive care unit admission | Bitsko et al. [6] (2022) | 166,891 | 11 | Case-control, cohort | Odds ratio | 1.50 (1.18–1.92) | Class III |
| Very low birth weight | Franz et al. [7] (2018) | 5,410 | 8 | Case-control, cohort | Odds ratio | 2.25 (1.56–3.26) | Class IV |
| Never breastfed | Bitsko et al. [6] (2022) | 13,229 | 9 | Case-control, cohort | Odds ratio | 1.55 (1.15–2.10) | Class IV |
| Extremely low birth weight | Franz et al. [7] (2018) | 1,345 | 4 | Case-control, cohort | Odds ratio | 4.05 (2.38–6.87) | Class IV |
| Apgar score <7 at 5 min | Bitsko et al. [6] (2022) | 8,189,263 | 15 | Case-control, cohort | Odds ratio | 1.30 (1.09–1.54) | Class IV |
Table 3.
| Study |
Item |
Rating | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 11 | 10 | 9 | 8 | 7 | 6 | 5 | 4 | 3 | 2 | 1 | ||
| Jenabi et al. [8] (2022) | C | Yes | No | No | No | Yes | No | C | Yes | Yes | No | 4/11 |
| Bitsko et al. [6] (2022) | C | Yes | No | No | No | Yes | Yes | C | C | No | No | 3/11 |
| Franz et al. [7] (2018) | C | Yes | No | No | No | Yes | No | No | Yes | Yes | Yes | 5/11 |
AMSTAR, a measurement tool to assess systematic reviews; C, can’t answer; NA, not applicable.
Item 1. Was an ‘a priori’ design provided? 2. Was there duplicate study selection and data extraction?.3. Was a comprehensive literature search performed? 4. Was the status of publication (i.e., grey literature) used as an inclusion criterion? 5. Was a list of studies (included and excluded) provided? 6. Were the characteristics of the included studies provided? 7. Was the scientific quality of the included studies assessed and documented? 8. Was the scientific quality of the included studies used appropriately in formulating conclusions? 9. Were the methods used to combine the findings of studies appropriate? 10. Was the likelihood of publication bias assessed? 11. The conflict of interest stated?





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


