Introduction
Menarche is an important event in the life of an adolescent girl, most of whom experience menarche before the age of 16 years. During this period, girls go through physical changes that are important in the psychological perception of sexual identity and that considerably influence mental maturity1-3). Historically, the age at menarche has gradually decreased, by about 4 months in every 10-year interval4). Over the last century, the age at menarche has fallen by 2 to 4 months every 10 years in the USA and Europe. Recently, mean age at menarche has stabilized at around 12 to 13 years of age, and has even increased in several countries, including Germany and the United Kingdom5,6). In Korea, age at menarche has declined, from 15 to 17 years in the 1960s to around 14 years in the 1980s, and to 12.8 years in the 1990s, because of consumption of an increasingly Westernized diet7). As age at menarche declines, the number of adolescents suffering from menstrual disorders such as dysmenorrhea and premenstrual syndrome has increased. Although 75% of girls in late puberty experience menstrual disorders, many hesitate to consult medical professionals, as they find open discussion of their problems embarrassing. Thus, such girls may not be adequately diagnosed or treated and may experience reproductive physiology problems during adulthood8). We therefore surveyed current changes in the age of menarche in Korean adolescents. We also evaluated general menstruation patterns, the incidence of common menstrual disorders, and the means used to cope with such problems.
Materials and methods
1. Subjects
The subjects of this study were 538 physically healthy, Korean, middle-high school students aged 14 to 18 years (mean age, 16.1 years) who visited our Pediatrics clinic in Seoul for health examinations between July and November, 2007.
Results
1. Change in age at menarche
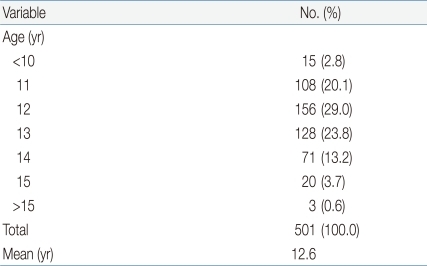
The mean age of all respondents was 16.1 years. Of the 538 subjects, 93.1% (n=501) reported that they had experienced menarche, 1.1% (n=6) reported they had not, and 5.8% (n=31) could not remember. Age at menarche was 12 years in 29.0% of respondents (n=156), 13 in 23.8% (n=128), 11 in 20.1% (n=108), and 14 in 13.2% (n=71). Mean age at menarche was 12.6 years (Table 1).
2. Menstruation pattern
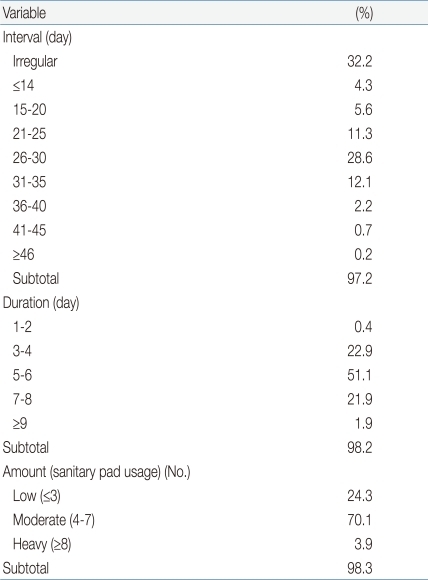
Of all respondents, 32.3% (n=173) reported that the menstrual cycle was irregular. Cycle length was 26 to 30 days in 28.6% (n=154), 21 to 25 days in 11.3% (n=61), 31 to 35 days in 12.1% (n=65), and 15 to 20 days in 5.6% (n=30). The duration of menstruation was 5 to 6 days in 51.1% (n=275), 3 to 4 days in 22.9% (n=123), and 7 to 8 days in 21.9% (n=118). When we assorted respondents by the number of pads used per day, with menstrual flow defined as "small", "moderate" and "large" upon use of ≤3, 4-7, and ≥8 pads, respectively, we found that 70.1% (n=377) of subjects had "moderate" menstruation (Table 2).
3. Premenstrual syndrome
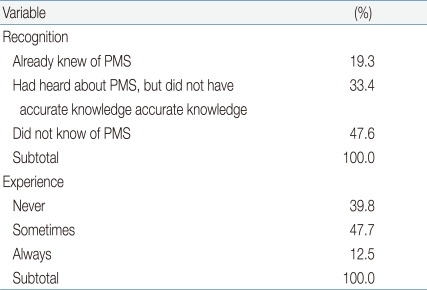
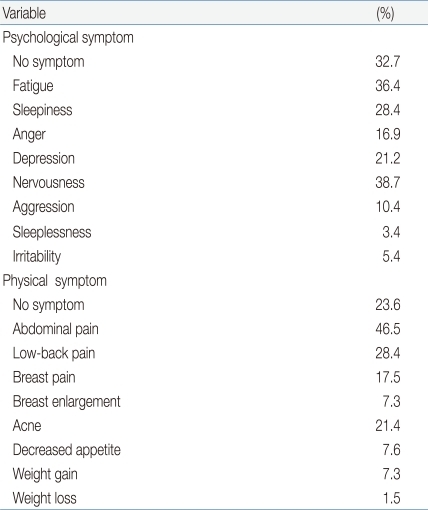
We found that 47.6% (n=256) of respondents had not heard of premenstrual syndrome, 32.3% (n=174) were familiar with the syndrome but lacked accurate knowledge thereof, whereas 19.3% (n=104) were well-informed. In contrast, when subjects were individually asked whether they had experienced symptoms of premenstrual syndrome, 47.7% (n=251) reported such events occasionally, 12.5% (n=67) replied that they always experienced symptoms, and 39.8% (n=214) said they had never experienced problems (Table 3). The most common psychological symptoms of premenstrual syndrome were nervousness (38.7%, 208 subjects), fatigue (36.4%, n=196), and sleepiness (28.4%, n=153); although 32.7% of subjects (n=176) reported no such symptoms. The most frequent physical symptoms of premenstrual syndrome were abdominal cramps (46.5%, 250 subjects), low back pain (28.4%, n=135), acne (21.4%, n=115), and breast pain (17.5%, n=94); although 23.6% of subjects (n=127) reported no such symptoms (Table 4).
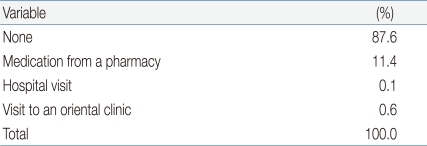
When subjects were asked how they coped with symptoms, 87.6% (n=276) reported that they did not access medication or treatment, and only 11.4% (n=36) took over-the-counter medications, such as pain killers, purchased from a pharmacy. Only 0.1% of subjects (n=1) visited a hospital and 0.4% (n=2) attended an Oriental clinic (Table 5).
4. Dysmenorrhea
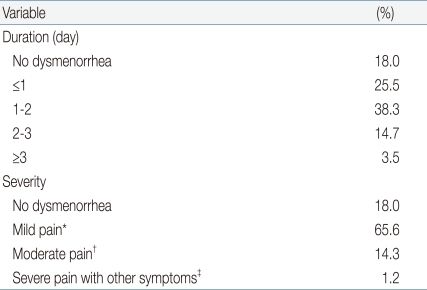
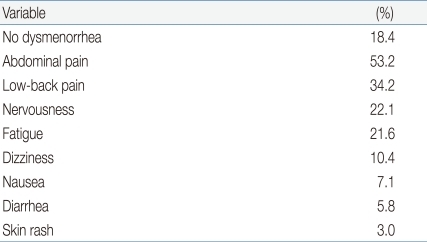
We found that 82% (n=435) of female students reported dysmenorrhea, 1 to 2 days in duration in 38.3% (n=206), ≤24 hours in 25.5% (n=137), 2 to 3 days in 12.5% (n=67), and ≥3 days in 3.5% (n=19). In most subjects, the principal symptom was mild pain that did not impact on normal daily life; painkillers were not needed (65.6%, n=353). However, 0.9% of subjects (n=5) had severe pain not relieved by pain killers, and other symptoms (Table 6). The most common dysmenorrhea problems were abdominal cramp (53.2%, 286 subjects), low-back pain (34.2%, n=184), fatigue (21.6%, n=116), and nervousness (22.1%, n=119) (Table 7).
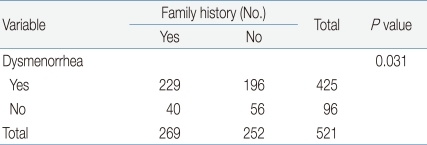
When we examined the relationship between dysmenorrhea and a family history thereof, we found that 53.9% of subjects (n=229) had dysmenorrhea shared with a family member (mother or sister), whereas 46.1% (n=196) did not have such a family member (Table 8). Statistical analysis showed a significant correlation between present dysmenorrhea and a family history thereof (P<0.05).
Discussion
Age at menarche is affected by various factors, including genetic and socio-economic parameters, nutritive state, psychological variables, individual physical structure, geography, weather, disease, and exercise, and may be a practical indicator of the maturity and health of both individuals and groups9,10). For example, age at menarche has been found to be lower in tropical than in temperate regions11). Menarche has been reported to be more frequent in summer and winter than in spring and autumn12), and to occur more commonly in spring and summer than in autumn and winter13).
However, reports on associations between age at menarche and season have not yielded consistent results. Compared with ordinary students, age at menarche has been found to be generally later in students (ballerinas or athletes) who engage in intensive physical activity during childhood, and earlier in students under psychological stress resulting from family discord or placement in an orphanage14). In addition, age at menarche has been reported to be closely correlated with parental educational level and occupation, being delayed by about 8 months when parents are poorly educated (unschooled, or mere high school graduates) compared to parents who are better educated (2-year college or higher-level graduates), and by about 6 months when parents are unemployed, or farmers, compared to parents working as civil servants or teachers15).
Recently, however, age at menarche has fallen regardless of region or race because of improvements in nutrition and physical growth, and changes in the socio-economic environment16). Thus, age at menarche is not fixed, but rather varies among population groups and over time17). For example, surveys of English adolescents in 1940 to 1960, during and after World War II, showed that age at menarche decreased by 3 to 4 months every 10 years18). In Western countries, age at menarche has fallen by 2 to 3 months every 10 years over the last 150 years19,20). In the USA and Europe, age at menarche was 17 years in the 19th century, 14.7 years in 187721), and 12.2 to 12.9 years in recent years22-24).
In Korea, research on the age of menarche commenced in 192325), and has continued since that time. Mean age at menarche has generally decreased; from 14.8 years in 1962 to about 14 years in 1973, 14.4 years in 1979, 13.4 years in 1986, 12.5 years in 1988, and 12.4±1.1 years in 199326). Regionally, age at menarche was 1 year earlier in Seoul than in other regions of Korea27). Age at menarche was 16.2 years in urban women and 16.3 years in rural women28), and 13.4 years in women living in large cities and 13.7 years in those residing in small cities or rural areas29). We found that the mean age at menarche was 12.6 years, confirming the recent trend toward earlier menarche in Koreans. Also, age at menarche did not differ greatly from that in European nations or the USA. However, our sampling area was confined to Seoul, and our results may thus not be representative of the entire Korean population.
When we surveyed general menstruation patterns, we found that, contrary to our expectations, many female students had irregular menstrual cycles. Moreover, increasingly earlier menarche was associated with an elevated prevalence of menstrual disorders including dysmenorrhea and premenstrual syndrome, and with the severity of these conditions. Dysmenorrhea, evident at the beginning of menstruation or just prior to menstruation, is categorized by presence of lower abdominal pain accompanied by gastrointestinal and neurological symptoms. We found that most students experienced dysmenorrhea during menstruation, although pain was mild. However, about 15% of students complained of severe symptoms, requiring use of painkillers and/or absence from school. In contrast, a previous study30) found that only 4% of subjects suffered disrupted school attendance because of dysmenorrhea, although another report found that 16% of subjects were in this category31). Premenstrual syndrome, another common menstrual disorder that usually features mood swings occurring about 1 week before menstruation, is experienced by most menstruating women, particularly those 20 to 40 years of age. The symptoms vary among individuals and may differ from cycle to cycle. The principal problems include headache, nausea, weight gain, breast tenderness, abdominal distention, fatigue, change in appetite, anxiety, irritation, and depression. These symptoms usually appear 7 to 10 days before menstruation and disappear several hours after commencement of menstruation8). The symptoms are subjective, and diagnostic tools including a premenstrual assessment form (PAF) have been formalized, but it continues to be difficult to objectively diagnose the syndrome because of the absence of accurate criteria. The PAF is composed of 10 questions addressing mood swing, water retention, and pain. Each question is answered on a 6-point scale ("no change," "very little change," "mild change," "moderate change," "severe change," and "extreme change"), with a higher score indicating more severe disturbance. Dysmenorrhea has been associated with premenstrual syndrome32), although the mechanism of interaction has not been determined. Other menstrual disorders include abnormal and dysfunctional uterine bleeding. As such problems were not explored in our survey, additional research on menstrual disorders common among adolescents is required.
We found that the major symptoms of premenstrual syndrome were nervousness, depression, inertia, abdominal cramps, and lower abdominal pain, similar to previous findings33). We also surveyed how our adolescent students perceived and coped with premenstrual syndrome. We found that more than half experienced premenstrual syndrome but could not accurately self-diagnose the condition, and some students did not recognize that they were experiencing the symptoms. Moreover, whereas many students suffered from premenstrual syndrome, only 0.1% consulted medical specialists; most simply endured the problems. This suggests that systematic approaches to treatment of menstrual disorders in adolescence should be developed, featuring a detailed clinical history-taking, counseling, and a treatment plan.
Although adolescent menstrual disorders have been actively studied in Korea, only a few reports have assessed the association between present dysmenorrhea and a family history thereof. When we explored this association, we found that 53.9% of students who had a family member (mother or sister) with dysmenorrhea also experienced the condition; this rate was higher than the values of 51.1%30) and 41.1%34) previously reported. Moreover, we found that the correlation between present dysmenorrhea and a family history thereof was statistically significant (P<0.05). Thus, a detailed family history of dysmenorrhea must be taken when a systematic therapeutic approach to dysmenorrhea in adolescents is envisaged.
Our study had several limitations. First, all study subjects were middle-high school students in Seoul, and our results thus cannot be automatically generalized to students in other regions of Korea. A large-scale nationwide survey is required, and data on age at menarche by birth cohort are needed. Second, premenstrual syndrome includes both physical and psychological symptoms evident before menstruation begins; however, most study subjects were not well acquainted with premenstrual syndrome per se. This means that our survey of symptom memory may have been influenced by information bias. In other words, those who did not understand "premenstrual syndrome" may not have given valid responses to the relevant questions.
Our study was cross-sectional in design, and the data are useful to aid understanding of current changes in age at menarche among Korean girls. Further, we canvassed the methods by which adolescent students coped with menstrual disorders. Our results suggest that pediatricians should accurately explain the normal menstrual cycle to adolescent patients, take detailed clinical histories, and perform physical examinations and order appropriate tests to determine causes of menstrual disorders in such patients, so that problems may be appropriately treated.



 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


