Article Contents
| Clin Exp Pediatr > Volume 67(6); 2024 |
|
Abstract
Fig. 1.
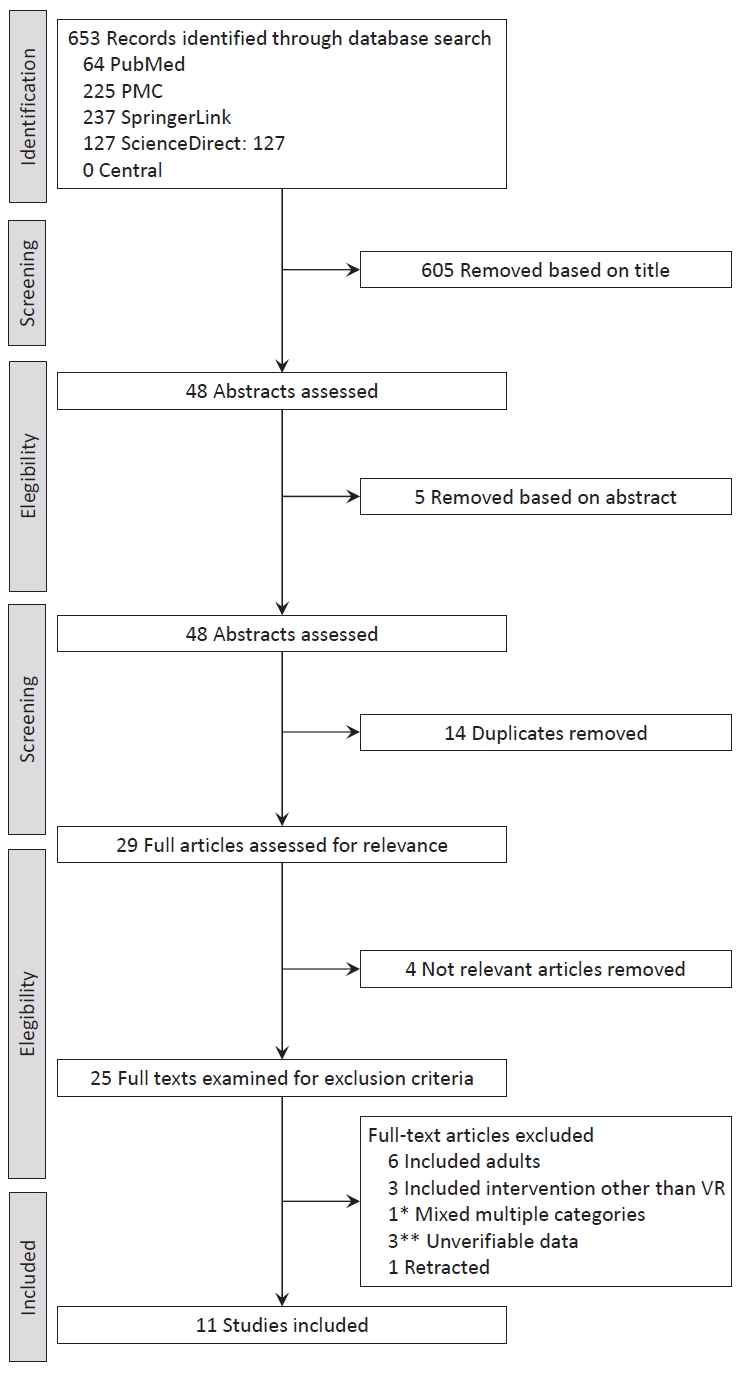
Table 1.
| Focus of the meta-analysis | Study | Question 1. | 2. | 3. | 4. | 5. | 6. | 7. | 8. | Score (maximum 8) |
|---|---|---|---|---|---|---|---|---|---|---|
| Cerebral palsy | Zhang et al. [10] | Yes | Yes | Yes | Yes | Unclear | Yes | Yes | Yes | 7 |
| Abdelhaleem et al. [9] | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes | 7 | |
| Ghai and Ghai [11] | Yes | No | Yes | Unclear | Yes | Yes | Yes | Yes | 6 | |
| Liu et al. [12] | Yes | Yes | No | Yes | Yes | Yes | Unclear | Yes | 6 | |
| Attention deficit hyperactivity disorder | Romero-Ayuso et al. [16] | Yes | Yes | Yes | Yes | Unclear | Yes | Yes | Yes | 7 |
| Parsons et al. [18] | Yes | Yes | Yes | Yes | Unclear | Yes | Yes | Yes | 7 | |
| Burns | Lauwens et al. [21] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 8 |
| Preoperative anxiety | Simonetti et al. [23] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 8 |
| Koo et al. [6] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 8 | |
| Needles | Czech et al. [24] | |||||||||
| Yes | No | Yes | Yes | Unclear | No | Yes | Yes | 5 | ||
| Saliba et al. [25] | Yes | Yes | Yes | No | Yes | Yes | No | Yes | 6 |
Question 1: Is a review based on focused questions adequately formulated and described?
Question 2: Were the eligibility criteria for included and excluded studies predefined and specified?
Question 3: Did the literature search strategy use a comprehensive, systematic approach?
Question 4: Were the titles, abstracts, and full-text articles dually and independently reviewed for inclusion and exclusion to minimize bias?
Question 5: Was the quality of each included study rated independently by 2 or more reviewers using a standard method to appraise internal validity?
Question 6: Were the included studies listed along with the important characteristics and results of each study?
Question 7: Was publication bias assessed?
Question 8: Was heterogeneity assessed? (This question applies only to meta-analyses.)
Table 2.
| Focus of the meta-analysis | Study |
Title |
Abstract |
Introduction |
Methods |
Results |
Discussion |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Meta-analysis? | Objectives | Data sources | Reviewer methods | Results | Conclusion | Explicit problem | Searching | Selection | Validity assessment | Data abstraction | Study features | Quantitative data synthesis | Trial flow (flowchart figure) | Study features | Quantitative data synthesis | Summary | Scores (maximum 18) | ||
| Cerebral palsy | Chen et al. [8] | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 17 |
| Zhang et al. [10] | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | 16 | |
| Abdelhaleem et al. [9] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 18 | |
| Ghai and Ghai [11] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | 17 | |
| Liu et al. [12] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 18 | |
| ADHD | Romero-Ayuso et al. [16] | Yes | Yes | No | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 16 |
| Parsons et al. [18] | Yes | Yes | No | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 16 | |
| Burns | Lauwens et al. [21] | Yes | Yes | No | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 16 |
| Operations | Simonetti et al. [23] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 18 |
| Koo et al. [6] | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 17 | |
| Needles | Czech et al. [24] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 18 |
| Saliba et al. [25] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | 18 | |
Table 3.
| Study | Study number | Age range (yr) | Inclusion criteria | Exclusion criteria | Outcomes | Result |
|---|---|---|---|---|---|---|
| Zhang et al. [10] | 17 RCTs | 2.8±0.86 to 11.8±2.4 | Children with CP aged 0 to 18 years | Participants who had severe cognitive impairment, hearing, or visual problems. | Activities of daily living | Virtual motor training had significant positive effects on activities of daily living (Hedge g=0.31; 95% CI, 0.10–0.51), grip (g=0.40; 95% CI, 0.08–0.71), gross motor function (g=0.71; 95% CI, 0.43–0.99). |
| 853 Participants | Comparator: usual care or conventional therapy. | Hand grip | ||||
| Outcomes: activities of daily living, hand grip, gross motor function. | Gross motor function | |||||
| Study design: RCT | ||||||
| Abdelhaleem et al. [9] | 19 RCTs | Experimental group: 7.05 to 11.67 | Children with any form of CP aged 3 to 18 years | Many publications of the same study reporting the same results | Fine motor function | VR seems to be effective for improving fine motor coordination. |
| 645 Participants | Control group 7.25 to 12.4 | Intervention: virtual reality therapy alone or combined with other intervention or within the setup of any other modality | Adults or patients suffering from any disability rather than CP | Gross motor function | Questionable effect on gross motor coordination. | |
| VR is not the intervention of choice. | PEDro scale is fairly correlated with Cochrane | |||||
| Comparator: no, placebo or routine physiotherapy treatment. Outcome measure: motor coordination | VR is used as an evaluative tool not a therapeutic modality. | RoB | ||||
| Robots are used as an orthosis only and not as VR component. | ||||||
| Study design: full-text RCTs | Single session intervention is used. | |||||
| Not published in English due to shortage of translation resources. | ||||||
| Ghai and Ghai [11] | Review: 16 studies (5 RCTs+11 CCTs) | 6 to 16.1±4.1 | Children with CP aged 6 -18 years | None mentioned. | Gait velocity | 88% of the 16 studies reported significant enhancements in gait performance after training with virtual reality. |
| 274 Participants. | Intervention: virtual reality (any training duration and setting) | Stride length | ||||
| Meta-analysis: 14 studies | Outcomes: spatiotemporal gait parameters, gross motor function and/or performance | Cadence | Meta-analyses revealed positive effects of virtual-reality training on gait velocity (Hedges g=0.68), stride length (0.30), cadence (0.66), gross motor function measure (0.44). | |||
| Peer-reviewed publications, with a ≥4 PEDro score | Stride width | |||||
| Language: English, German, Hindi, Punjabi, and Sanskrit languages | Gross motor function | Subgroup analysis for gait velocity reported a training duration of 20–30 min per session, ≤4 times per week across ≥8 wk to allow maximum enhancements | ||||
| Liu et al. [12] | Review: 18 RCTs | 4 to 18 | Children with CP aged no more than 18 years | Studies including patients with comorbidities. | Balance function | VR therapy has a positive effect on improving balance function in children with CP. |
| 474 Patients. | Intervention: VR as a therapeutic tool were assessed using a balanced outcome measure. | VR as an intervention supplement. | At least 20 min per session, twice a week for 6 wk or more of regular VR therapy is more effective for improving balance function in children with CP. | |||
| Meta-analysis: 16 RCTs | Outcomes: effects on balance measured by motor performance scales or test instruments used pre- and post-VR | Studies including other interventions that affected VR balance and performance data. | ||||
| 470 Patients. | Study design: RCTs | Case reports, non-peer-reviewed publications, conference abstracts. | ||||
| Language: English |
Table 4.
| Study | Study number | Age range (yr) | Inclusion criteria | Exclusion criteria | Outcomes | Result |
|---|---|---|---|---|---|---|
| Romero-Ayuso et al. [16] | Review: 6 studies | 8 to 18 | Children or adolescents with ADHD | No VR-based interventions or AR | Cognitive performance (omissions, commissions, correc t hits, perceptual sensitivity) | Improvements through VR in attentional vigilance, increased number of correct responses, decreased number of errors of omission. |
| Meta-analysis: 4 studies | Mean 12.9 | VR-based interventions | Case series, RCT protocols or clinical-trial protocols that did not present results or that focused on the application of VR exclusively to the educational field as a method of teaching a subject. | |||
| 125 Participants. | Study design: RCT or nonrandomized CCTs | No improvements in impulsivity responses. | ||||
| Language: English or Spanish | ||||||
| Parsons et al. [18] | Population comparison for VR (3D)- CPT=8 studies | 8 to 14 | Studies that utilized a virtual reality classroom | No report of interval data, ratio data and/or attention-symptom data. | Cognitive performance (omissions, commissions, hit reaction times) | The virtual classroom CPTs reliably differentiate attention performance in persons with ADHD |
| Population comparison for 2D-CPT= 6 studies | Language: English. | Insufficient report of study results (e.g., no means and standard deviations) to allow for effect size computation. | ||||
| Intervention studies, conference presentations, dissertations |
Table 5.
| Study | Study number | Age range (yr) | Inclusion criteria | Exclusion criteria | Outcomes | Result |
|---|---|---|---|---|---|---|
| Lauwens et al. [21] | Review: 8 studies 142 participants. | 2 to 18 | Children with burns | Studies where results of children and adults were pooled and remained indistinguishable or with missing data. | Pain during dressing changes | Adjuvant full immersive VR significantly reduces pain experienced during dressing changes in children and adolescents with burns |
| Meta-analysis: 4 studies | Primary research papers and those published in peer-reviewed journals | Other kind of wound care besides burns | ||||
| 104 Participants. | Intervention: immersive VR | Nonimmersive VR, semi-immersive VR. Other distraction interventions | ||||
| Language: English or Dutch |
Table 6.
| Study | Study number | Age range (yr) | Inclusion criteria | Exclusion criteria | Outcomes | Result |
|---|---|---|---|---|---|---|
| Koo et al. [6] | Meta-analysis: 5 RCTs | 5 to 8.3 (mean ages) | Patients were to undergo surgery. | Not RCT. No full-length article | Preoperative anxiety | VR lowered anxiety in children. |
| 449 Participants | VR before surgery in intervention group | |||||
| Study design: RCTs | ||||||
| Simonetti et al. [23] | Review: 7 RCTs | 4 to 12 | Children under 19 | Adults or neonates | Preoperative anxiety | VR lowered anxiety in children undergoing surgery |
| 602 Participants | Intervention/outcome: VR to reduce perioperative anxiety | Cognitive impairments | ||||
| Meta-analysis: 6 RCTs | Nonoperative setting | |||||
| Study design: RCTs | Use of anxiolytics | |||||
| 562 Participants | Anxiety not primary or secondary outcome | |||||
| Grey literature |
Table 7.
| Study | Study number | Age range (yr) | Inclusion criteria | Exclusion criteria | Outcomes | Result |
|---|---|---|---|---|---|---|
| Czech et al. [24] | 6 RCTs | 5 to 18 | Intervention: use of VR in needle procedures | None mentioned. | Primary outcomes: pain, anxiety, fear | Significant reduction in pain in studies using the Wong-Baker scale. Nonsignificant reduction in pain in studies using the Faces Pain Scale-Revised |
| 554 Participants | Outcomes: pain or fear/anxiety | Secondary outcomes: changes in blood pressure and heart rate, satisfaction | The data for fear was not pooled due to large heterogeneity and different scales. | |||
| Study design: RCTs | ||||||
| Saliba et al. [25] | 9 RCTs | 4 to 12 | Patients 4 to 12 years old | Patients under 4 or over 12 years old | Primary outcome: pain | VR reduced pain and fear/anxiety in children undergoing vascular access procedures |
| 930 Participants | Intervention: VR used as distraction | Secondary outcome: fear and anxiety | ||||
| Comparators: no distraction, any other form of distraction or standard care | Nonvascular access procedures | |||||
| Outcomes: pain as primary and possibly fear/anxiety as secondary | Non-RCTs | |||||
| Study design: RCTs |





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


